OChem Test 2 Reagents
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms

hydrate formation
H3O+ or H2O & OH

hemiacetal formation
NaOEt & EtOH

acetal formation
EtOH, H+

immine formation
NH3 (optional: H+, Na2SO4, MgSO4)
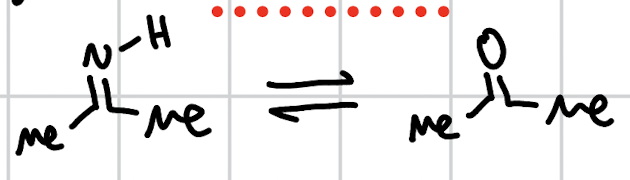
hydrolysis of immines
H2O, H+
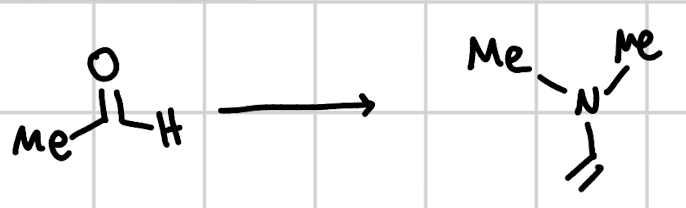
enamine formation
NH3
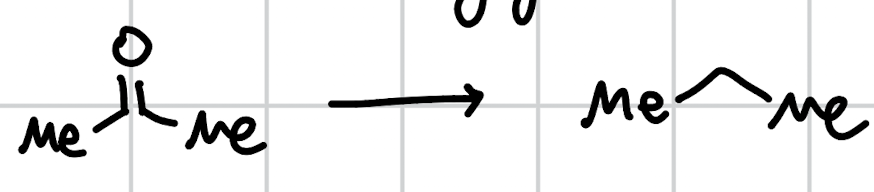
Wolff Kischner Deoxygenation
H2NNH2, -OH, heat x2
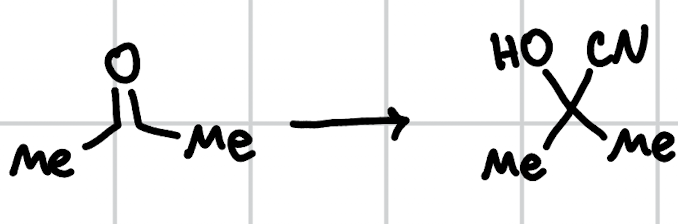
cyanohydrin formation
KCN, HCl
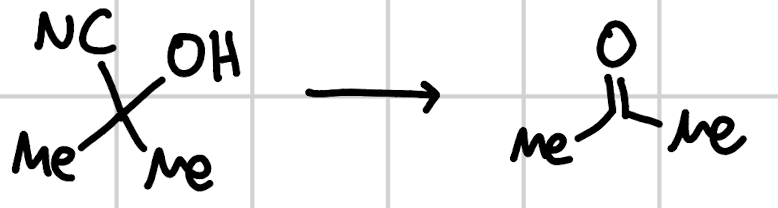
cyanohydrin destruction (idk lol)
base (-OH, pyridine, etc)
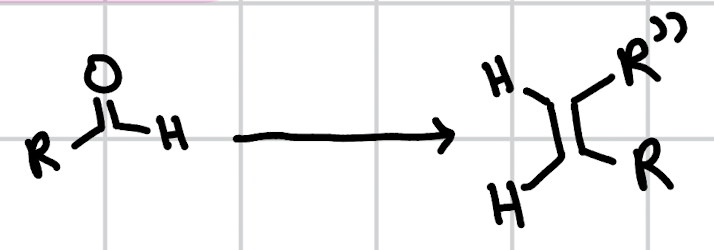
wittig reaction
Ph3P=–R
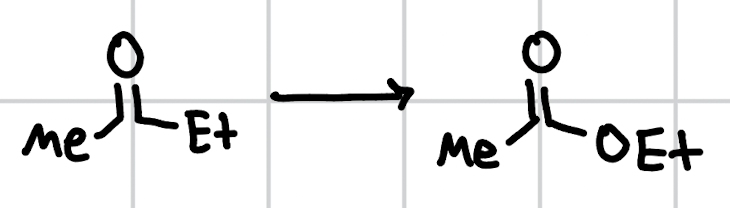
Baeyer-Villiger oxidation
mCPBA
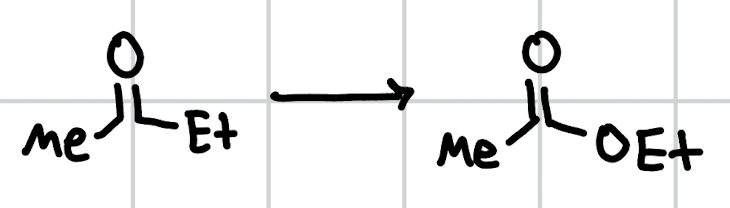
carbonation of organometallic reagents (bunny rabbit rxn)
1. CO2 2. H3O+

conversion of carboxylic acid to acid chloride
SOCl2 or PCl3 or PBr3
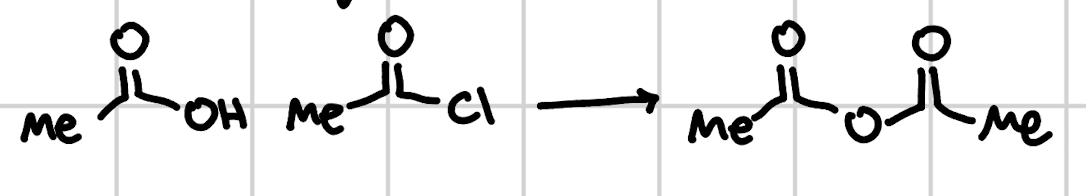
carboxylic acid to anhydride
NEt3 or heat
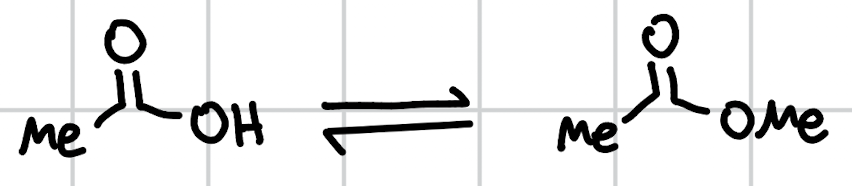
fischer esterification
MeOH, H+
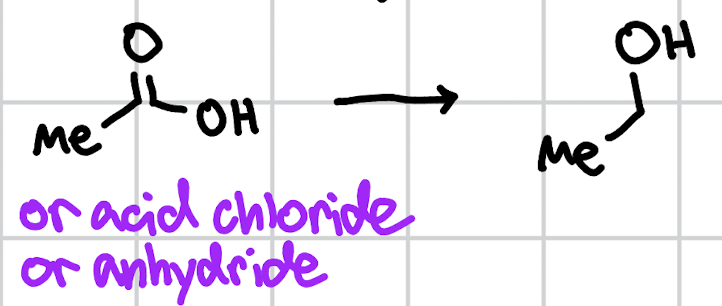
reduction of carboxylic acids
1. LAH 2. H3O+
hydrolysis of acid chlorides
H2O
hydrolysis of anhydrides
H2O
hydrolysis of amides
conc acid/base or heat
hydrolysis of esters
acid/base
hydrolysis of nitriles
conc acid/base, heat
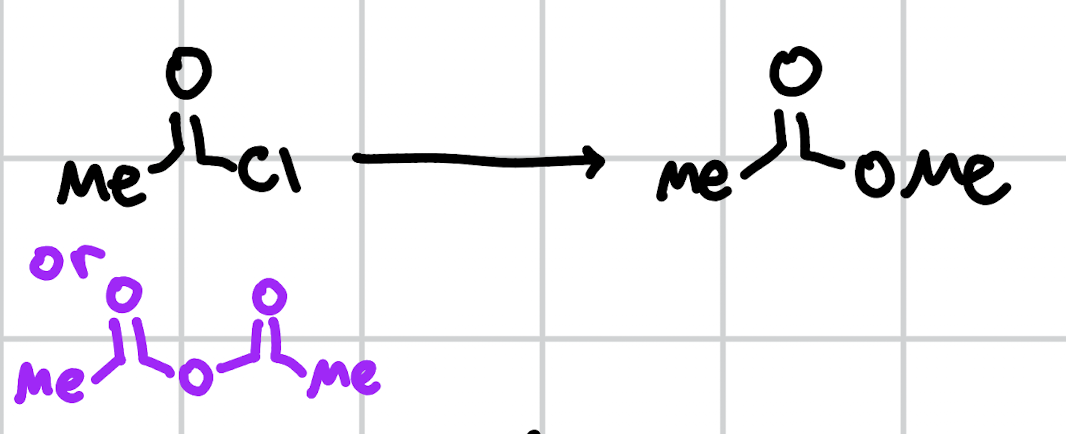
acid chloride/anhydride to ester
NaOMe or MeOH, H+
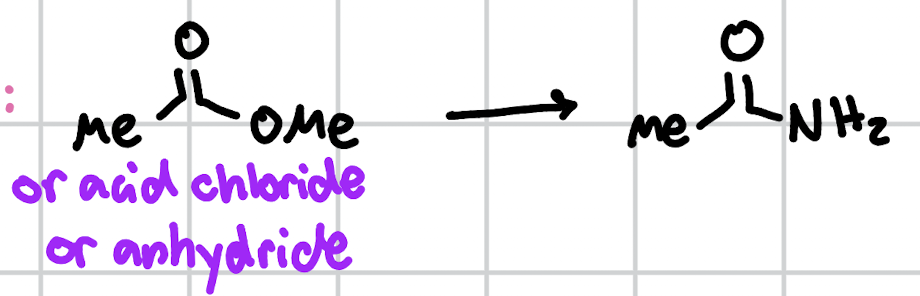
amidation
NH3, heat/DMAP
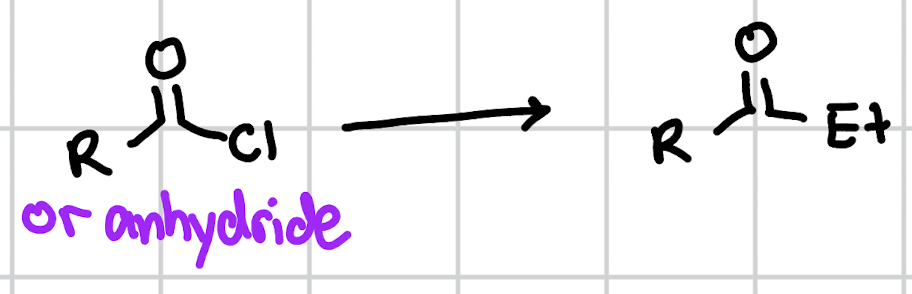
conversion of acid chloride/anhydride to ketone
1. Et2LiCu 2. H3O+

conversion of acid chloride/anhydride to aldehyde
1. LiAl(OtBu)3H 2. H3O+
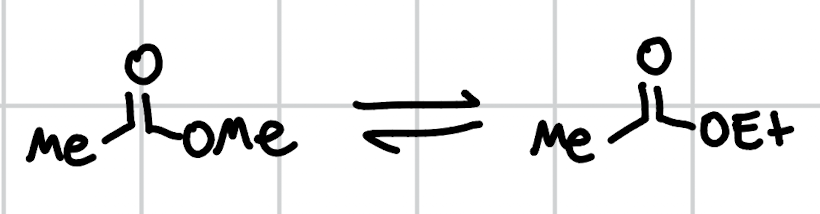
transesterification
EtOH, H+
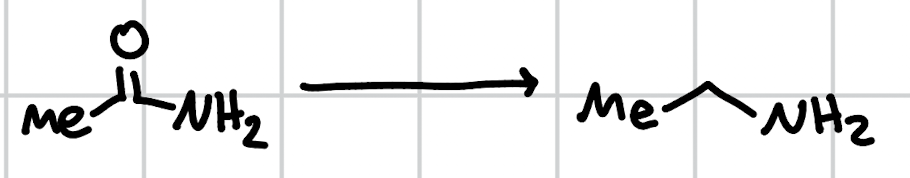
reduction of amides w/ LiAlH4
1. LiAlH4 2. H3O+
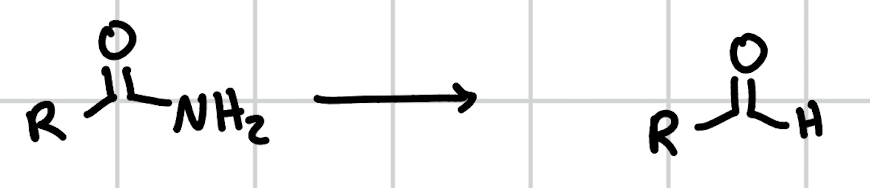
reduction of amides w/ DiBAl
1. DiBAl (1 eq) 2. H3O+
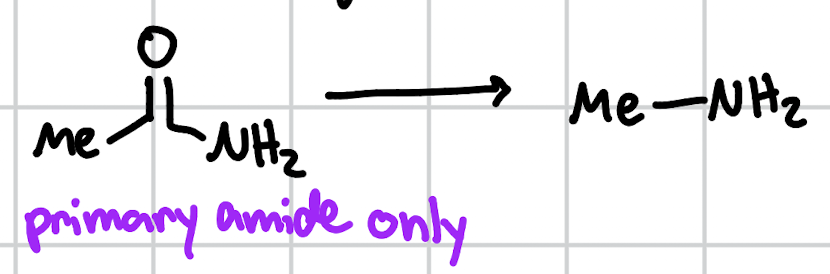
Hoffman rearrangement
KOH, Br2