BIOC 4331 Lecture 4
1/19
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
What does Ka represent?
The acid dissociation constant, an equilibrium constant.

What is the general acid dissociation reaction?

What is the Ka expression for HA?
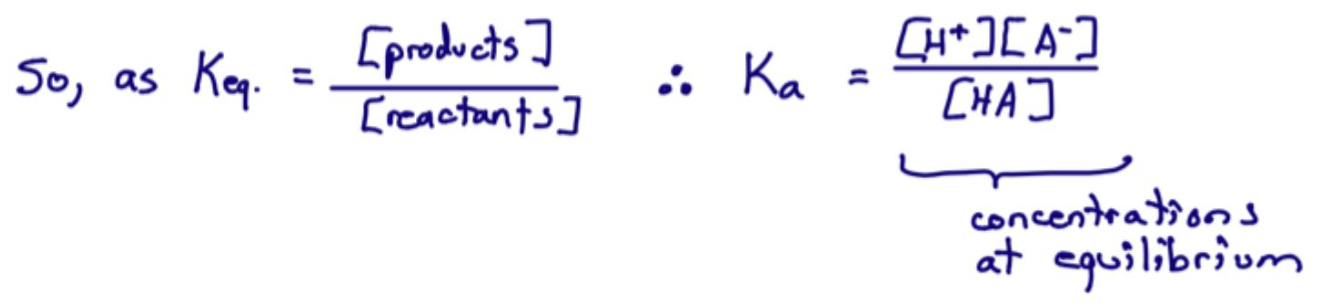
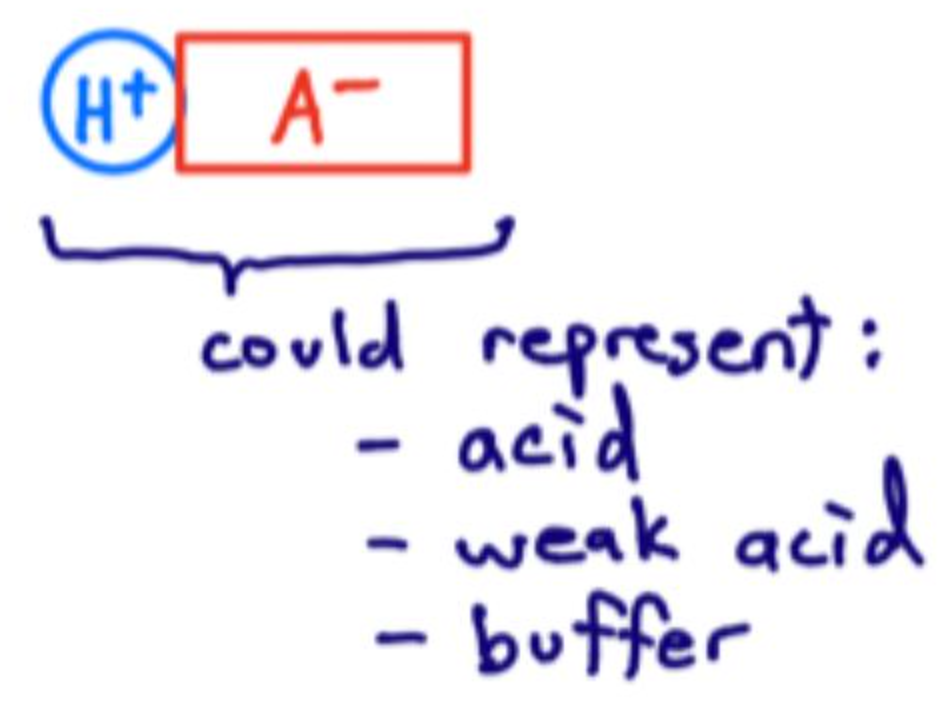
What does HA represent?
An acid, weak acid, or a buffer component.
At equilibrium, what is the relationship between [H+] and [A-]?
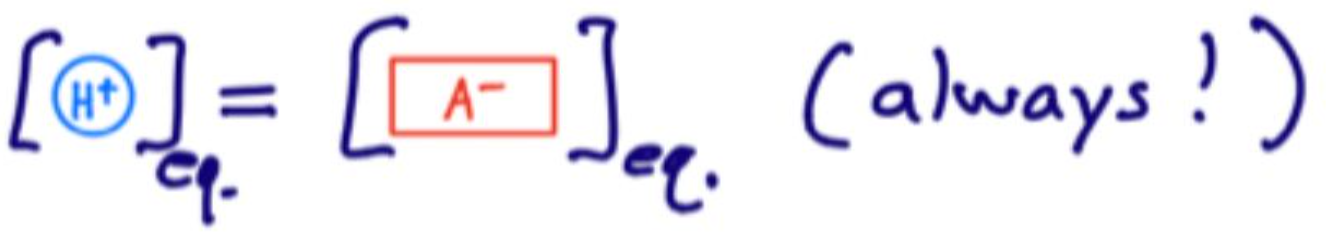
"What does "dissociated" mean in acid-base problems?
The amount of HA that has formed H+ and A-.
What is the relationship between dissociated species at equilibrium?
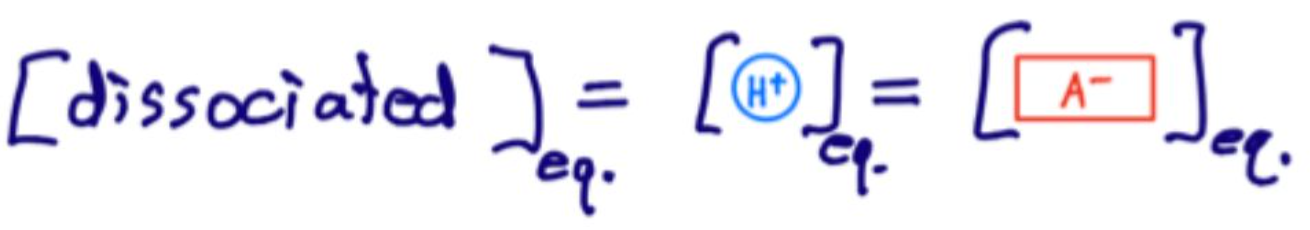
How do you calculate the amount still associated at equilibrium?
[HA]eq = [starting] - [dissociated]
![<p>[HA]<sub>eq</sub> = [starting] - [dissociated]</p>](https://knowt-user-attachments.s3.amazonaws.com/8cada526-0e13-4b97-901a-79c226b678ac.png)
If x dissociates, how do you rewrite Ka?
Ka = x2/([starting] - x)
![<p>Ka = x<sup>2</sup>/([starting] - x)</p>](https://knowt-user-attachments.s3.amazonaws.com/c2ea431e-aba9-4bd6-a9bd-ea2207ad4b87.png)
What is the general quadratic form?
ax2 - bx - c = 0
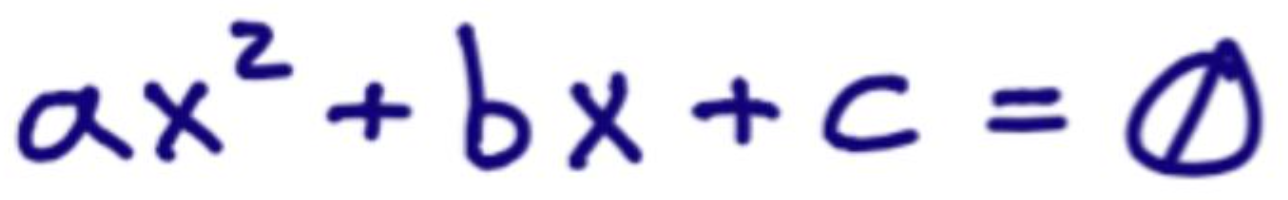
What is the quadratic formula?
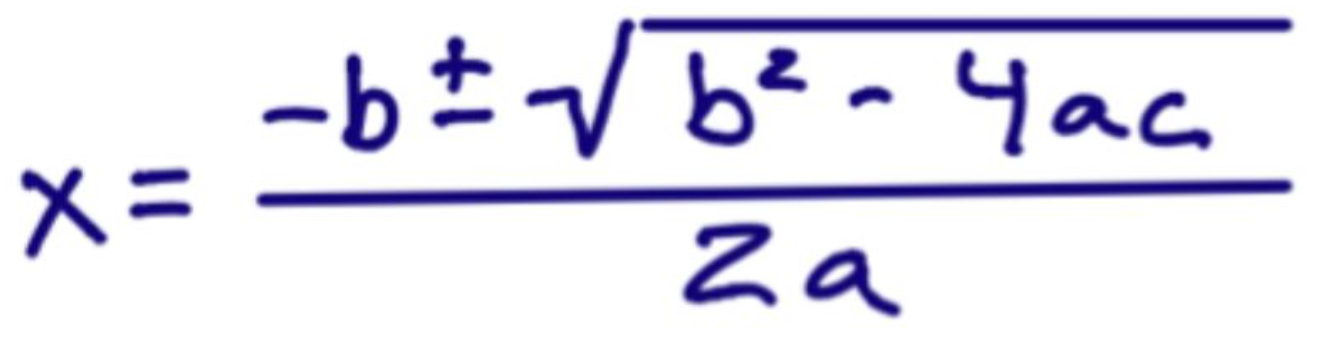
What is the Henderson-Hasselbalch equation?
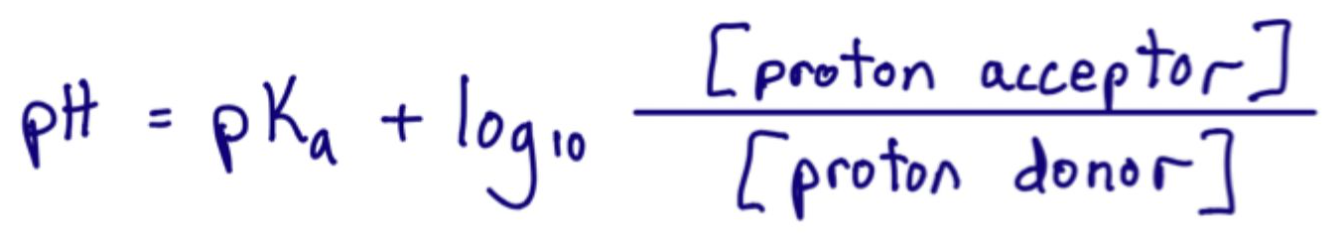
In a buffer, what is the proton donor and what is the proton acceptor?
The proton donor is HA (acid) and the proton acceptor is A- (conjugate base)
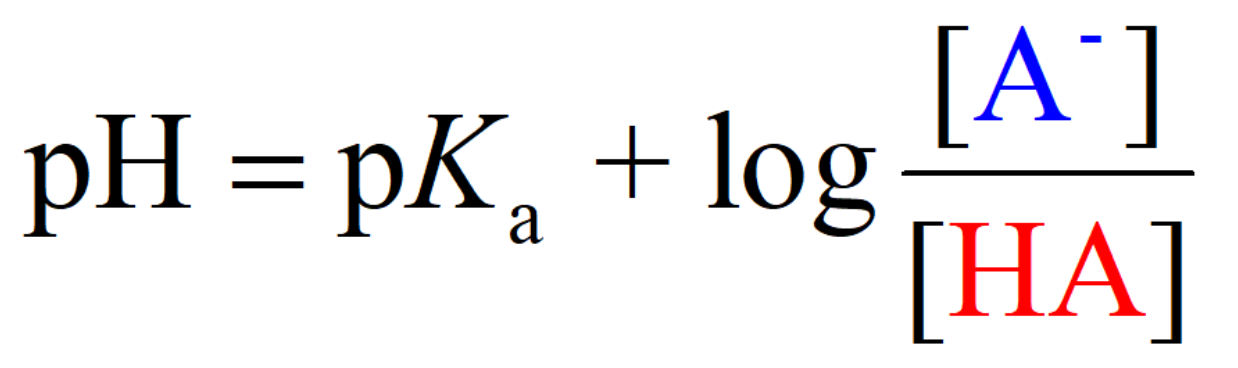
What happens when pH = pKa?
[HA] = [A-]
How do you calculate moles from molarity?
moles = M*V
At the equivalence point of a strong acid–strong base titration, what relationship is true?
moles acid = moles base
For a strong acid, what is the relationship between [H+] and molarity?
[H+] = Macid
How do you calculate pH from hydrogen ion concentration?
pH = −log[H+]
What is the inverse-log form used to find [H+] concentration?
[H+] = 10−pH
What is the definition of pKa?
pKa = −log[Ka]