21. Blood Gas Interpretation
1/46
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
47 Terms
maintenance of constant conditions through dynamic equilibrium of internal environment of the body via hydrogen ions
what is homeostasis
lungs, kidneys, liver, GI
what organs regulate homeostasis
carnivores
____ produce CO2 and excess H+ precursors
herbivores
____ produce CO2 and excess HCO3- precursors
1. chemical: rapid first line of defense
2. respiratory: change H+ and pCO2 in minutes to hours
3. renal: excrete H+ w/in hours to days
what are the 3 principal mechanisms to buffer H+
buffer bicarbonate within seconds
the extracellular compenent of chemical buffers...
buffer phosphate, hemoglobin, and proteins in 2-4hrs
the intracellular component of chemical buffers...
reviewed
review the four primary disturbances and expected compensatory responses

respiratory/lungs
if there is a metabolic/kidney problem, there is _____ compensation

metabolic/kidneys
if there is a respiratory/lung problem, there is ____ compensation

1. assess pH as acidotic or alkalosis
2. is the primary change/abnormalities CO2/respiratory or HCO3/metabolic
3. consequences and how it affects tx.
4. assess whole blood gas
how do you approach a blood gas
two separate disorders occurring in a patient at one time...follow the path and you will find all the problems
what is a mixed disturbance and how do you approach them
utilize "independent variables" such as PCO2, strong ion differences (SID), and total concentration of weak acids (Atot) to give you a deeper understanding
how might you describe the Stewart's approach to dx. acid-base

anesthetic drugs, equipment, and dead space
what is a primary cause of acidosis (increased CO2) related to this class
acidosis--increase CO2
these would all be related to respiratory causes of _____

alkalosis--decreased CO2
these would all be related to respiratory causes of _____

acidosis--decreased HCO3
these would all be related to metabolic causes of _____

alkalosis---increase HCO3
these would all be related to metabolic causes of _____

1. enzymes do not function as well
2. cardiac system impaired, decreased CO, and decreased renal/hepatic flow
3. ventricular arrhythmias or fibrillation
4. O2 binding curve shifted to the right--->decreased hemoglobin-O2 binding
5. depression/coma/death
what are some consequences of acidosis
1. agitation and other CNS signs
2. seizures due to hypocalcemia
3. hypokalemia
4. O2-hemoglobin curve shifts left inhibited O2 release
what are some consequences of alkalosis
quickly obtained results to monitor respiratory and acid-base status, electrolytes and lactate, and oxygen in both awake and anesthetized patients
why is it important to know about blood gas analysis
respiratory gas exchange like O2 vs CO2
arterial blood gas will provide what information
metabolic status and primarily hepatic or renal issues like acid-base balance
venous blood gas will provide what information
lower; higher
venous blood gas has a _____ pH and ____ pCO2 than arterial blood gas due to local tissue metabolism
1. PaO2 ~80-110mmHg
2. bright red color in a pulsatile flow
how do we know if we got an arterial sample for blood gas
1. PvO2 ~35-45mmHg
2. dark red color in a non-pulsatile flow
how do we know if we got an venous sample for blood gas
1. clip and clean
2. heparin syringe filling with ~1mL of blood with air bubbles taken out quickly to protect from air exposure
when it comes to actually collecting a blood gas sample...
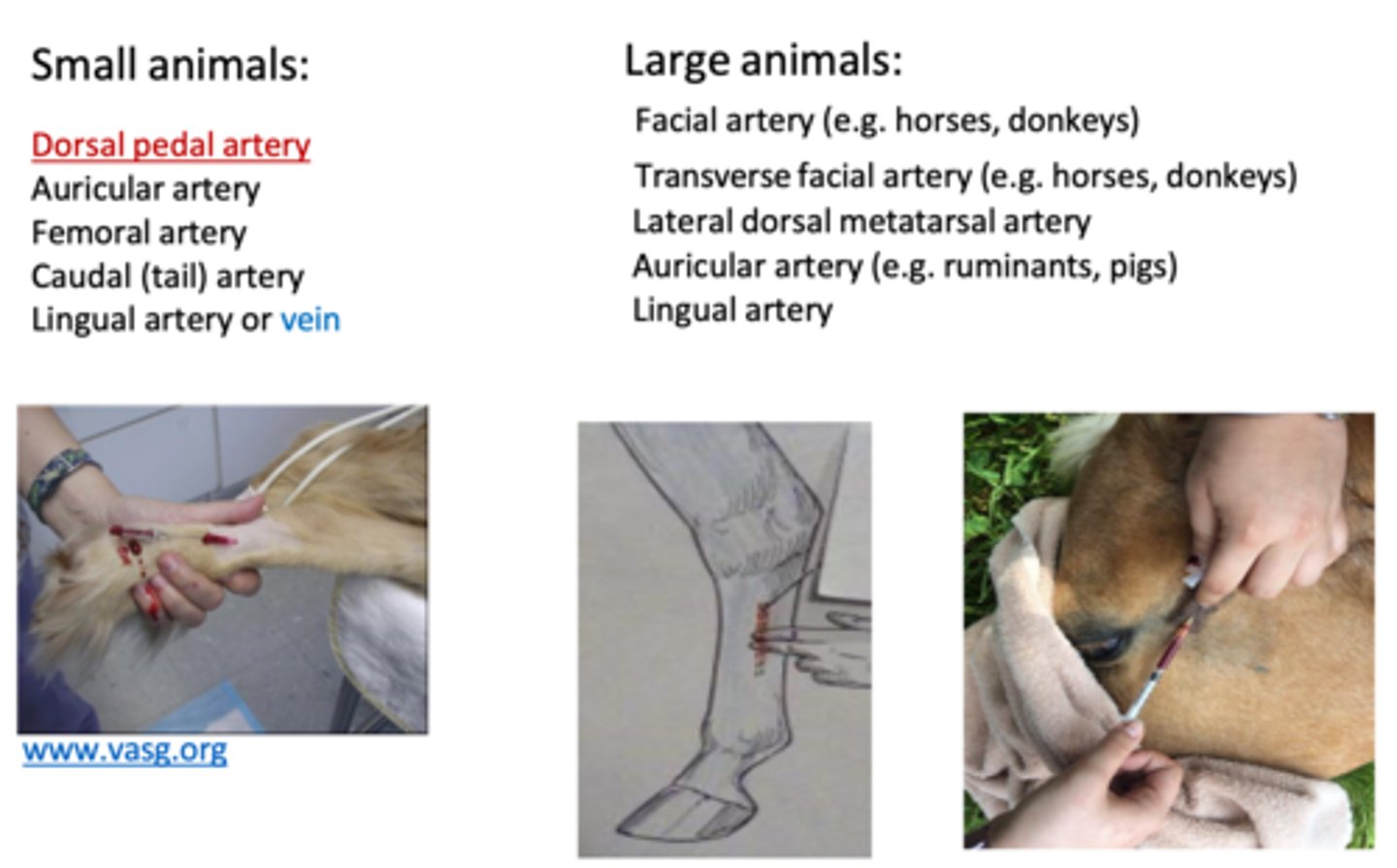
dorsal pedal aa
primary vessel to collect blood-gas sample from small animal
reviewed
review collection sites
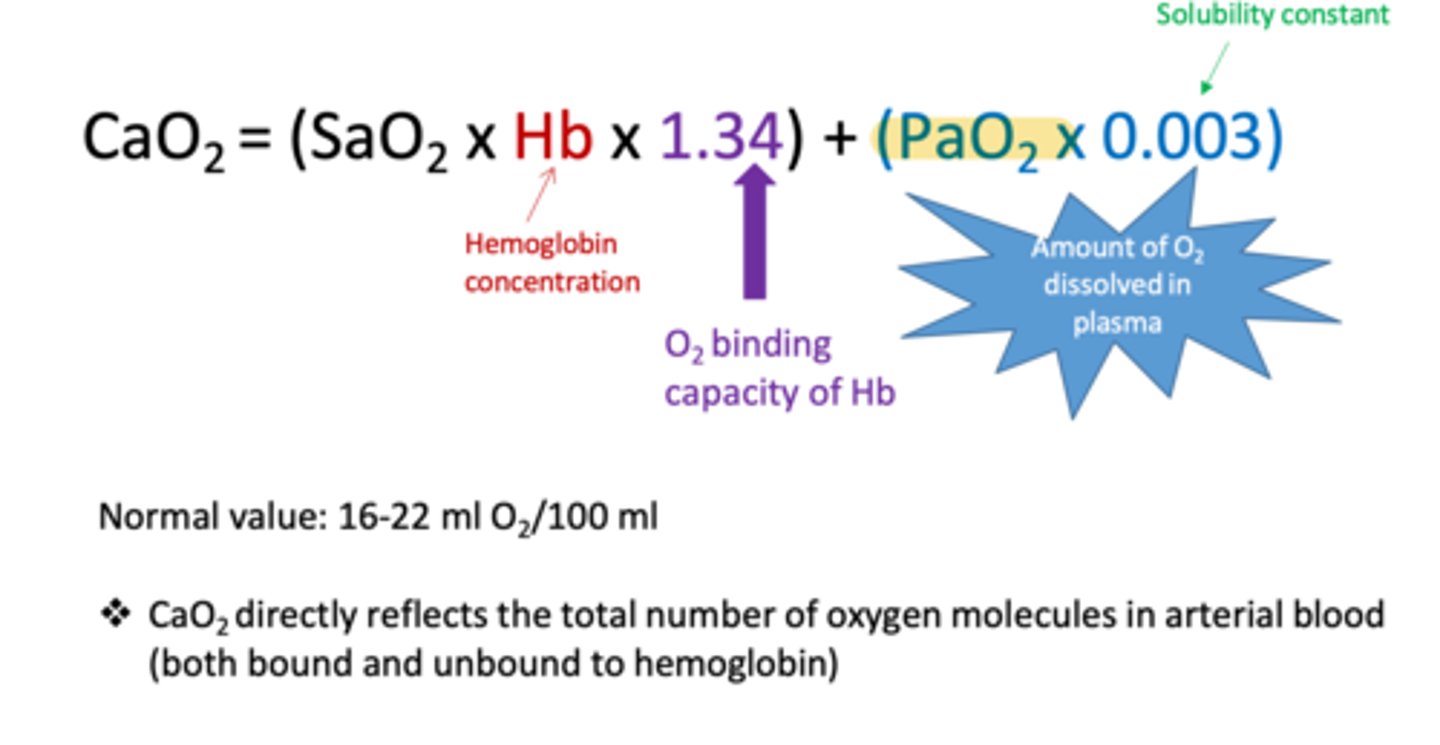
1. pH: H+ concentration
2. partial pressures of O2- PO2: O2 dissolved in plasma
3. partial pressures of carbon dioxide- PCO2: respiratory component of acid-base
what do blood gas analyzers measure directly
1. HCO3-
2. BE
3. SaO2
what do blood gas analyzers calculate
1. regulate pH of body fluids as an immediate buffer for fixed acids in the blood
2. facilitate transport of CO2 from the body to the lungs
functions of bicarbonate/HCO3-
metabolic component
bicarbonate value is an assessment of the ______ of acid base
total amount of bicarbonate as it is 85% of TCO2
total carbon dioxide (TCO2) is a measure of...

1. amount of strong acid base needed to titrate 1L of blood to 7.4pH
2. base excess = metabolic alkalosis
3. base deficit = metabolic acidosis
4. used to calculate bicarbonate therapy
what is base excess (BE)

% of available heme-binding sires saturated with O2 from an arterial sample calculated based on position of O2-hemoglobin dissociation curve and PaO2
what is SaO2
is sample arterial or venous
-->SaO2 >88% = arterial
-->SaO2 <88% = mixed or venous
step 1 is to determine...
acid/base status
step 2 is to determine....

assess ventilatory statue (PaCO2)
step 3 is to...

assess how the animal is oxygenating via A-a and PaO2: FiO2
step 4 is to...
alveolar-arterial (A-a) O2 gradient to determine effect of hypoventilation
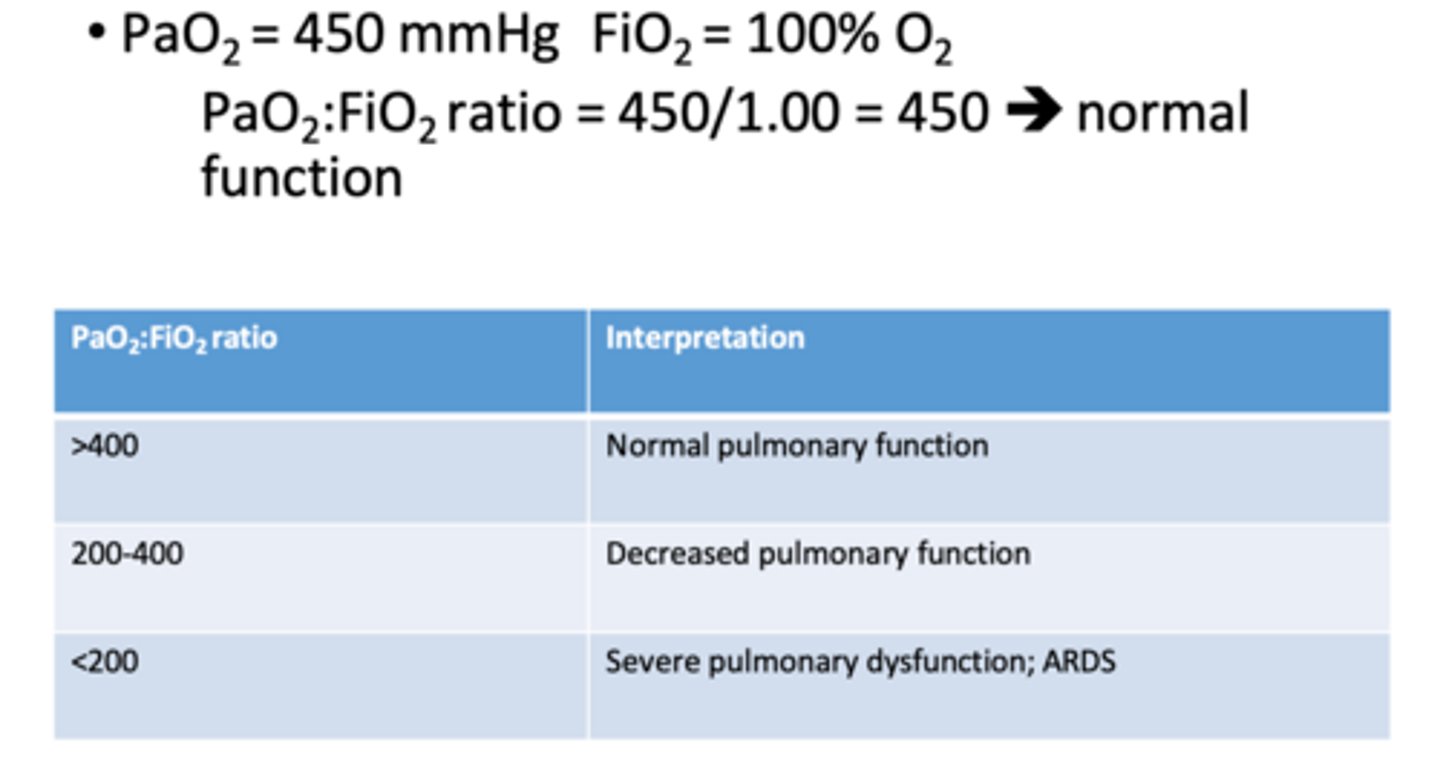
what calculation do we use to assess how an animal is oxygenating

explain the fraction of inspired O2 compared to the PaO2 as a ratio
what is the significance of the FiO2 >0.21 (5x) rule
determine anion gap HE SKIPPED THIS SLIDE
step 5 is to...

1. air bubbles increase PaO2 and decreased PaCO2
2. excess heparin decreases pH
3. delay in analysis decreases PaO2 and pH and increased PaCO2
4. blood clots
5. temperature and barometric pressure
what are some things that can affect accuracy of a blood-gas analysis
decrease PaO2 (<60 mmHg), SaO2 (<90%), or hemoglobin content
hypoxemia is defined as
general term for impaired O2 delivery (DO2)
hypoxia is defined as
measures how much O2 has moved across the lungs and dissolved into the plasma as PaO2
overall significance of blood gas when it comes to O2 measurements