BC 3- Amino acid metabolism
1/159
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
160 Terms
What is required for amino acid synthesis?
Carbon backbones and reduced nitrogen.
Where do carbon backbones for amino acids come from?
Intermediates of glycolysis, the citric acid cycle, and the pentose phosphate pathway.
What provides reduced nitrogen (NH₄⁺) for amino acid synthesis?
Glutamate and glutamine.
Where does amino acid biosynthesis occur in the cell?
cytosol
What form are biosynthetic amino acids in?
The L-form.
L-amino acid visual
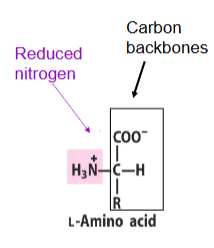
What is transamination?
A reaction that transfers an amino group from an amino acid to a keto acid.
What type of molecules participate in transamination?
An α-amino acid and an α-keto acid.
What enzyme catalyzes transamination reactions?
Transaminases (aminotransferases).
What coenzyme is required for transamination?
Pyridoxal phosphate (PLP).
What vitamin is PLP derived from?
Vitamin B6.
What is glutamate’s main role in amino acid metabolism?
It is a major donor of amino groups for the synthesis of most other amino acids.
What is produced when glutamate donates an amino group in transamination?
α-ketoglutarate.
What is glutamine used for in nitrogen metabolism?
It provides amide nitrogen for biosynthesis of nitrogen-containing compounds.
What biomolecules can receive amide nitrogen from glutamine?
Asparagine, carbamoyl phosphate, and nucleotides.
What are the main routes for incorporating inorganic nitrogen into organic molecules?
Synthesis of glutamate and glutamine.
What roles do glutamate and glutamine play in metabolism?
They serve as major nitrogen donors in many metabolic reactions.
How is glutamate synthesized?
From α-ketoglutarate + NH₄⁺ + NADPH → glutamate + NADP⁺ + H₂O.
What is the key precursor for glutamate synthesis?
α-ketoglutarate.
How is glutamine synthesized?
Glutamate + NH₄⁺ + ATP → glutamine + ADP + Pi + H⁺.
What energy molecule is required to form glutamine?
ATP
What is glutamate dehydrogenase?
An enzyme that catalyzes the reversible conversion between glutamate and α-ketoglutarate.
What reaction does glutamate dehydrogenase catalyze (direction toward glutamate)?
α-ketoglutarate + NH₄⁺ + NAD(P)H → glutamate + NAD(P)⁺ + H₂O.
What reaction does glutamate dehydrogenase catalyze (breakdown direction)?
Glutamate → α-ketoglutarate + NH₄⁺ + NAD(P)H (or NAD(P)⁺ depending on direction).
What is the role of glutamate dehydrogenase in nitrogen metabolism?
It helps incorporate or release inorganic nitrogen (NH₄⁺) in amino acid metabolism.
Where does glutamate dehydrogenase act in metabolism?
It links amino acid metabolism with the citric acid cycle via α-ketoglutarate.
What is glutamine synthetase?
An enzyme that catalyzes the formation of glutamine from glutamate and ammonia.
What reaction does glutamine synthetase catalyze?
Glutamate + NH₄⁺ + ATP → glutamine + ADP + Pi + H⁺.
What is the main function of glutamine synthetase?
To incorporate inorganic nitrogen (NH₄⁺) into organic form (glutamine).
Why is glutamine synthetase important in metabolism?
It helps detoxify ammonia and provides nitrogen for biosynthesis.
What is required for glutamine synthetase activity?
ATP.
How is alanine synthesized?
By transamination of pyruvate.
What is the amino group donor in alanine synthesis?
Glutamate.
What is produced when glutamate donates an amino group in alanine synthesis?
α-ketoglutarate.
What is the key reaction in alanine synthesis?
Pyruvate + glutamate ⇌ alanine + α-ketoglutarate.
What is glutamate aminotransferase?
An enzyme that catalyzes transamination reactions involving glutamate, transferring its amino group to an α-keto acid to form a new amino acid and α-ketoglutarate.
What is the main reaction for alanine synthesis?
Alanine is formed by transamination of pyruvate using glutamate as the amino donor.
What enzyme catalyzes alanine synthesis?
Alanine aminotransferase (ALT)
What are the reactants in alanine synthesis?
Pyruvate + Glutamate
What are the products of alanine synthesis?
Alanine + α-ketoglutarate
Why is alanine synthesis important in metabolism?
It links amino acid metabolism with glycolysis (via pyruvate)
What is glutamate aminotransferase?
An enzyme that transfers amino groups between amino acids and α-keto acids (transamination).
How is aspartate synthesized?
By transamination of oxaloacetate using glutamate as the amino donor
What enzyme synthesizes aspartate?
Aspartate aminotransferase (AST)
What are the reactants for aspartate synthesis?
Oxaloacetate + Glutamate
What are the products of aspartate synthesis?
Aspartate + α-ketoglutarate
What cofactor is required for aspartate synthesis?
Pyridoxal phosphate (PLP, vitamin B6)
How is asparagine synthesized?
By amidation of aspartate
What enzyme synthesizes asparagine?
Asparagine synthetase
What are the reactants for asparagine synthesis?
Aspartate + Glutamine + ATP
What are the products of asparagine synthesis?
Asparagine + Glutamate + AMP + PPi
Key difference between aspartate and asparagine synthesis?
Aspartate: formed by transamination
Asparagine: formed by amidation using glutamine
What is the difference between essential and nonessential amino acids?
Essential: cannot be synthesized → must come from diet
Nonessential: can be synthesized by the body
How many amino acids can humans synthesize?
About half (nonessential amino acids)
Which organisms can synthesize all 20 amino acids?
plants and microbes
List the essential amino acids in humans
Histidine, Isoleucine, Leucine, Lysine, Methionine, Phenylalanine, Threonine, Tryptophan, Valine
List the nonessential amino acids
Alanine, Arginine, Asparagine, Aspartate, Cysteine, Glutamate, Glutamine, Glycine, Proline, Serine, Tyrosine
How is alanine synthesized?
Transamination of pyruvate
How is aspartate synthesized?
Transamination of oxaloacetate
How is asparagine synthesized?
From aspartate + glutamine (NH₃ donor)
How is glutamate synthesized?
Transamination of α-ketoglutarate
How is glutamine synthesized?
From glutamate + NH₃ via glutamine synthetase
How is cysteine synthesized?
From serine + methionine (provides sulfur)
How is tyrosine synthesized?
From phenylalanine
What are the main metabolic “starting points” for amino acid synthesis?
Pyruvate
Oxaloacetate
α-ketoglutarate
3-phosphoglycerate
What is the common reaction type for many amino acids?
Transamination (using glutamate)
What is the main nitrogen donor in amino acid synthesis?
Glutamate (directly) or glutamine (for NH₃ transfer)
What two key components are required for amino acid synthesis?
Carbon backbone + nitrogen source
Where do carbon backbones for amino acids come from?
Glycolysis
Citric acid cycle
Pentose phosphate pathway
Which glycolysis intermediates serve as carbon skeletons?
3-phosphoglycerate
Phosphoenolpyruvate (PEP)
Pyruvate
Which citric acid cycle intermediates are used for amino acid synthesis?
Oxaloacetate
α-ketoglutarate
Which pentose phosphate pathway intermediates are used?
Ribose-5-phosphate
Erythrose-4-phosphate
What provides the nitrogen for amino acid synthesis?
NH₄⁺ (ammonia)
Which molecules deliver nitrogen (NH₄⁺) in amino acid synthesis?
Glutamate and glutamine
Which molecule is the main amino group donor in transamination?
Glutamate
Which molecule donates free ammonia (NH₃) in biosynthesis reactions?
Glutamine
What happens to amino acids when they are degraded?
Nitrogen → NH₄⁺ → urea
Carbon skeleton → α-keto acids
What happens to the carbon skeleton (α-keto acids) after amino acid degradation?
Enter the citric acid cycle
Used for energy or glucose production
What happens to nitrogen removed from amino acids?
Converted to urea for excretion
What cycle is responsible for nitrogen excretion?
Urea cycle
How are amino acid metabolism and the citric acid cycle connected?
Through α-keto acid intermediates
Why is ammonia (NH₄⁺) toxic?
It disrupts cellular metabolism and must be converted to urea
What are the two major sources of amino acids for catabolism?
Dietary proteins and intracellular (body) proteins
What is the first step in amino acid catabolism?
Removal of nitrogen (deamination/transamination)
What happens to amino acid carbon skeletons after nitrogen removal?
They become α-keto acids
Where do α-keto acids go in metabolism?
Citric acid cycle (energy production)
Gluconeogenesis (glucose formation)
Ketogenesis (ketone bodies)
Lipid synthesis (fatty acids)
How are amino acids linked to energy production?
Their carbon skeletons enter the citric acid cycle for ATP production
How are amino acids linked to glucose production?
Carbon skeletons can be converted into glucose via gluconeogenesis
Which amino acids are glucogenic?
Those that form intermediates that enter the citric acid cycle or gluconeogenesis
Which amino acids are ketogenic?
Those that form acetyl-CoA or acetoacetyl-CoA
What do acetyl-CoA and acetoacetyl-CoA form?
Ketone bodies
When are ketone bodies produced from amino acid breakdown?
During low glucose availability (fasting/starvation)
What other major biomolecules can amino acid carbon skeletons become?
Fatty acids (via acetyl-CoA)
Where does the urea cycle occur?
liver
What is the central metabolic hub of amino acid catabolism?
Citric acid cycle
What are the three major metabolic fates of amino acid carbon skeletons?
Glucose (gluconeogenesis)
Fatty acids (lipogenesis)
Ketone bodies (ketogenesis)
What connects amino acid metabolism to carbohydrate and fat metabolism?
Acetyl-CoA and citric acid cycle intermediates
where are the amino acids that are catabolized coming from?
released from degradation of cellular proteins or ingested from dietary proteins and degraded down in the stomach or small intestine
absorbed by the intestinal cells and transported in the blood to the tissues
How are dietary proteins digested?
By proteases in the GI tract
Which enzyme digests proteins in the stomach?
Pepsin