National 5 Physics - Energy
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
What is the specific latent heat of a substance?
The amount of heat energy required to change the state of 1kg of the substance
What is the specific heat capacity of a substance?
The amount of heat energy required to raise the temperature of 1kg of a substance by 1°C
True or false: different materials can store different amounts of heat energy and still be at the same temperature.
True, because they could have different specific heat capacities
What is temperature a measurement of?
The average kinetic energy of particles within a substance
Which formula shows the relationship between heat energy, specific heat capacity, mass, and change in temperature?
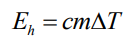
What is specific heat capacity measured in?
J kg-1 °C-1
What are the two types of specific latent heat?

What is the specific latent heat of fusion?
The energy required to change 1kg of a substance from a solid into a liquid at its melting point
What is the specific latent heat of vaporisation?
The energy required to change 1kg of a substance from a liquid into a gas at its boiling point
Can one material have two different values for its latent heat of fusion and its latent heat of vaporisation?
Yes, they always do
Which formula shows the relationship between heat energy, mass, and the specific latent heat?
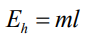
What is specific latent heat measured in?
J kg-1
If you want to calculate the heat energy used to change water into steam, you’ll need to use the latent heat of…
Vaporisation
What happens to the temperature of a substance whilst it changes state?
The temperature remains constant
What is boiling?
When a substance changes state from a liquid to a gas
What is melting?
When a substance changes state from a solid to a liquid
What happens to particles during boiling?
Heat energy is used to break the intermolecular forces
Why do substances not change temperature whilst changing state?
Because the energy added or removed is used to break or form intermolecular bonds rather than changing the kinetic energy of the particles. Temperature is a measure of average kinetic energy, so it remains constant until the structural change is complete
What is the definition of pressure?
The force exerted per unit area
If you increase the force exerted on a solid surface, what happens to the pressure?
It increases because the area remains constant but the force is increasing
How does the kinetic model of gases represent them?
It represents gas particles as numerous small spheres that are far apart and moving constantly in all directions, colliding with one another and the walls of the container
What is the relationship between the kinetic model of gases and pressure?
When the gas particles collide with the walls of the container, they exert forces on it, and the total force (which depends on the number of collisions and the energy they each have) divided by the total area will give us the pressure of the gas within the container
What is absolute zero?
The temperature at which atoms stop moving completely, which is 0K or -273oC
How do you convert from degrees Celsius to Kelvin?
You add 273
How do you convert from Kelvin to degrees Celsius?
You subtract 273
What two factors affect the latent heat of a substance?
The type of substance and the mass of substance
What is the law of the conservation of energy?
Energy cannot be created or destroyed, only transferred from one form to another