AP Chemistry- Unit 3
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
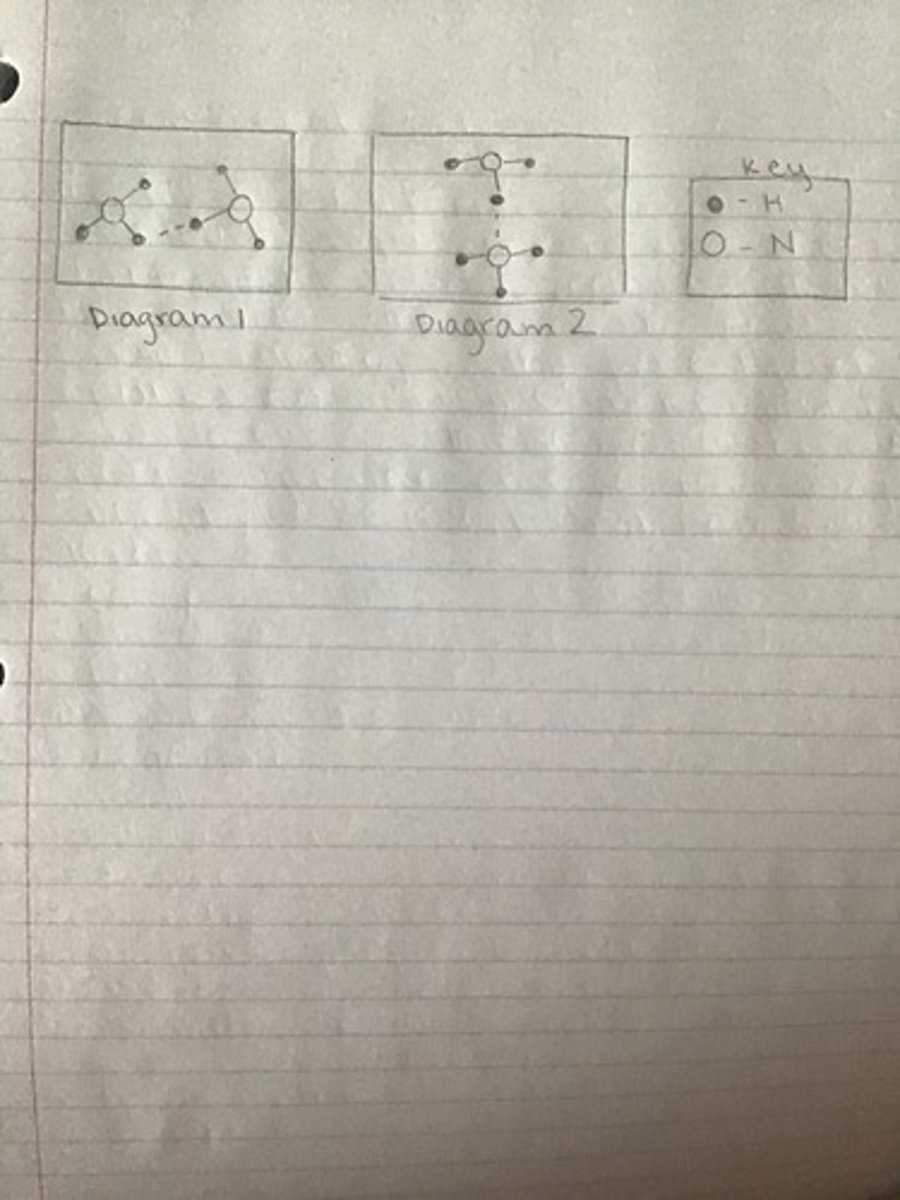
shown above best represents the strongest intermolecular force between two ammonia, NH3, molecules?
Diagram 2- hydrogen bonding between nitrogen and hydrogen atoms in different molecules.
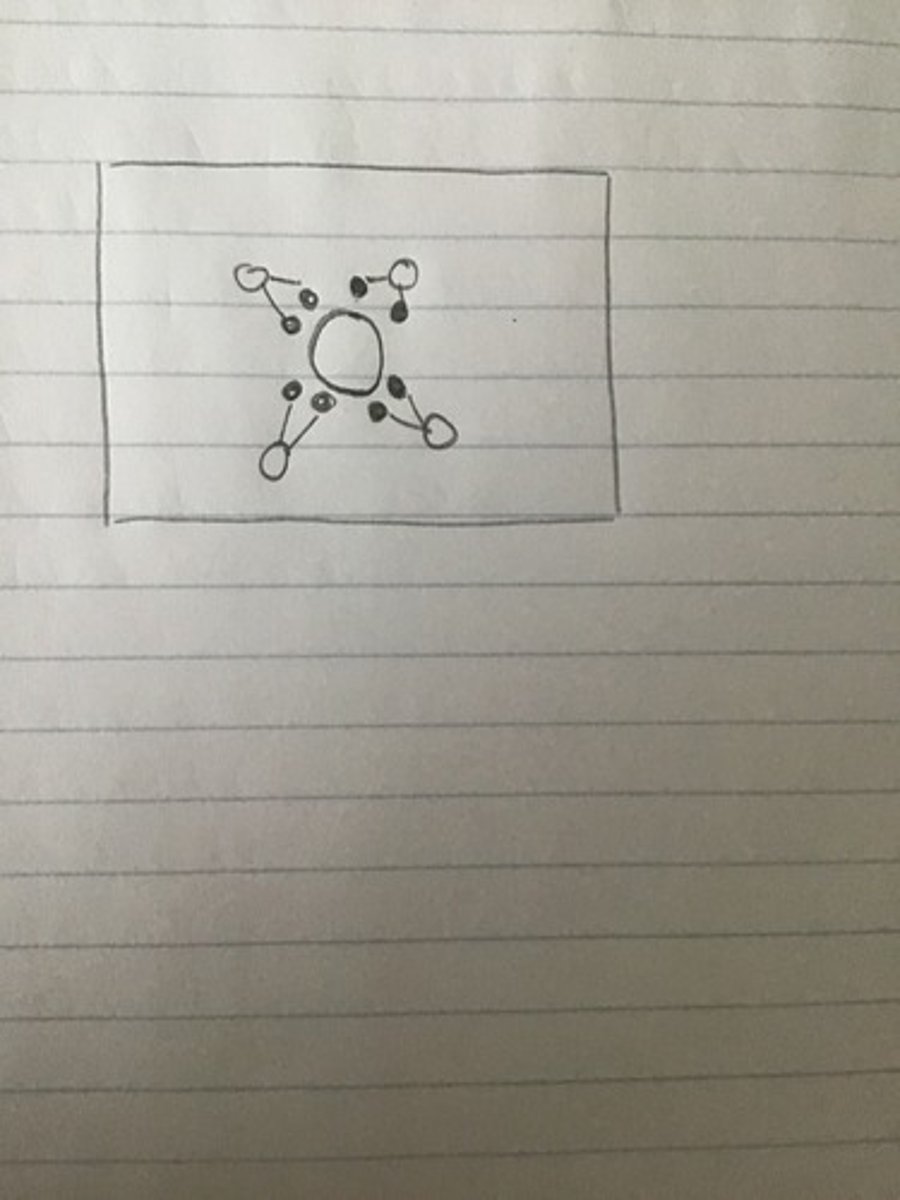
A solid compound of a group 1 (alkali) metal and a group 17 (halogen) element dissolves in water. The diagram above represents one type of solute particle present in the solution. Which of the following identifies the solute particle and best helps explain how the solute particle interacts with water molecules?
The particle is negative and produces ion dipole attractions with the hydrogen atoms in water.
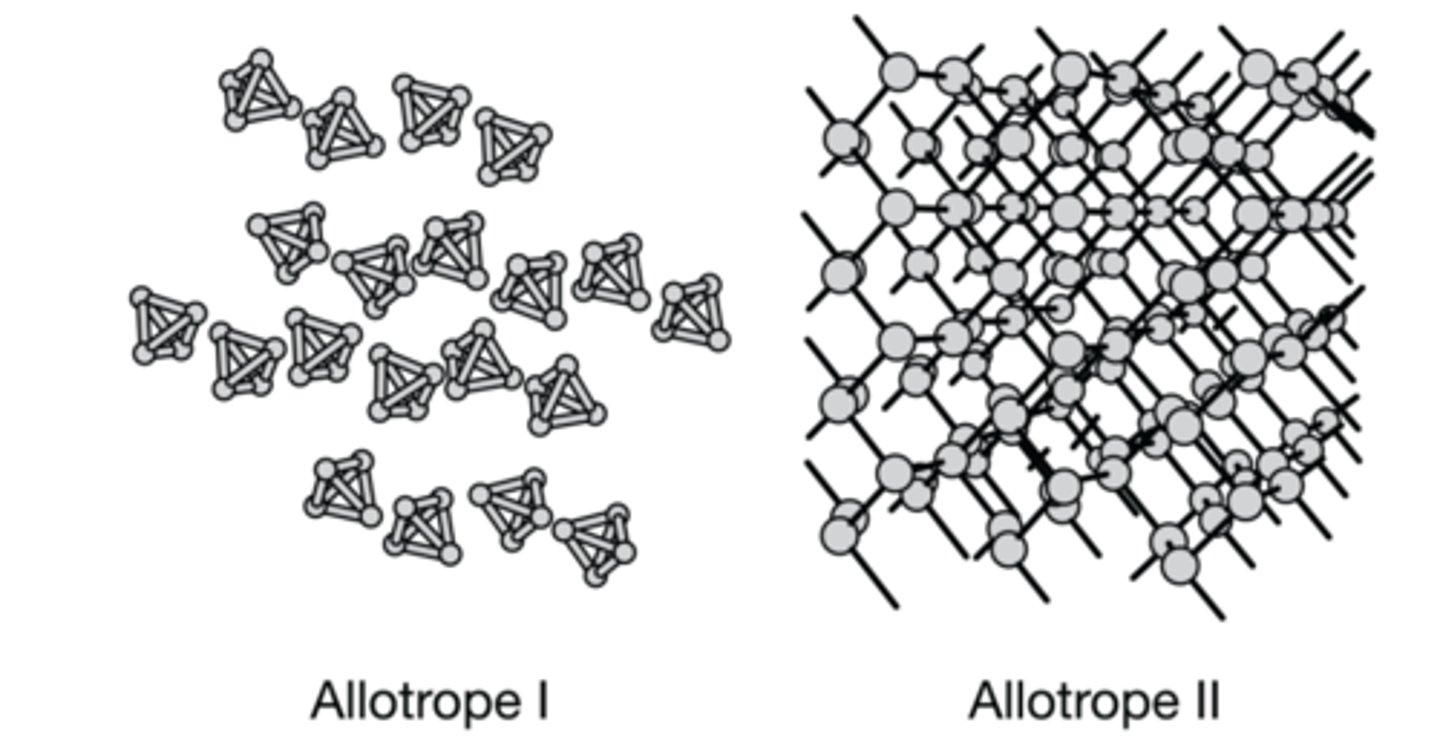
The diagrams above represent two allotropes of solid phosphorus. Which of the following correctly identifies the allotrope with the lower melting point and explains why?
Allotrope 1 because it doesn't contain the covalent bonds between the phosphorus atoms that are found in allotrope 2
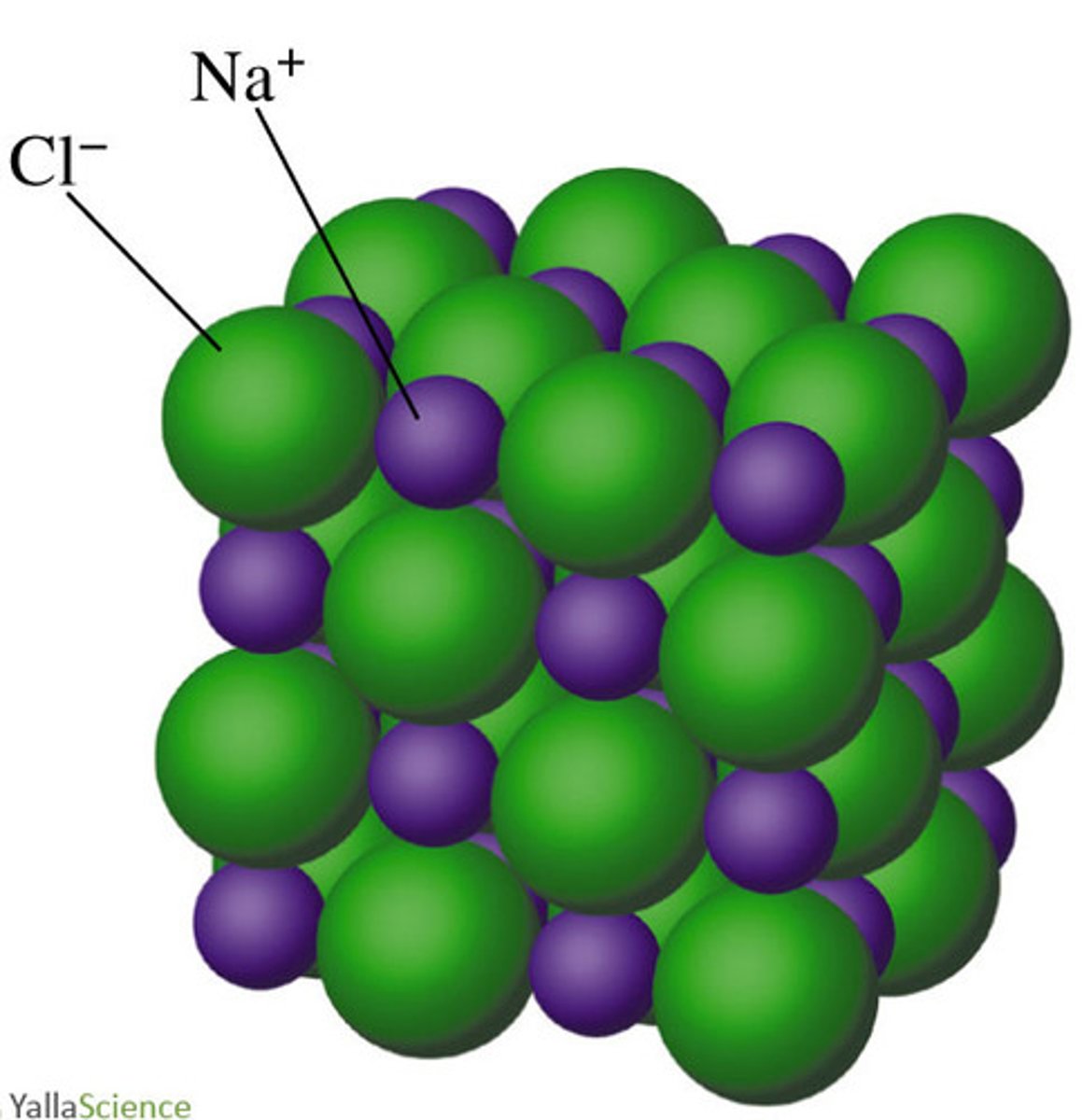
The crystal structure of NaCl is shown above. Which statement correctly compares crystalline NaCl(s)to molten NaCl(l) in terms of electrical conductivity?
Crystalline NaCl contains no freely moving electrons to conduct electricity, but molten NaCl is composed of freely moving Na+ and Cl− ions, which allows it to be a good conductor of electricity.
What type of solid will each of the following substances form:
a) Diamond
b) NaOH
c)SiO2
d) CaCO3
a) Network
b)Ionic
c)Network
d)Ionic
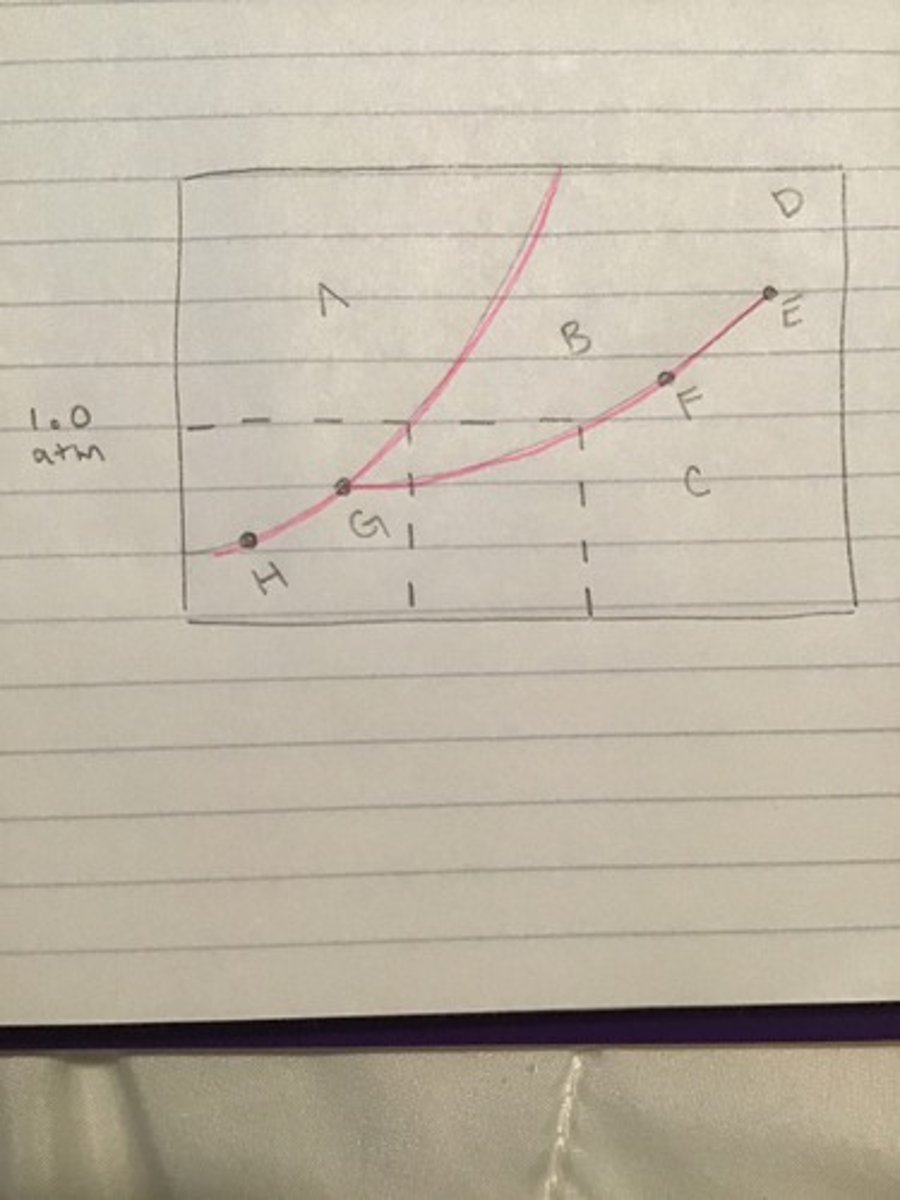
Label points A-H on the phase diagram. Include triple point and critical point.
A) Solid
B)Liquid
C)Gas
D)Vapor
E)Critical Point
F)Liquid+Gas
G)Triple Point
H)Solid+Gas
A gaseous air‑fuel mixture in a sealed car engine cylinder has an initial volume of 360.mL at 1.0 atm. To prepare for ignition of the fuel, a piston moves within the cylinder, reducing the volume of the air‑fuel mixture to 40.mL at constant temperature. Assuming ideal behavior, what is the new pressure of the air‑fuel mixture?
About 9 atm, because the volume of the gas mixture decreased by a factor of 9.
A sample of neon gas occupies 27.2 liters at stp. Determine the mass of the neon sample.
24.49g Neon
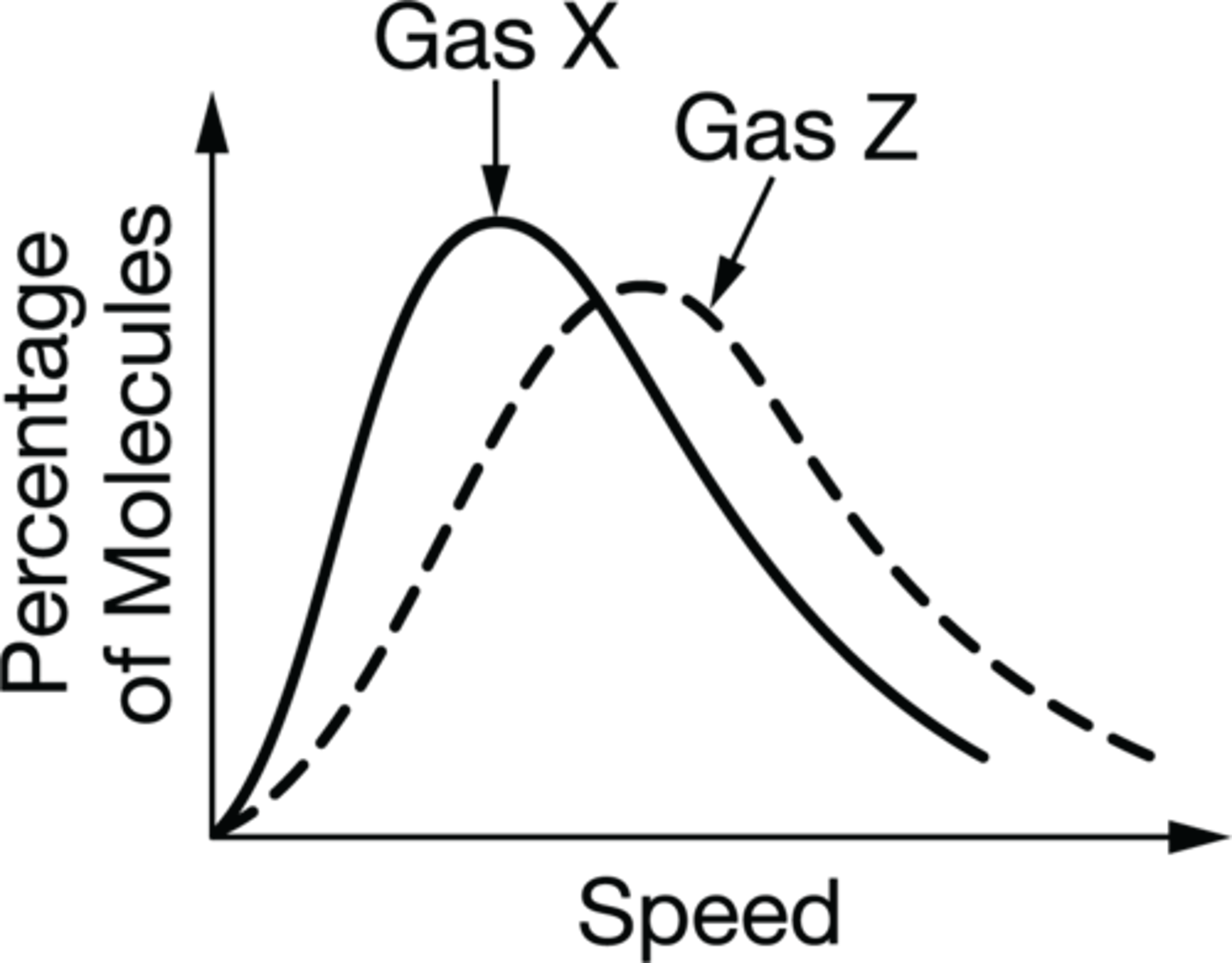
Draw a diagram that best represents the speed of gas particles at a fixed temp. What was it drawn this way?
The gas particles at a fixed temperature will not all have the same speed. They will have variety of speed as described by the Maxwell-Boltzmann distribution.
Which of the following gasses would have a smaller molar mass and why?
Gas Z would have a smaller molar mass. This is because the two gasses are at the same temperature so their kinetic energies are the same. However the curve for gas Z shows that it has a higher average speed than the molecules in gas X. Therefore Gas Z has a smaller molar mass.
Which of the following would deviate the most from an ideal gas at high temperatures and why?
A) H2
B)F2
C)Ne
D)SO2
D) SO2
SO2 would deviate the most from an ideal gas at high temperatures because of its large molecular volume. At higher temperatures, the volume consumed by the gas will be more significant and deviate greatly from an ideal gas with a volume that is negligible.
The gas C2H6 has a larger, more polarizable electron cloud than the gas CH4. How does the deviation of C2H6 from an ideal gas compare to that of CH4, explain?
The deviation from an idea, gas will be greater in C2H6. This is because it has a larger, more polarizable electron cloud that increases and strengthens its intermolecular forces. In an ideal gas the attraction between particles is negligible so C2H6 will have a larger deviation than CH4.
What volume of .2 M HCl stock solution should be used to prepare 500 mL of .02 M HCl?
50 mL
How many grams of MgCl2 (molar mass 95.21 g/mol) are need to prepare 250 mL of .05 M MgCl2 solution.
1.19 g MgCl2
Describe how H2O and KCl molecules would react in solution.
In solution the H+ atoms in H2O would orient themselves towards the negatively charged Cl- and the O2- atoms in H2O would orient themselves toward the positive K+.
Explain why NaCl is a good conductor when dissolved in water.
NaCl is a good conductor when dissolved in water because when it dissolves in separates into its ions Na+ and Cl-. These freely moving ions are able to conduct electricity which then causes NaCl to be a good conductor.
What would be the best procedure to separate a mixture of NaCl and H2O?
Distillation
How can you use paper chromatography to find the number of components in a sample?
Paper chromatography can be used to find the number of components in a sample because it Is able to separate small quantities of a mixture into its parts. This is done by using different solutions with various polarities as the mobile phase.
What is the major attractive force that exists when methanol is completely dissolved in water?
Hydrogen bonding
What can be said about an unknown, brittle solid that conducts electricity when fully dissolved in water?
The unknown solid is ionic. This is because in conducts electricity in solution meaning that it dissociates into its ions.
What occurs when photons of visible light are absorbed by molecules of dye?
The absorption of the photons in the visible range will cause the electrons in the dye molecules to transition between energy levels.
Explain what happens at the molecular level as CO2 molecules absorb photons of the infrared radiation.
As CO2 absorbs the infrared radiation photons, the vibrations amongst the molecules will increase. This is because the bonds are bending and stretching and infrared radiation is associated with transitions in molecular vibration levels.
What is the approximate frequency that corresponds to a wavelength of 650 nm ( 6.5*10^-7 m) ?
4.61*10^14 m
In an experiment on the photoelectric effect, a photon with a frequency of 9*10^11 s^-1 was absorbed by a sample. What is the approximate energy of this photon?
5.96*10^-22 J
How would fingerprints left on a cuvette effect the results given from the spectrophotometer?
The fingerprints left on the cuvette would scatter the lights causing less light to pass through the solution and result in a higher amount of light absorbed.
Explain how excess water left in a cuvette could skew the results of measured absorbance.
Excess water in the cuvette would cause the results of measured absorbance to be lower than expected because the water would dilute the sample solution.