Chemistry OCR - Module 4: Section 2 Alcohols, Haloalkanes & Analysis
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
What is a haloalkane?
An alkane with at least on halogen atom
Why is the Carbon-Halogen bond in haloalkanes polar?
Halogens are more electronegative than carbon, so the carbon-halogen bond is polar.
The δ+ carbon is electron deficient. This means it can be attacked by a nucleophile.
What is a nucleophile?
A nucleophile is an electron pair donor. It could be a negative ion or an atom with a lone pair of electrons. It donates an electron pair to somewhere without enough electrons.
OH-, CN-, and NH3 are all nucleophiles which react with haloalkanes. Water is a nucleophile too, but it reacts slowly.
How can haloalkanes be hydrolysed to make alcohols?
This is a nucleophilic substitution reaction. You have to use a warm aqueous alkali, for example sodium hydroxide, or potassium hydroxide or it will not work. The general equation is: R-X + OH- → R-OH + X-
(over the arrow is OH-/H2O and below is reflux)
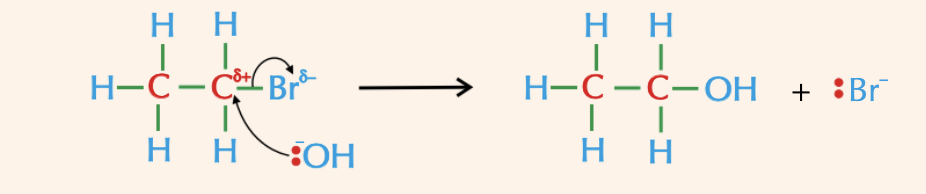
Why can water act as a nucleophile?
The water molecule is a weak nucleophile, but it will eventually substitute for the halogen, it is just a much slower reaction than one with a halogen.
You get an alcohol produced again: R-X + H2O → R-OH + H+ + X-
Why are iodoalkanes hydrolysed the fastest?
How quickly different haloalkanes are hydrolysed depends on bond enthalpy.
Weaker carbon-halogen bonds break more easily so they react faster.
Iodoalkanes have the weakest bonds, so they hydrolyse the fastest.
Fluoroalkanes have the strongest bonds, so they are the slowest at hydrolysing.
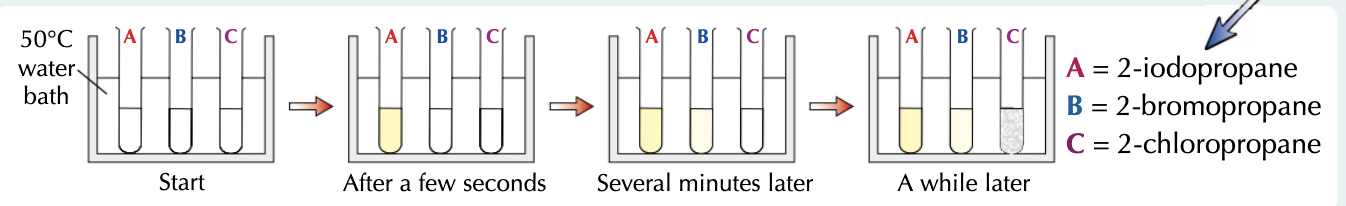
How can you compare the reactivity of chloroalkanes, bromoalkanes and iodoalkanes?
When you mix a haloalkane with water, it reacts to form an alcohol.
If you put silver nitrate solution in the mixture too, the silver ions react with the halide ions as soon as they form, giving a silver halide precipitate.
To compare the reactivities, set up three test tubes each containing a different haloalkane, ethanol (as a solvent) and silver nitrate solution (this contains the water).
A pale yellow precipitate quickly forms with 2-iodopropane, so iodoalkanes must be the most reactive haloalkanes. Bromoalkanes react slower than iodoalkanes to form a cream precipitate, and chloroalkanes form a white precipitate the slowest of all.
What are CFCs?
Chlorofluorocarbons
They are very stable, volatile, non-flammable and non-toxic. They were used a lot, e.g. in fridges, aerosol cans, dry cleaning and air conditioning, until scientists realised they were destroying the ozone layer.
Why are CFCs destroying the Ozone layer?
The ozone absorbs UV radiation which can cause sunburns or skin cancer.
Ozone formed naturally, when an oxygen molecule is broken down into two free radicals by ultraviolet radiation. The free radicals attack other oxygen molecules forming ozone: O2 → O + O → O2 + O → O3
The holes in the ozone are formed because CFC’s in the upper atmosphere absorb UV radiation and split to form chlorine free radicals. These free radicals catalyse the destruction of ozone, they destroy ozone molecules and are then regenerated to destroy more ozone.
CF2Cl2(g) → ·CF2Cl(g) + Cl·(g)
Cl·(g) + O3(g) → O2(g) + ClO·(g)
ClO·(g) + O(g) → O2(g) + Cl·(g)
Overall: O3(g) + O(g) → 2O2(g)
How can nitrogen oxides also break down ozone?
NO· free radicals from nitrogen oxides destroy ozone too. Nitrogen oxides are produced by car and aircraft engines and thunderstorms. NO· free radicals affect ozone in the same way as chlorine radicals.
What alternatives have been developed for CFCs?
HCFCs (hydrogchlorofluorocarbons) and HFCs (hydrofluorocarbons) are being used as temporary alternatives to CFCs until safer products are developed.
Hydrocarbons are also used.
HCFCs are broken down in the atmosphere in 10-20 years. The still damage the ozone layer, but their effect is much smaller than CFCs.
HFCs are also broken down in the atmosphere. Unlike HCFCs, they do not contain chlorine, so they do not affect the ozone layer.
HFCs and HCFCs are greenhouses gases and they are 1000 times worse than carbon dioxide.
Some hydrocarbons are being used in fridges but these are greenhouse gases too.
Most aerosols have been replaced by pump spray systems or use nitrogen as a propellant. Many industrial fridges and freezers now use ammonia as the coolant gas, and carbon dioxide is used to make foamed polymers.
What is the general formula for alcohols?
CnH2n+2OH
What are alcohols divided into?
Primary - OH is bonded to a carbon with 1 alkyl group
Secondary - OH is bonded to a carbon with 2 alkyl group
Tertiary - OH is bonded to a carbon with 3 alkyl groups
Why are alcohols generally polar molecules?
Alcohols are generally polar molecules due to the electronegative hydroxyl group which pulls the electrons in the C-H bond away from the carbon.
The electronegative oxygen in the polar hydroxy group draws electron density away from the hydrogen, giving it a slightly positive charge. This positive charge can attract the lone pairs on an oxygen from a neighbouring molecule, forming hydrogen bonds.
Due to the hydrogen bonds in alcohols, what properties do they obtain?
When you mix an alcohol with water, hydrogen bonds form between the -OJ and H2O. If it is a small alcohol, hydrogen bonding lets it mix freely with water, it is soluble in water.
In larger alcohols, most of the molecule is a non-polar carbon chain, so there is less attraction for the polar H2O molecules. This means that as alcohols increase in size, their solubility in water decreases.
Alcohols also from hydrogen bonds with each other. Hydrogen bonding is the strongest kind of intermolecular force, so it gives alcohols a relatively low volatility compared to non-polar compounds.
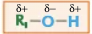
How can the -OH be swapped for a halogen to make a haloalkane?
Alcohols will react with compounds containing halide ions in a substitution reaction.
The hydroxyl (-OH) group is replaced by the halide, so the alcohol is transformed into a haloalkane.
The reaction also requires the addition of an acid such as H2SO4.
How can alcohols be dehydrated to form alkenes?
You can make alkenes by eliminating water from alcohols in an elimination reaction. (An elimination reaction where water is eliminated is called a dehydration reaction.)
The alcohol is mixed with an acid catalyst, either concentrated sulfuric acid (H2SO4) or concentrated phosphoric acid (H3PO4). The mixture is then heated.
When an alcohol dehydrates it eliminates water.
The water molecule is made up from hydroxyl group and a hydrogen atom that was bonded to a carbon atom adjacent to the hydroxyl carbon.
This means that often there are two possible alkene products from one elimination reaction depending on which side of the hydroxyl group the hydrogen is eliminated from.
If any of the alkene products can form E/Z isomers, then a mixture of both isomers will form.
How does burning alcohols oxidise them?
In a combustion reaction, the C-C and C-H bonds are broken as the alcohol is completely oxidised to make carbon dioxide and water.
How does the structure of the alcohol impact how much an alcohol can be oxidised?
You can use the oxidising agent acidified dichromate(VI) (Cr2O7-2/H+, e.g. K2Cr2O7/H2SO4) to mildly oxidising alcohols.
The orange dichromate(VI) ion is reduced to the green chromium(III) ion, Cr3+.
Primary alcohols are oxidised to aldehydes and then to carboxylic acids.
Secondary alcohols are oxidised to ketones only.
Tertiary alcohols will not be oxidised.
Aldehydes and ketones are carbonyl compounds, they have the functional group C=O. Their general formula is CnH2nO.
Aldehydes have a hydrogen and one alkyl group attached to the carbonyl carbon atom.
Ketones have two alkyl groups attached to the carbonyl carbon atom.
Why do primary alcohols oxidise into aldehydes and carboxylic acids?
Primary alcohols can be oxidised twice, first to form aldehydes and the the second to from carboxylic acids.
How can you control how far the alcohol is oxidised?
Control the reaction conditions
E.g.
Gently heating ethanol with potassium dichromate(VI) solution and sulfuric acid in a test tube should produce “apple” smelling ethanal. However, it is really tricky to control the amount o f heat and the aldehyde is usually oxidised to form “vinegar” smelling ethanoic acid.
To get just the aldehyde, you need to get it out of the oxidising solution as soon as it is formed. You can do this by gently heating excess alcohol with a controlled amount of oxidising agent in distillation apparatus, so the aldehyde (which boils at a lower temperature than the alcohol) is distilled off immediately.
To produce the carboxylic acid, the alcohol has to be vigorously oxidised. The alcohol is mixed with excess oxidising agent and heated under reflux.
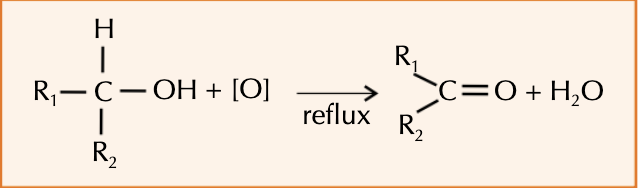
Why do secondary alcohols oxidise to ketones?
Refluxing a secondary alcohol with acidified dichromate(VI) will produce a ketone.
Ketones cannot be oxidised easily, so even prolonged refluxing will not produce anything more.
Why are tertiary alcohols not oxidised easily?
Tertiary alcohols do not react with potassium dichromate(VI) at all the solution stays orange.
The only way to oxidise tertiary alcohols is by burning them.