34.2 phenylamine and azo compounds
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
12 Terms
Describe the preparation of phenylamine
Nitration of benzene
Reduction of nitrobenzene with hot Sn/ concentrated HCl
Deprotonation: add NaOH
List the 2 ways that phenylamine can react
Reaction of phenylamine with Br2 at room temperature
Reaction of phenylamine with H2O2 or NaNO2
Explain how phenylamine reacts with Br2
Electrophilic substitution
reacts at RTP
Aqueous Br2
Lone pair of electrons in nitrogen atom in phenylamine amine donate electron density into benzene ring
Delocalisation of electrons causes increased electron density in the benzene ring
Therefore benzene ring becomes activated and more readily attacked by electrophiles.
Incoming electrophiles directed to 2,4,6 positions
Explain the reaction of phenylamine with HNO2
The amine group of phenylamine reacts with nitrous acid at temperature below 10 to form diazonium salts
Since nitric acid is unstable it has to be made in the test tube by reacting sodium nitrite and dilute acid.
Upon further heating with water, diazonium salts form phenol.
Describe and explain the relative basicities of aqueous ammonia, ethylamine and phenylamine
(Strongest) Ethylamine
Ammonia
(Weakest) Phenylamine
In ethylamine, the electron-donating alkyl group donates electron density to the N atom causing its lone pair to become more available to form a dative covalent bond with a proton.
Ammonia lacks an electron-donating group
Hence, it is less basic than ethylamine
However it is more basic than phenylamine as the lone pair on the nitrogen is not delocalised
In phenylamine, the lone pair of electrons overlap with conjugated system on benzene ring and become delocalised.
Therefore, lone pair of electrons become less readily available to form bond with proton.
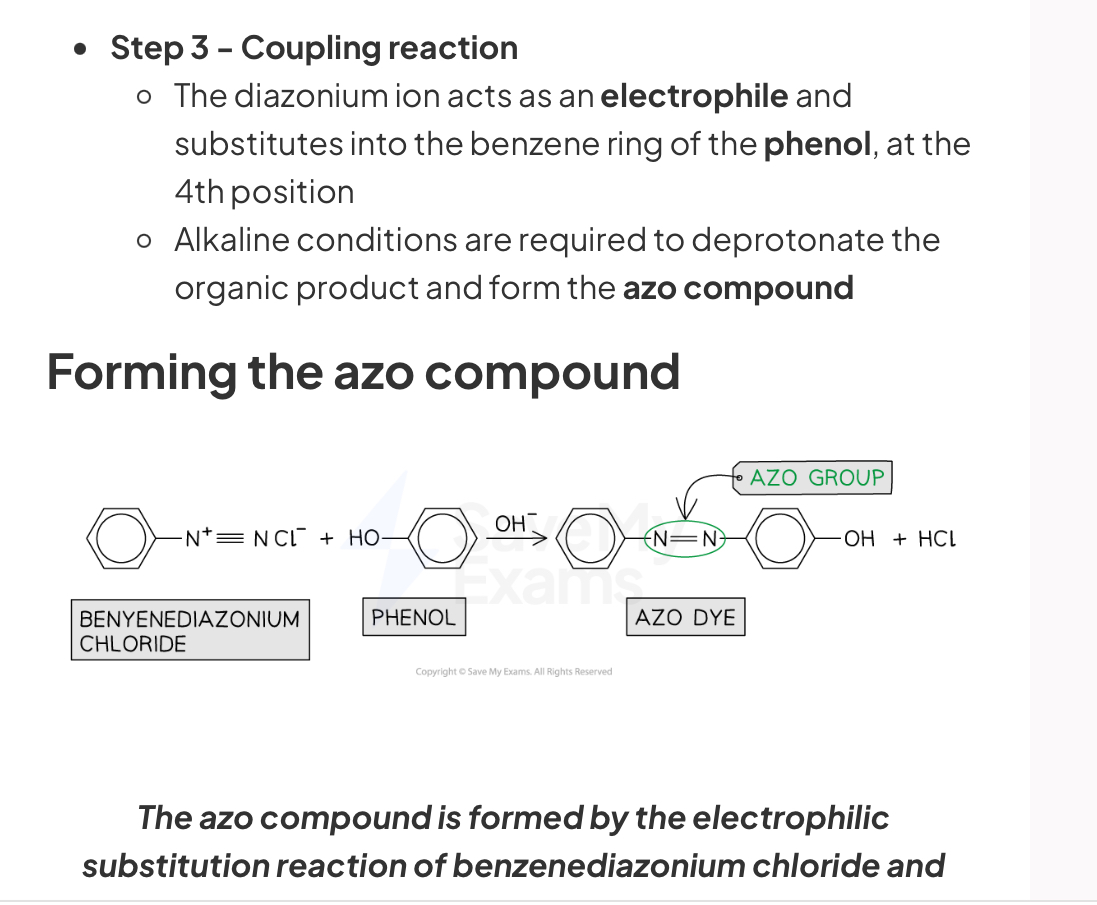
How are azo compounds formed?
Coupling reaction of benzenediazonium chloride salt with alkaline phenol
Diazonium ion acts as an electrophile and substitutes into the benzene ring of phenol, at the 4th position
Alkaline conditions are required to deprotonate the organic product and form the azo compound
Why are azo compounds so stable?
Delocalised electrons in the pi bonding systems of 2 benzene rings are extended through N=N which acts as a bridge between the 2 rings
What is the azo group?
R1-N=N-R2
How are azo dyes made?
Formation of nitrous acid
Diazotisation: reaction between nitrous acid and phenylamine to form diazonium ion
Coupling reaction: The diazonium ion acts as an electrophile and substitutes into the benzene ring of the phenol, at the 4th position
Alkaline conditions are required to deprotonate the organic product and form the azo compound
Forming the azo compound: The azo compound is formed by the electrophilic substitution reaction of benzenediazonium chloride and phenol
The delocalised electrons in the π bonding systems of the two benzene rings are extended through the -N=N- which acts as a bridge between the two rings
How can other azo dyes be made?
Via a similar route
the yellow dye can be formed from the coupling reaction between benzenediazonium chloride and C6H5N(CH3)2 instead of phenol (C6H5OH)
Draw the coupling reaction between benzenediazonium choride and phenol