Protic and Polar Aprotic Solvents
0.0(0)
Card Sorting
1/10
There's no tags or description
Looks like no tags are added yet.
Study Analytics
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
11 Terms
1
New cards
Water
Protic
2
New cards
Methanol
Protic
3
New cards
Ethanol
Protic
4
New cards
Ammonia (NH3)
Protic
5
New cards
Acetic Acid (CH3COOH)
Protic
6
New cards
Acetone (CH3COCH3)
Polar Aprotic
7
New cards
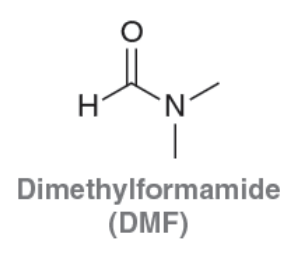
Dimethylformamide (DMF)
Polar Aprotic
8
New cards
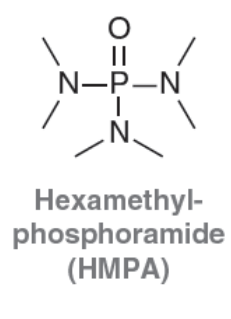
Hexamethylphosphoramide (HMPA)
Polar Aprotic
9
New cards
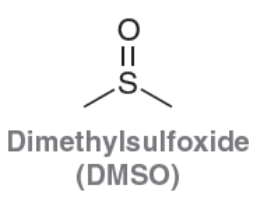
Dimethylsulfoxide (DMSO)
Polar Aprotic
10
New cards
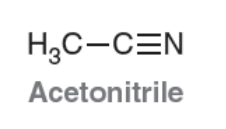
Acetonitrile
Polar Aprotic
11
New cards
Which solvent type is ideal for SN2 reactions?
polar aprotic - these solvents lack hydrogen bonding abilities, and therefore do not interact with the nucleophile, leaving it free for the backside attack.