Electrode potentials
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
What are the components of an electrochemical cell:
Two half cells
Connected with a wire => allows electrons to pass through
High resistant voltmeter
salt bridge, consists of filter paper dipped in concentrated KNO3 => allows ions to move through to complete the circuit
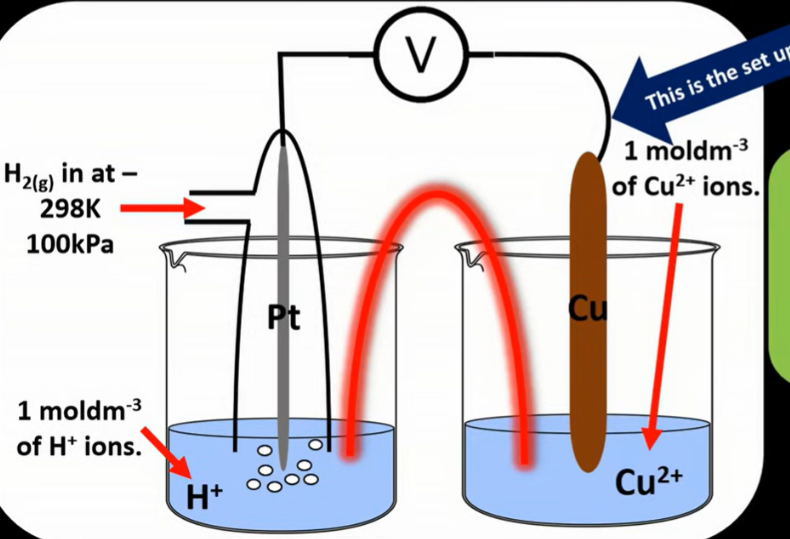
Draw the standard hydrogen electrode, including what side the hydrogen half cell is connected, and standard conditions
Standard conditions:
All solutions 1mol/dm3
298K
100kPa
For hydrogen half cell is connected on the left side, where platinum electrode is used, as it is inert and a good conductor of electricity. A platinum electrode, allows the transfer of electrons, and provides a reaction surface.
How to calculate the EMF of a cell?
When EMF is positive, the reaction is feasible
When the EMF is negative, the reaction is not feasible
Right - left
NO PRoblem (the acronymn)
Sometimes in a multiple choice question, you have to look at the diagram and then it is the right electrode - the left electrode

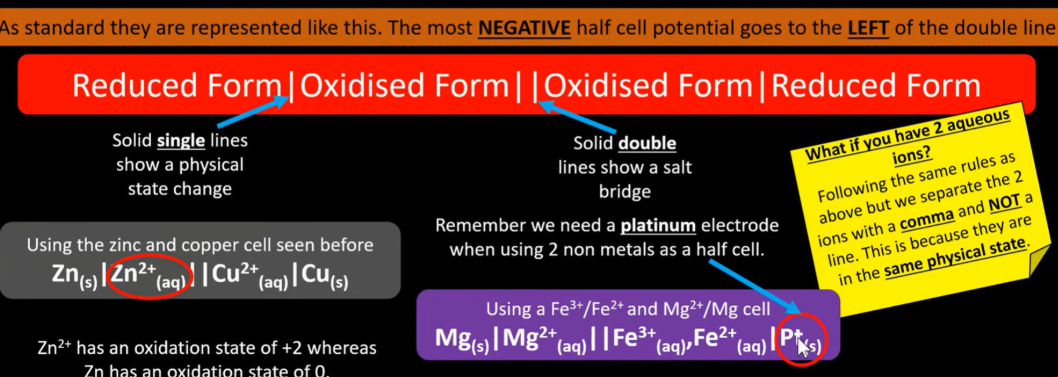
How to write cell notation rules
The most negative half cell is on the left of double line
The oxidised form is closest to the salt bridge
The double verticle lines represent the salt bridge
The single line shows a state change
A comma is used to separate same physical state
What is the electrochemical series? (+) and (-) represent what?
Electrochemical series is the list of EMF values given for a metal in standard electrodes.
The more - EMF on this series shows a stronger reducing agent
The more + EMF on the series shows a stronger oxidising agent.
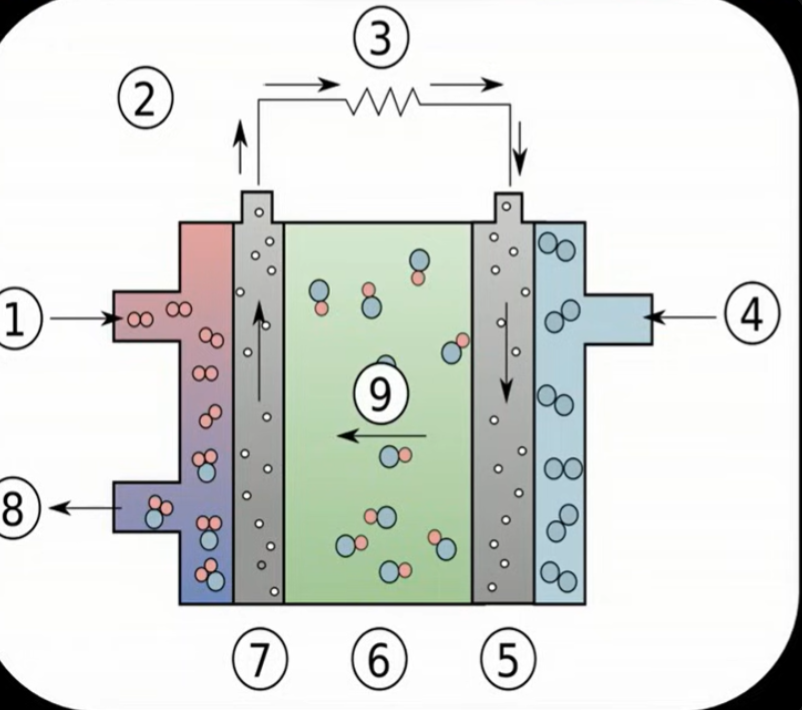
Describe the electrode reactions in a hydrogen-oxygen fuel cell:
Hydrogen is fed here, and reacts with the OH- ions. The reaction is 2H2(g)+ 4OH-(aq)→ 4H2O(l) +4e-
Flow of electrons through the platinum electrode, platinium is a good conductor of electricity but is inert
Flow of electrons is used to power something
Oxygen is fet here, where it reacts with water and 4 electrons from step one to make OH- ions. The reaction is O2(g)+2H2O(l)+4e- → 4OH-
Electrons flow to the negative electrode made from platinium
The electrolyte is made from KOH solution, carries the OH ions from cathode to anode
electrons flow from the positive electrode which is made from platinum
Water is emitted the product of step 1
OH ions from reaction 4 are carried towards the anode via the electrolyte
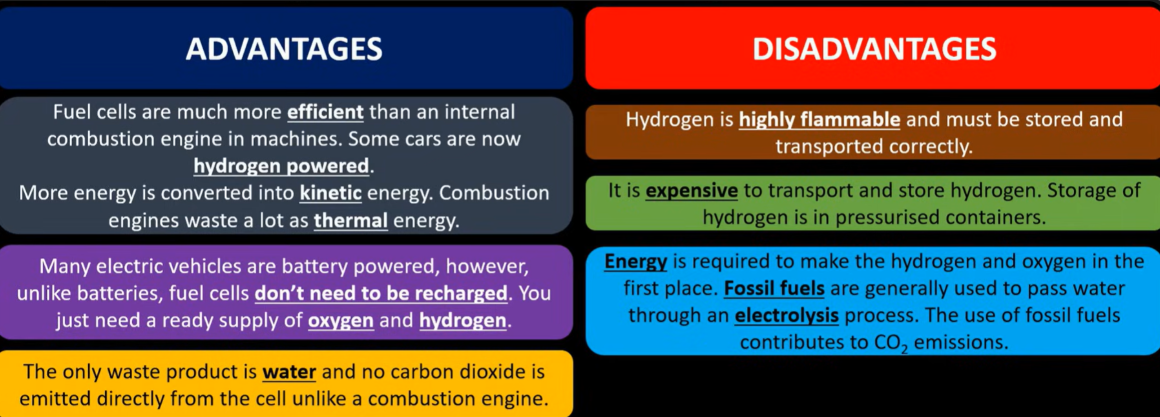
The advantages and disadvantages of hydrogen fuel cell:
Summary of reachargeable and non-rechargeable cells:
Non-rechargable cells: When the cell reactions cannot be reversed
(+) Cheaper to buy
(-) Single use … finite … landfill … Toxic substances
Rechargeable cells: The electrode reactions can be reversed, by applying a greater potential in the reverse direction (-) It requires electricity … which releases carbon dioxide
(+) Minimise waste and resource use
(-) More expensive
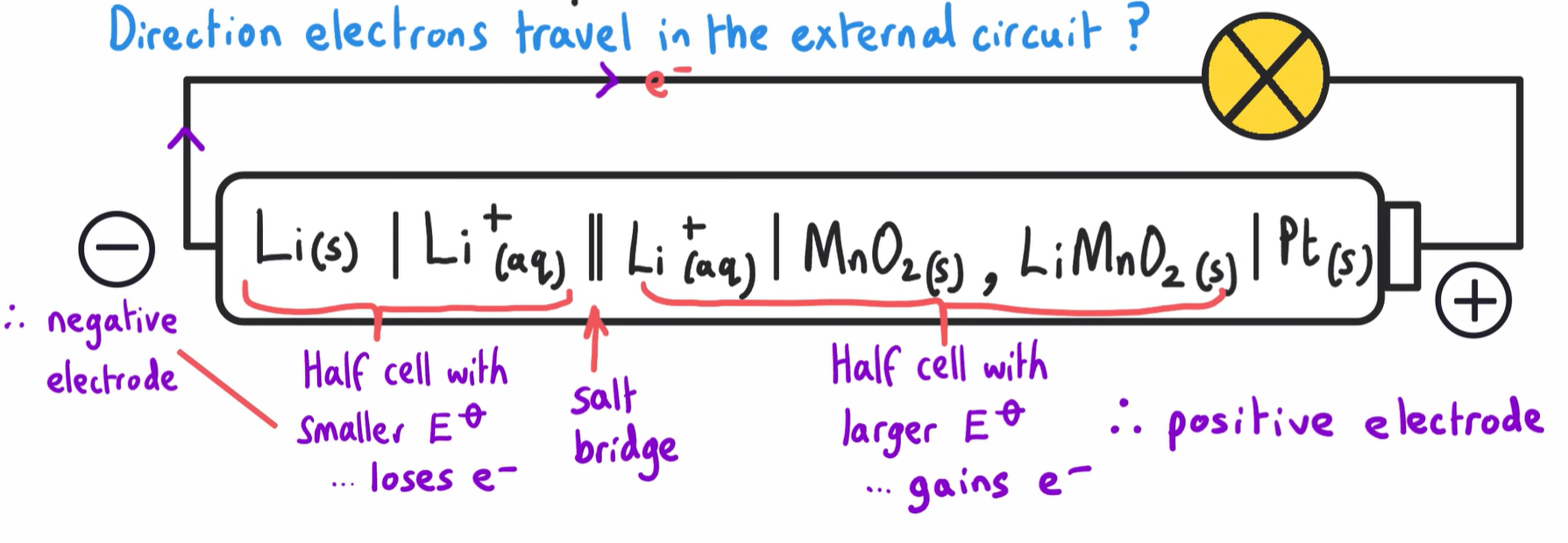
POROUS SEPERATOR ACTS LIKE A SALT BRIDGE, COMPLETES THE CIRCUIT AS IT ALLOWS IONS TO FLOW.
INFORMATION MUST NEED TO KNOW FOR THE LITHIUM ION CELL
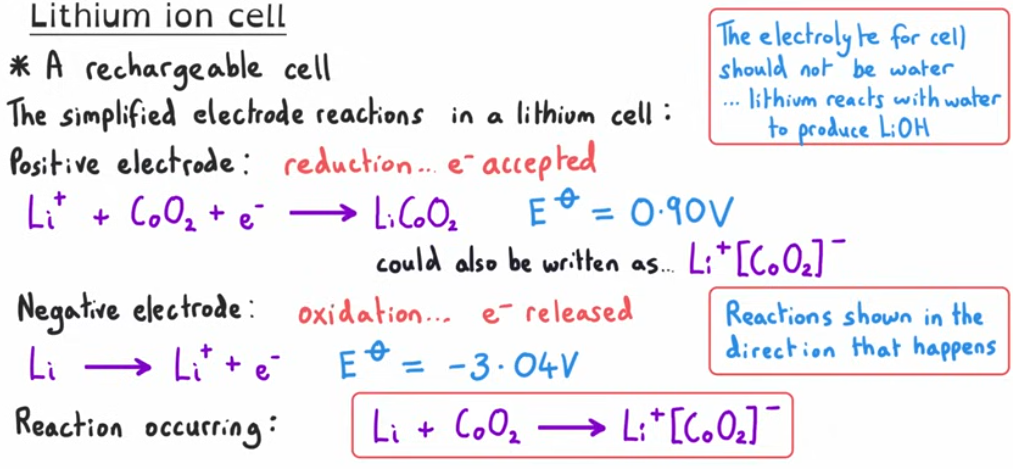
What is the direction of electrons travelling through the circuit?