Bio: Cellular Respiration and Fermentation
5.0(2)
Studied by 28 peopleCard Sorting
1/71
Last updated 12:22 PM on 1/22/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
72 Terms
1
New cards
Autotrophs
* Produce their own organic molecules through photosynthesis.
2
New cards
Heterotrophs
* Eat organic compounds produced by other organisms
3
New cards
Cellular Respiration
* the process of releasing the energy contained in organic molecules
(mainly Glucose) to do work.
* The process uses the energy from the
organic, biological macromolecules to make ATP.
* a series of coordinated enzyme-catalyzed reactions that capture energy from biological macromolecules.
* releases heat (unusable energy) and **free electrons**(used to produce ATP).
(mainly Glucose) to do work.
* The process uses the energy from the
organic, biological macromolecules to make ATP.
* a series of coordinated enzyme-catalyzed reactions that capture energy from biological macromolecules.
* releases heat (unusable energy) and **free electrons**(used to produce ATP).
4
New cards
Oxidation
* loss of electrons(LEO)
5
New cards
Reduction
* gain of electron(GER)
6
New cards
Dehydrogenization
* lost electrons are accompanied by protons(***hydrogen***)
* a hydrogen atom is lost (1 electron, 1 proton)
* To follow the e-, follow the H’s
* a hydrogen atom is lost (1 electron, 1 proton)
* To follow the e-, follow the H’s
7
New cards
Redox Reactions
* e- carry energy from one molecule to another
8
New cards
Nicotinamide adenine dinucleotide
* NAD+
* NADH
* NAD+
* NADH
\*Meaning of NAD+
* Oxidized form?(ready to accept electrons)
* Reduced Form?(accepted electrons)
* Oxidized form?(ready to accept electrons)
* Reduced Form?(accepted electrons)
9
New cards
Flavin adenine dinucleotide
* FAD+
* FADH2
* FAD+
* FADH2
\*Meaning of FAD+
* Oxidized form?(ready to accept electrons)
* Reduced Form?(accepted electrons
* Oxidized form?(ready to accept electrons)
* Reduced Form?(accepted electrons
10
New cards
Aerobic Respiration
* Final electron acceptor is oxygen (O2 )
11
New cards
Anaerobic Respiration
* Final electron acceptor is an inorganic molecule (not O2 )
12
New cards
Fermentation
* Final electron acceptor is an organic molecule, converted into lactic acid or ethanol + CO2
13
New cards
Substrate-level phosphorylation
* A mechanism for synthesis of ATP
* Transfer phosphate group directly to ADP
* Uses an enzyme (kinase)
* During glycolysis & Krebs (Citric Acid) Cycle
* Transfer phosphate group directly to ADP
* Uses an enzyme (kinase)
* During glycolysis & Krebs (Citric Acid) Cycle
14
New cards
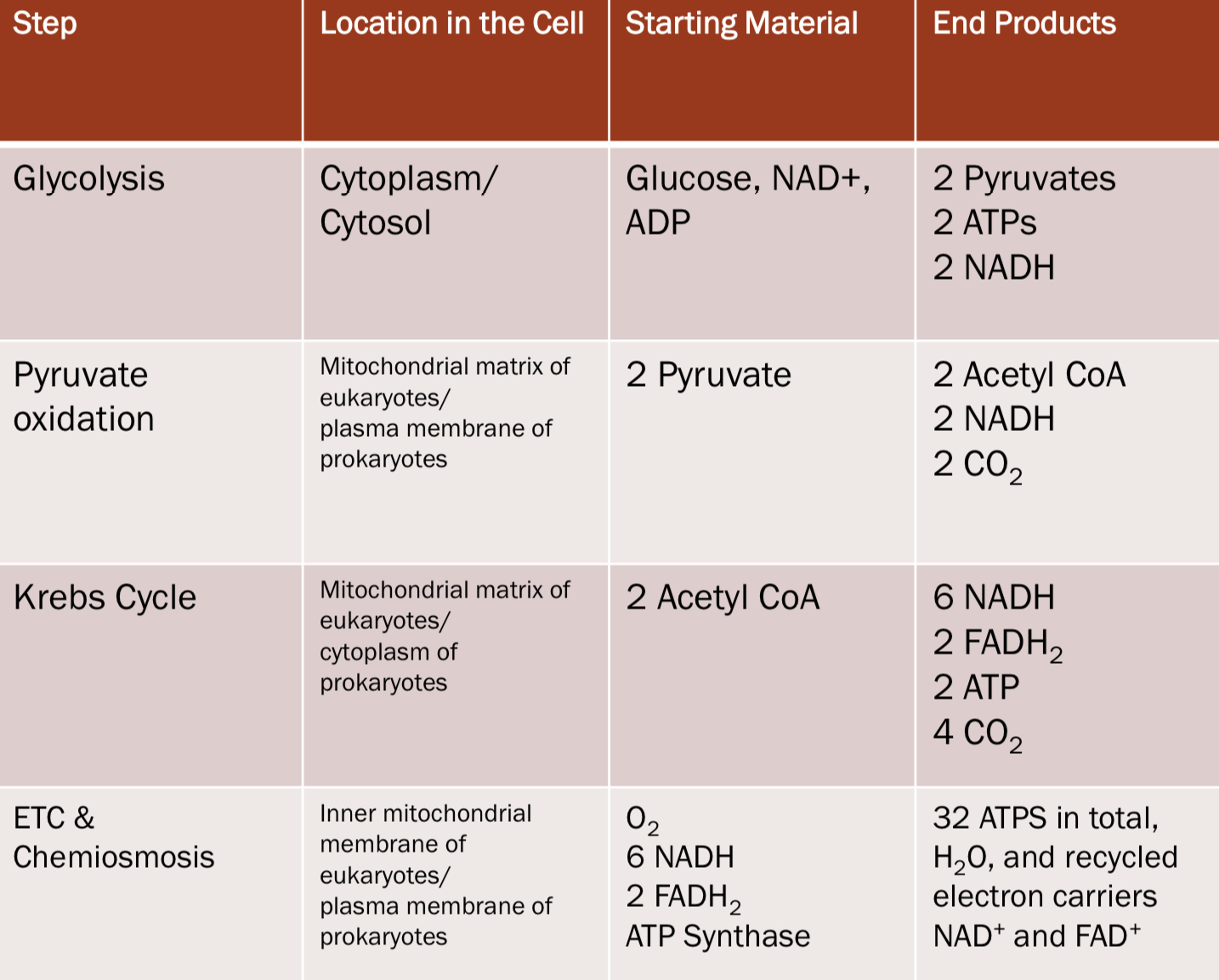
Glycolysis
* occurs in the **cytosol/cytoplasm**
* converts 1 Glucose(6 carbons) to 2 Pyruvate(3 carbons)
* can be Aerobic(32 ATP produced) or Anaerobic(2 ATP produced) so it occurs with/out the presence of O2
* converts 1 Glucose(6 carbons) to 2 Pyruvate(3 carbons)
* can be Aerobic(32 ATP produced) or Anaerobic(2 ATP produced) so it occurs with/out the presence of O2
15
New cards
Energy Investment Stage(Priming Reaction)
* During this stage, __2 ATP molecules are required__ to act as the activation energy for each glucose molecule that enters the process.
* The ATPs are used to phosphorylate glucose. The phosphorylation makes glucose unstable.
* This ultimately leads to the breaking of glucose into 2 G3P molecules.
* First to Fifth step
* The ATPs are used to phosphorylate glucose. The phosphorylation makes glucose unstable.
* This ultimately leads to the breaking of glucose into 2 G3P molecules.
* First to Fifth step
16
New cards
\*Step 1
^^? Hexokinase^^
? Glucose 6-phosphate
^^? Hexokinase^^
? Glucose 6-phosphate
* _____________ transfers a phosphate group from ATP to glucose, making it more chemically reactive. The charged phosphate also traps the sugar in the cell.
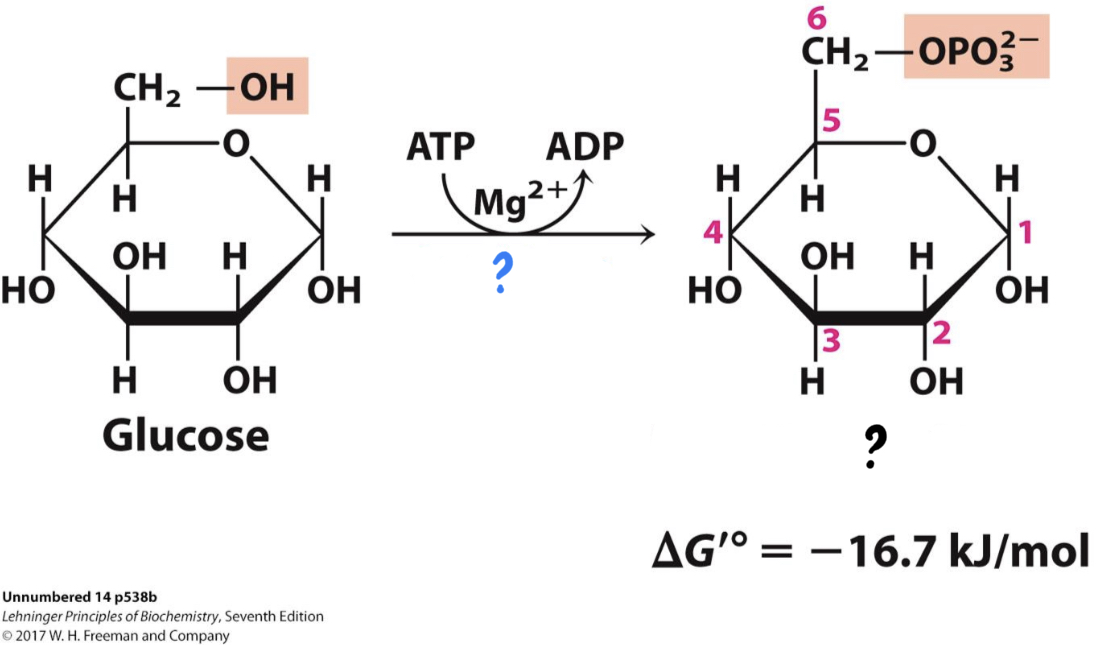
17
New cards
? ATP
?? ADP
?? ADP
18
New cards
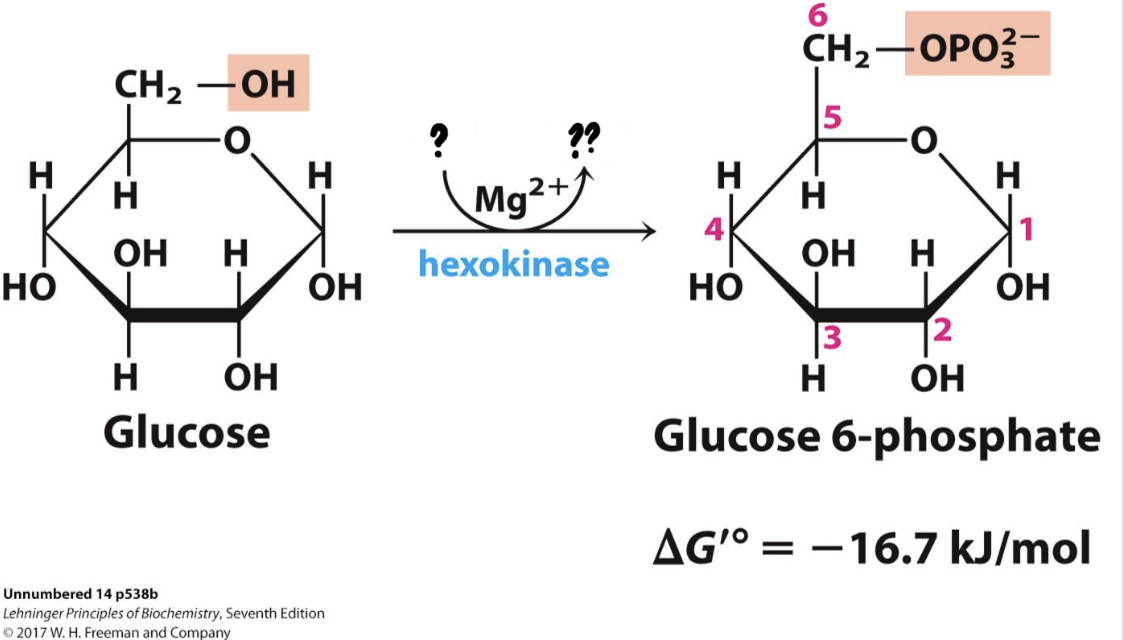
\*Step 2
^^? Phosphohexose isomerase^^
? Fructose 6-phosphate
^^? Phosphohexose isomerase^^
? Fructose 6-phosphate
* This step from phosphoglucose to phosphofructose allows the eventual split of the sugar into two three-carbon molecules
19
New cards
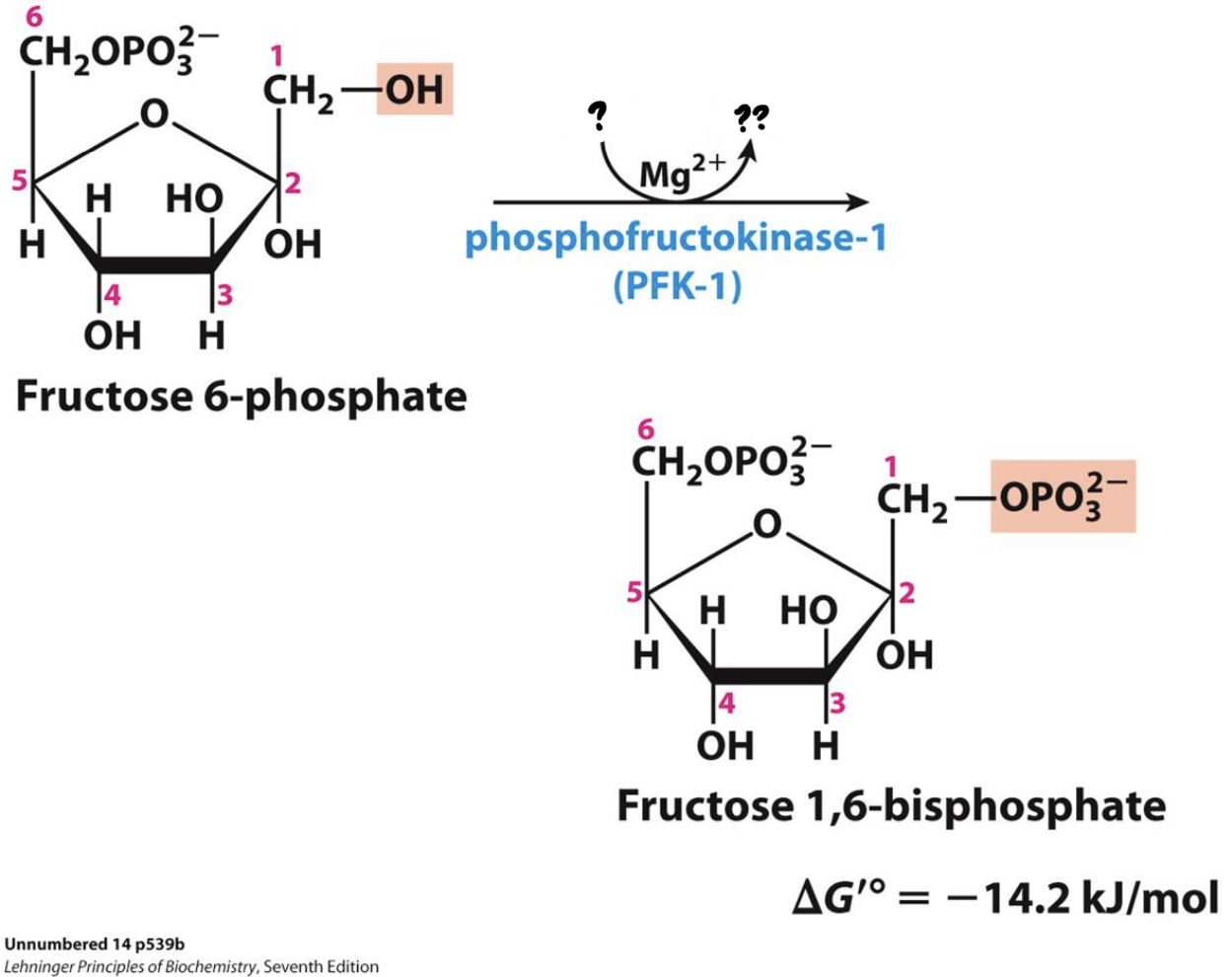
\*Step 3
^^? Phosphofructokinase -1 (PFK-1)^^
? Fructose 1,6-bisphosphate
^^? Phosphofructokinase -1 (PFK-1)^^
? Fructose 1,6-bisphosphate
* ^^_____________________^^ transfers a phosphate group from ATP to the opposite end of the sugar, __investing a second molecule of ATP__. This is a key step for the regulation of glycolysis.
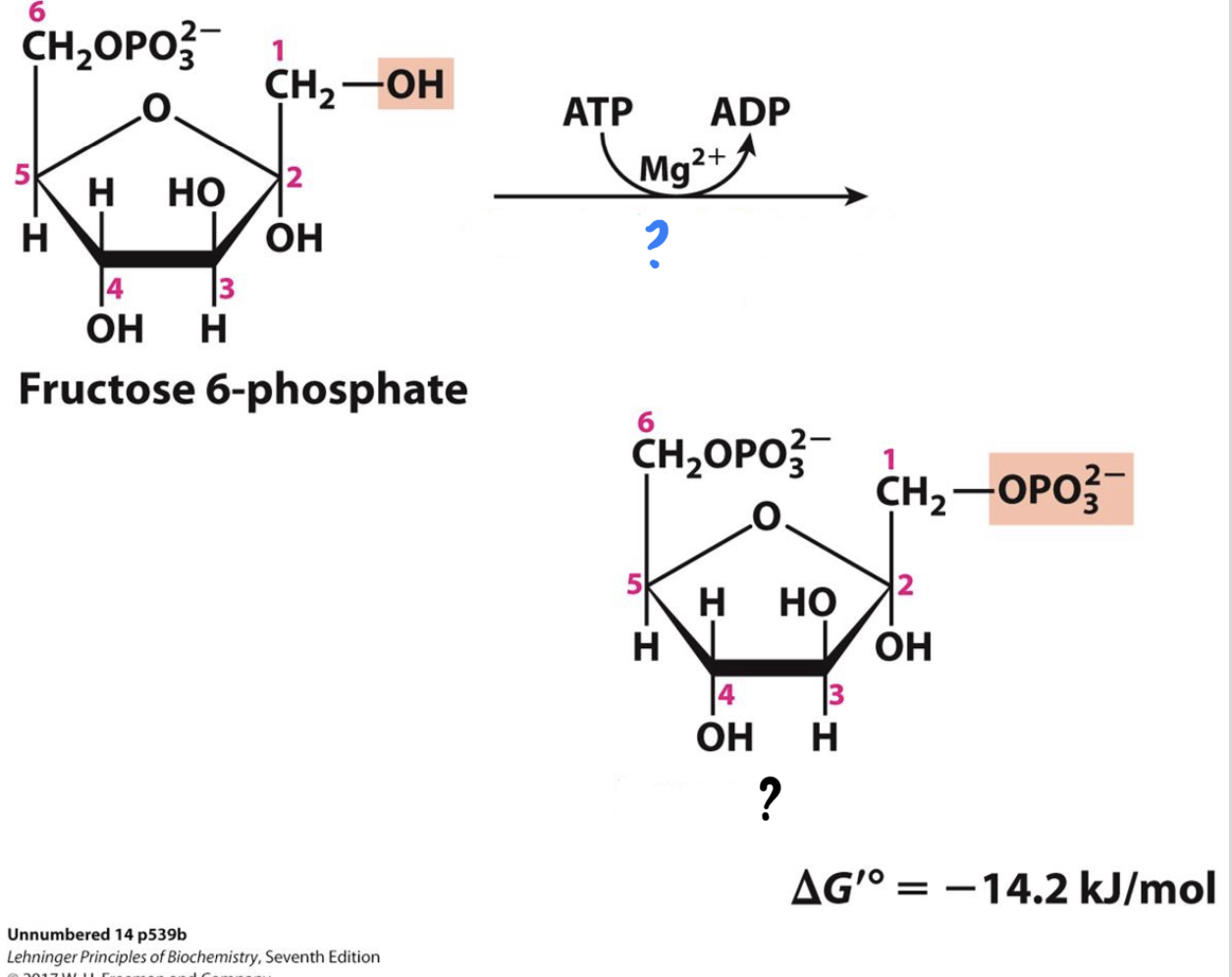
20
New cards
? ATP
?? ADP
?? ADP
21
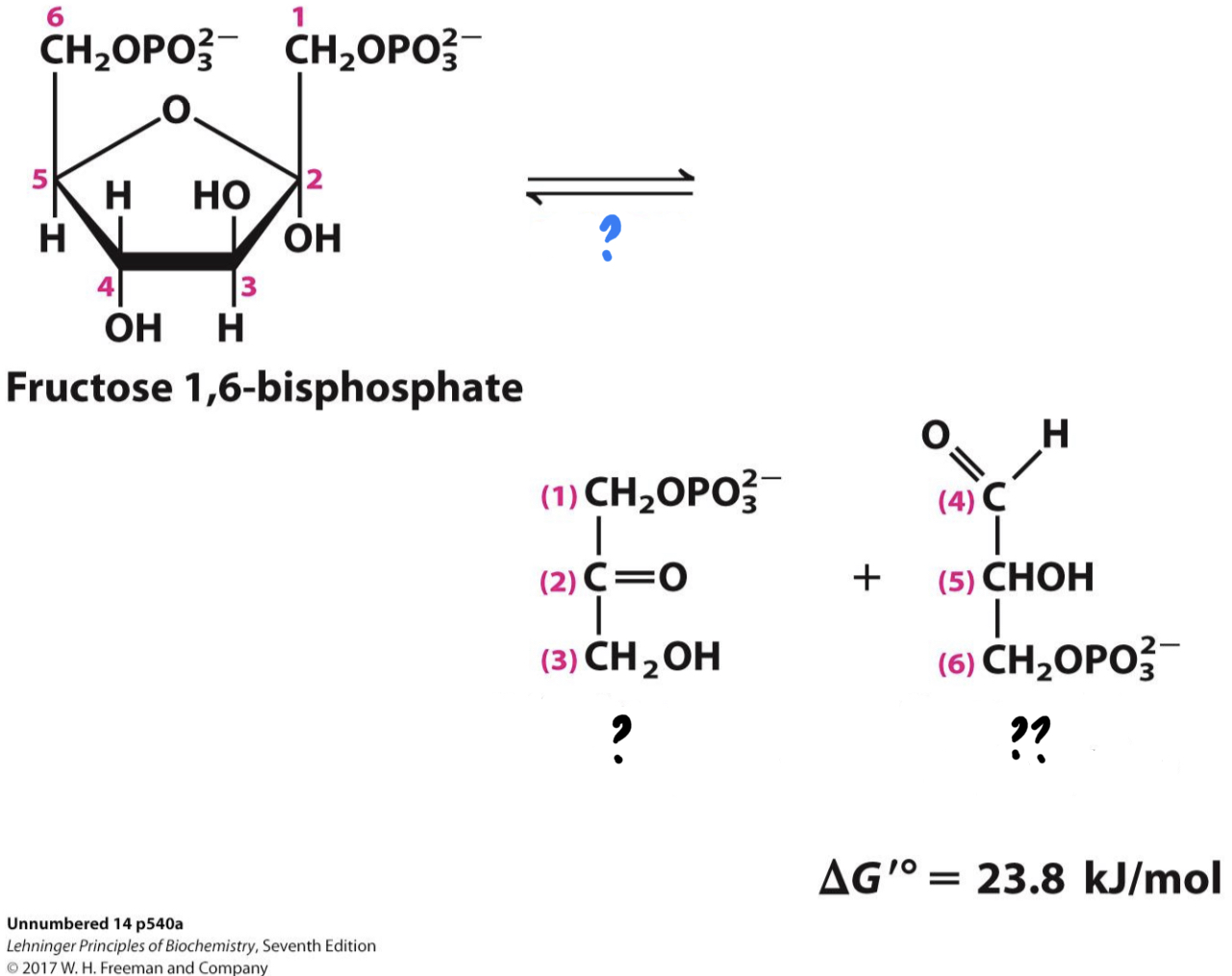
New cards
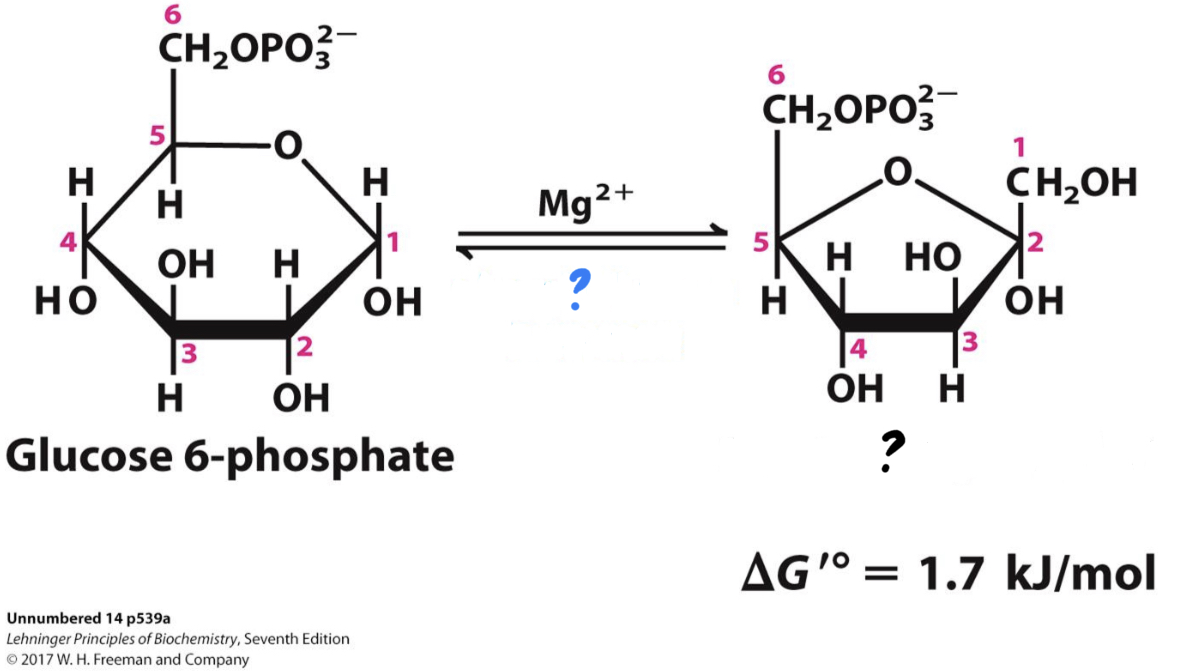
\*Step 4
^^? Aldolase^^
? Dihydroxyacetone phosphate(DHP)
?? Glyceraldehyde 3-phosphate(G3P)
^^? Aldolase^^
? Dihydroxyacetone phosphate(DHP)
?? Glyceraldehyde 3-phosphate(G3P)
* _________ cleaves the sugar molecule into two different three-carbon sugars.
22
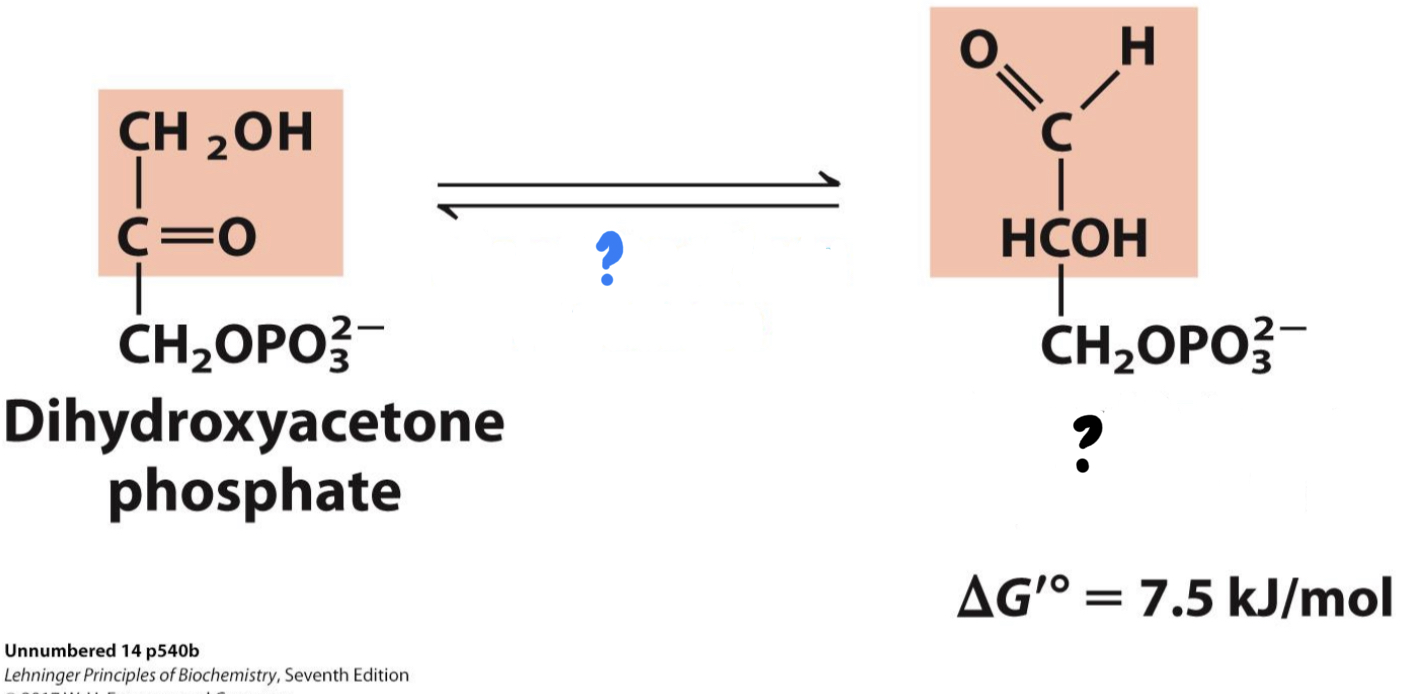
New cards
\*Step 5
^^? Triose Phosphate Isomerase^^
? Glyceraldehyde 3-phosphate(G3P)
^^? Triose Phosphate Isomerase^^
? Glyceraldehyde 3-phosphate(G3P)
\
23
New cards
Energy Payoff Stage
* During this stage, each of the two G3P molecules formed during the energy investment phase is oxidized.
* The energy and electrons from the 2 G3P molecules are used to create 2 molecules of NADH, 4 ATP molecules per glucose(*though substrate-level phosphorylation*), and 2 molecules of Pyruvate
* Sixth to Tenth step
* The energy and electrons from the 2 G3P molecules are used to create 2 molecules of NADH, 4 ATP molecules per glucose(*though substrate-level phosphorylation*), and 2 molecules of Pyruvate
* Sixth to Tenth step
24
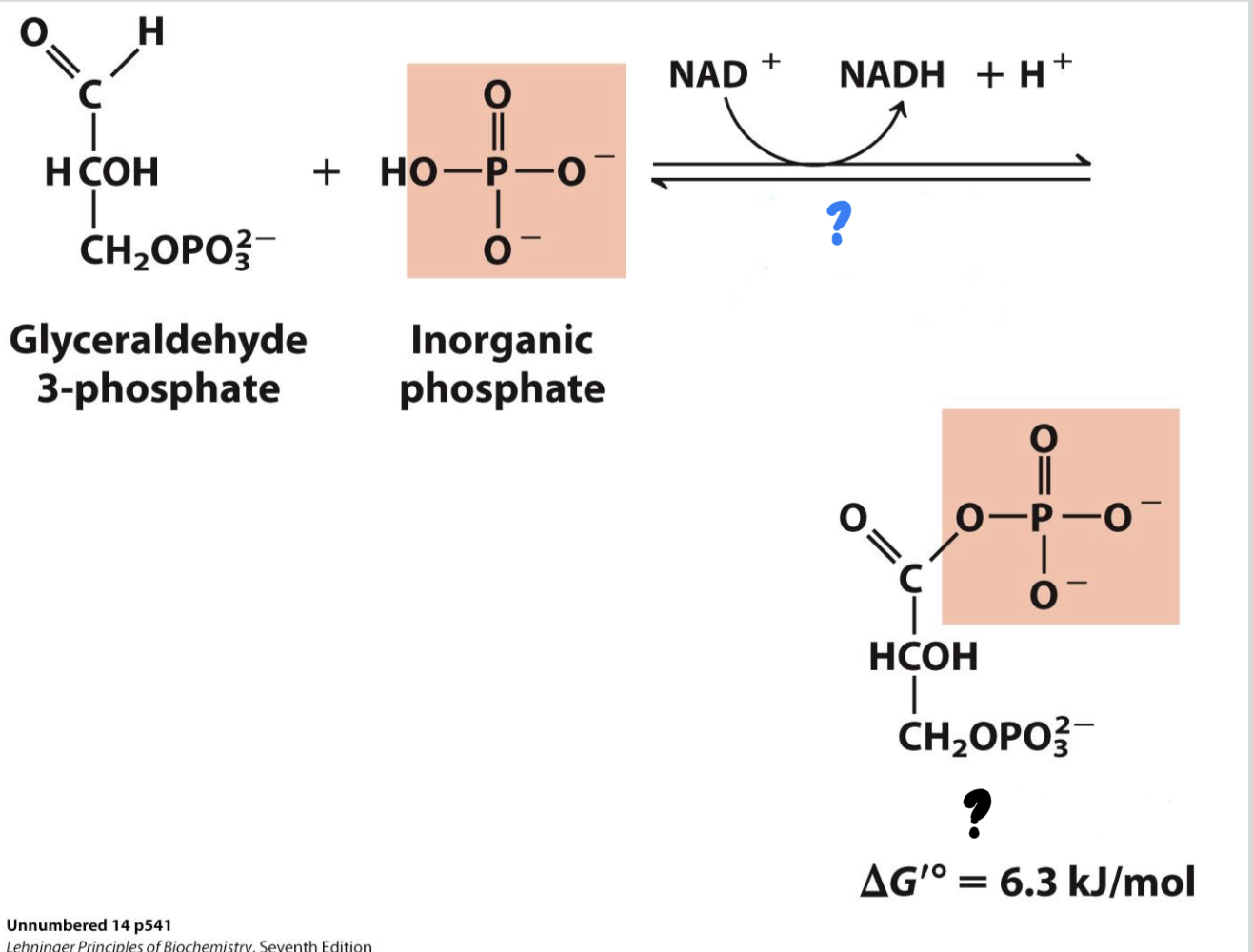
New cards
\*Step 6
^^? Glyceraldehyde 3-phosphate dehydrogenase^^
? 1,3-Bisphophoglycerate
^^? Glyceraldehyde 3-phosphate dehydrogenase^^
? 1,3-Bisphophoglycerate
* Two sequential reactions: (1) G3P is oxidized by transferring electrons to NAD+, forming NADH. (2) Using energy from this exergonic redox reaction, a phosphate group is attached to the oxidized substrate, making a high- energy product.
25
New cards
? NAD+
?? NADH(x2) + H+
?? NADH(x2) + H+
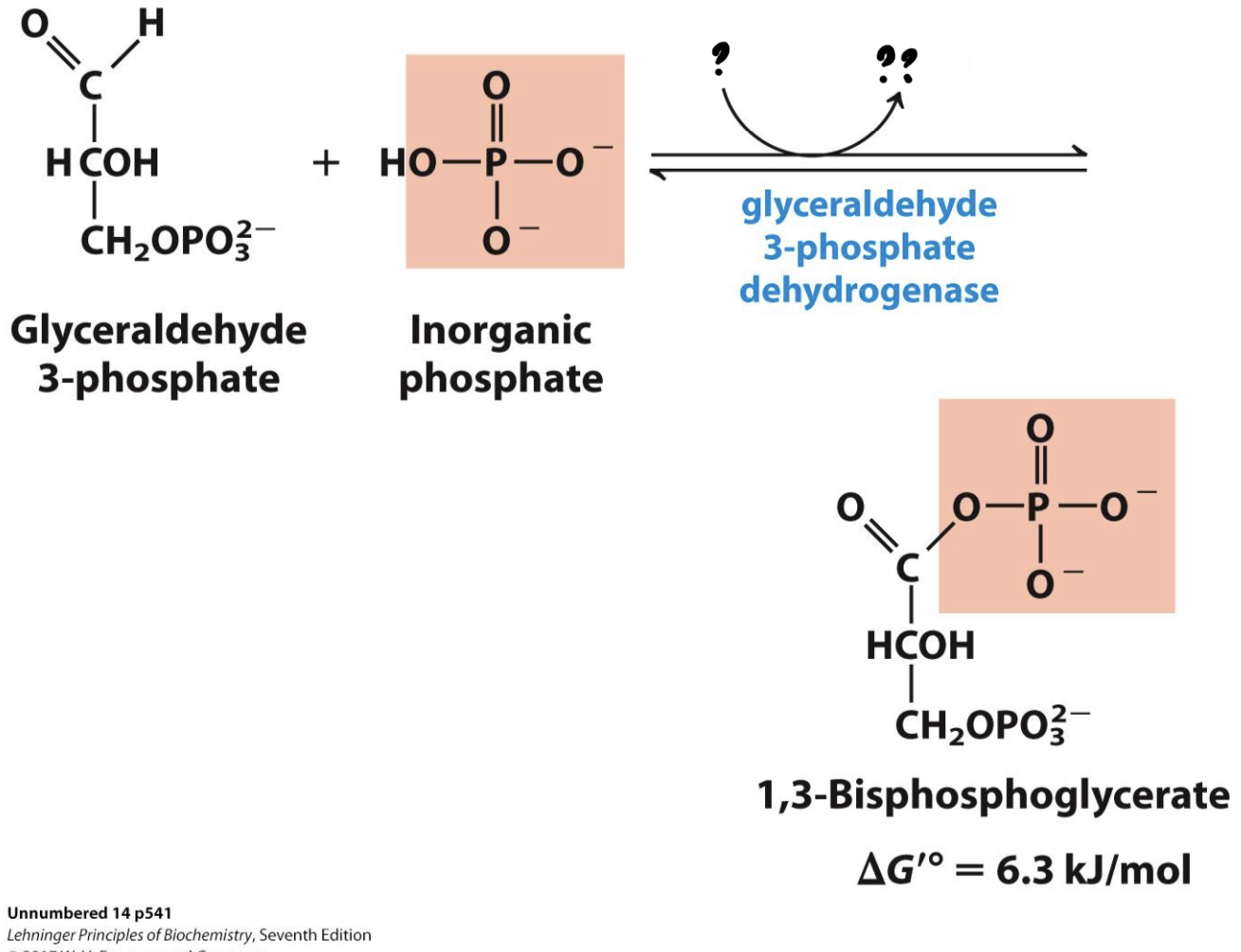
26
New cards
\*Step 7
? ^^Phosphoglycerate kinase^^
? 3-Phosphoglycerate
? ^^Phosphoglycerate kinase^^
? 3-Phosphoglycerate
* The phosphate group is transferred to ADP (substrate-level phosphorylation) in an exergonic reaction.
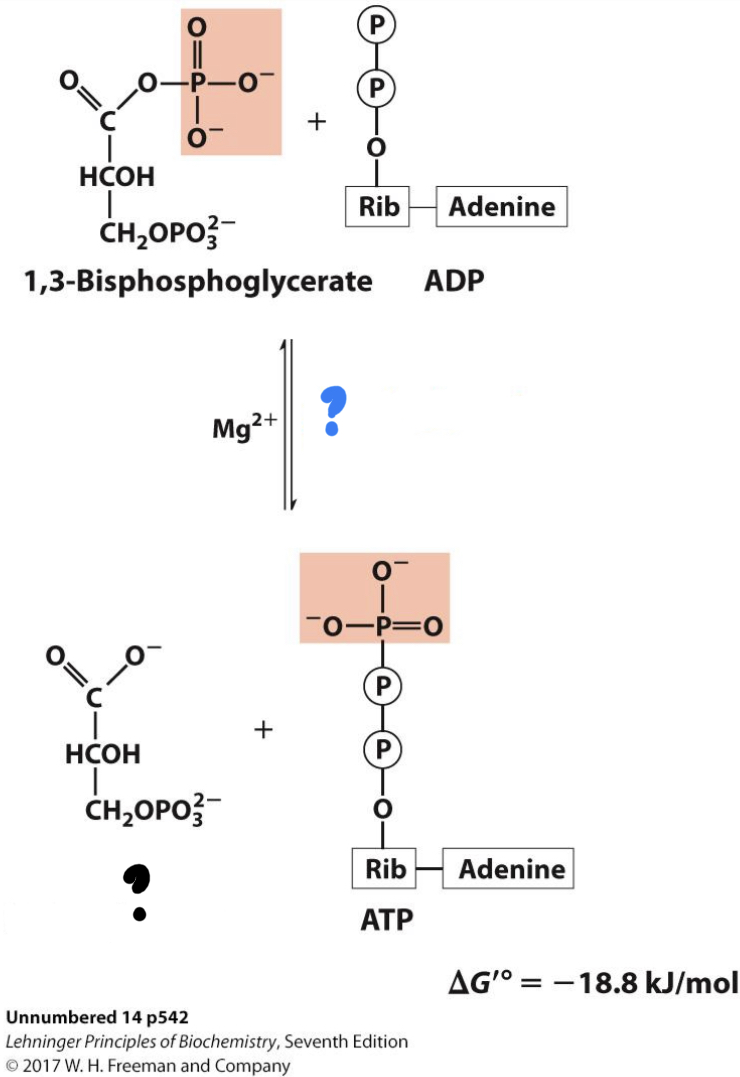
27
New cards
? ADP(x2)
?? ATP(x2)
?? ATP(x2)
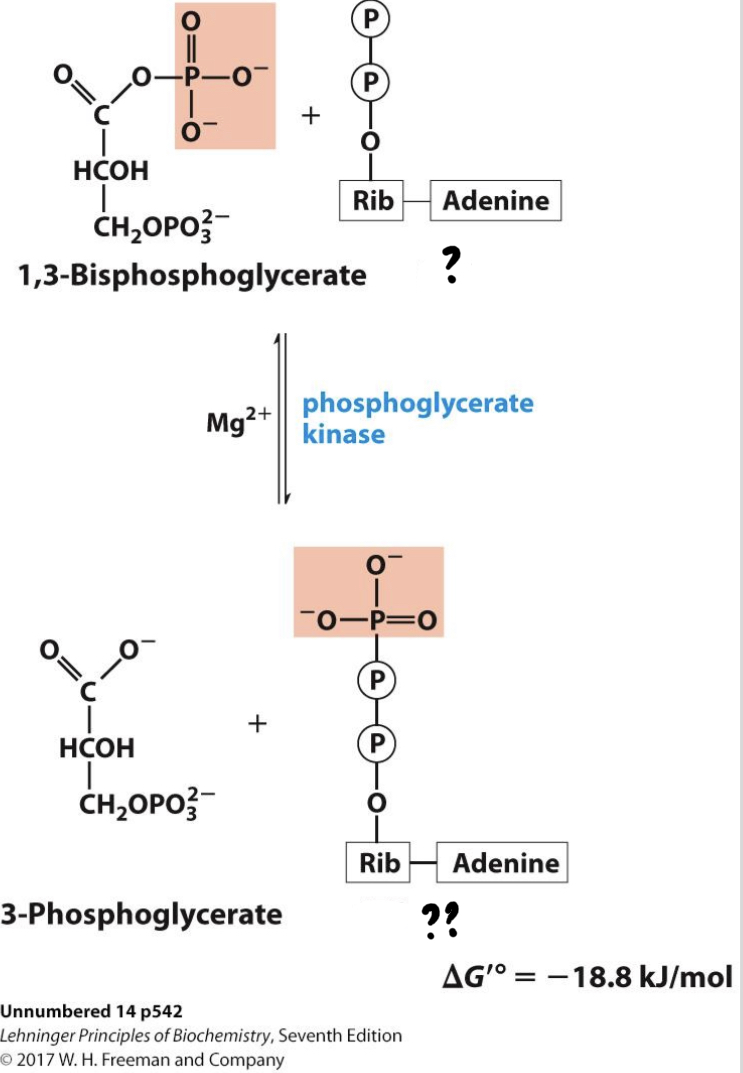
28
New cards
\*Step 8
^^? Phosphoglycerate mutase^^
? 2-Phosphoglycerate
^^? Phosphoglycerate mutase^^
? 2-Phosphoglycerate
* ^^_______________^^ relocates the remaining phosphate group.
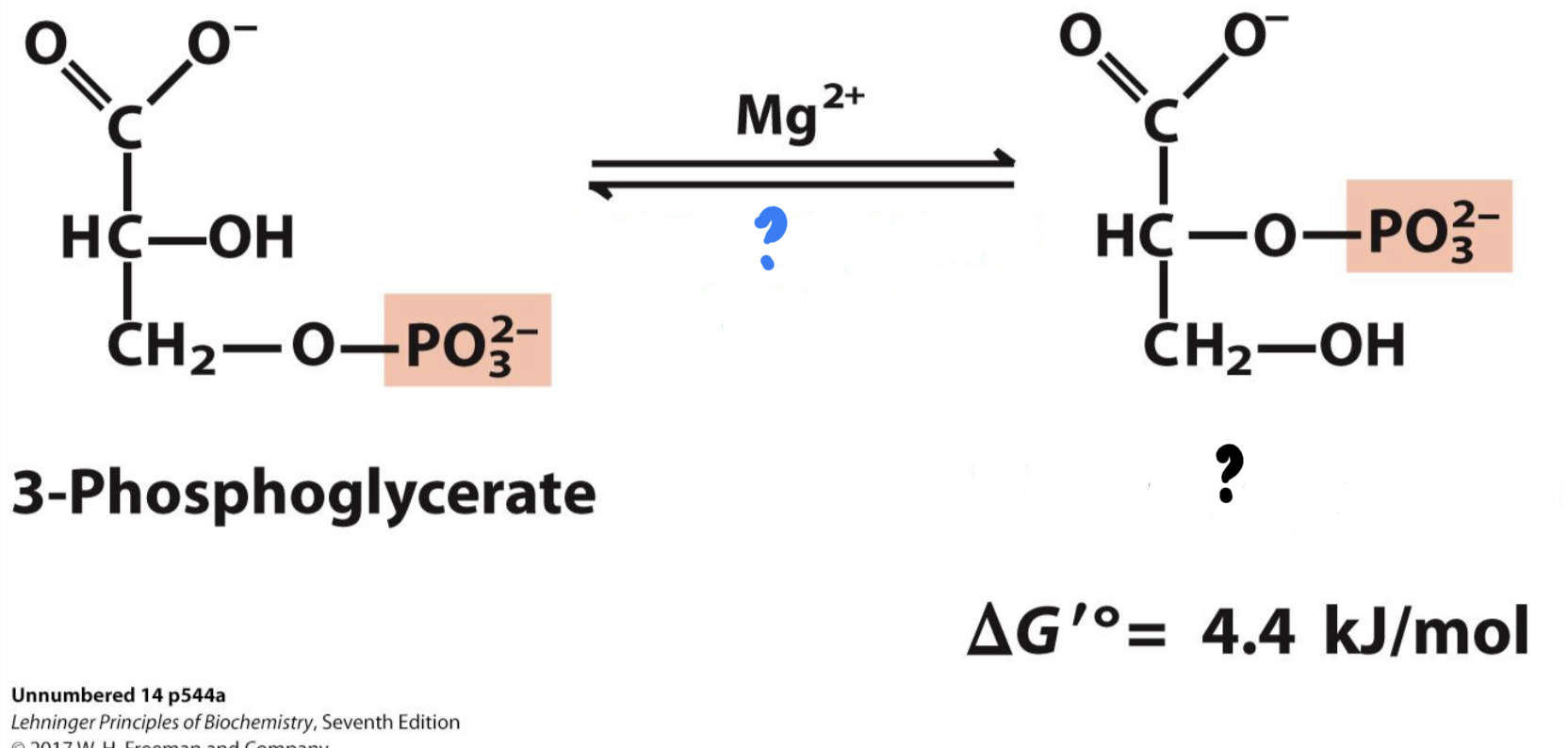
29
New cards
\*Step 9
^^? Enolase^^
? Phosphoenolpyruvate
^^? Enolase^^
? Phosphoenolpyruvate
* ^^_____^^ causes a double bond to form in the substrate by extracting a water molecule, yielding _______________, a compound with very high potential energy.
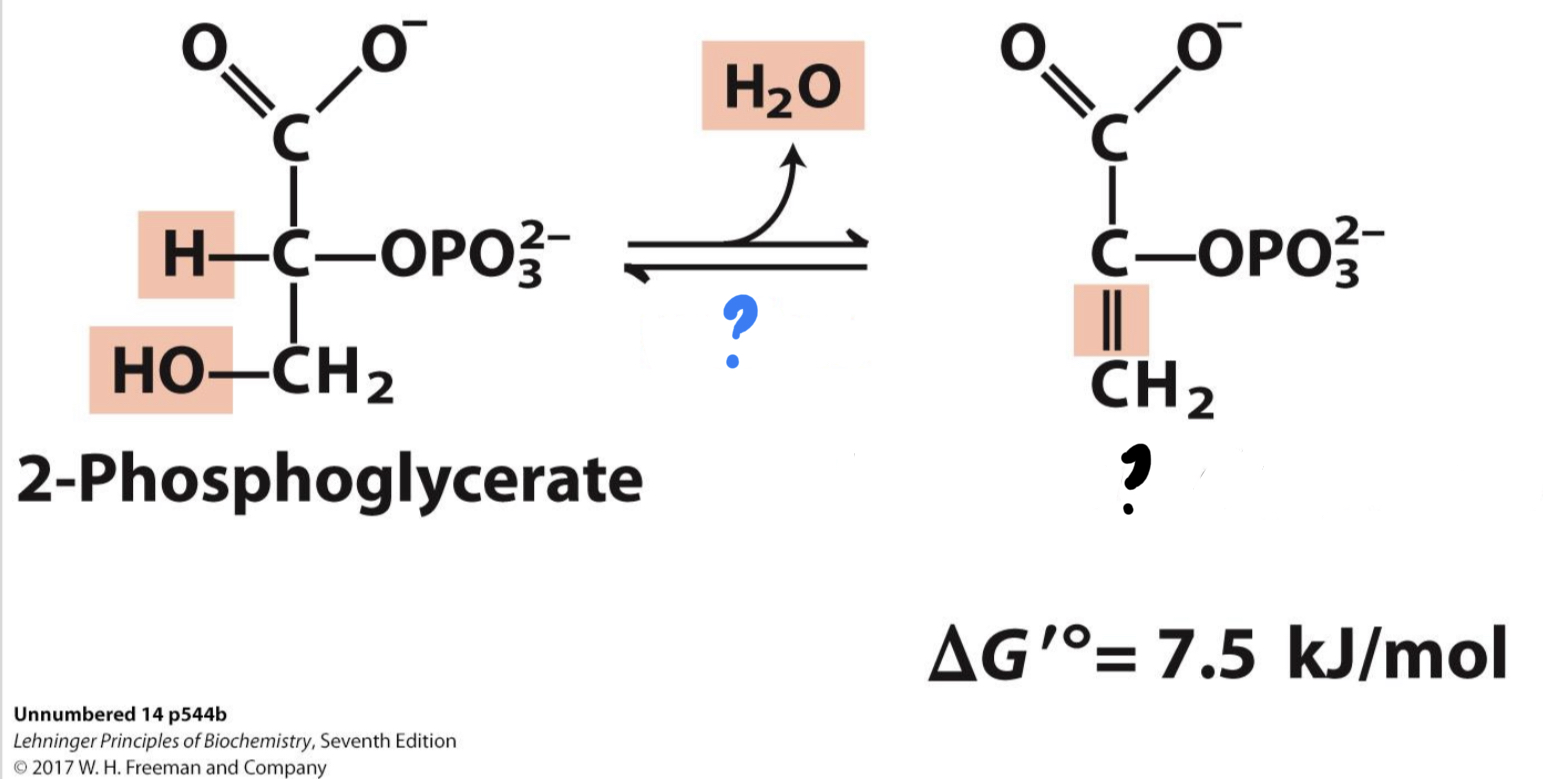
30
New cards
? H2O
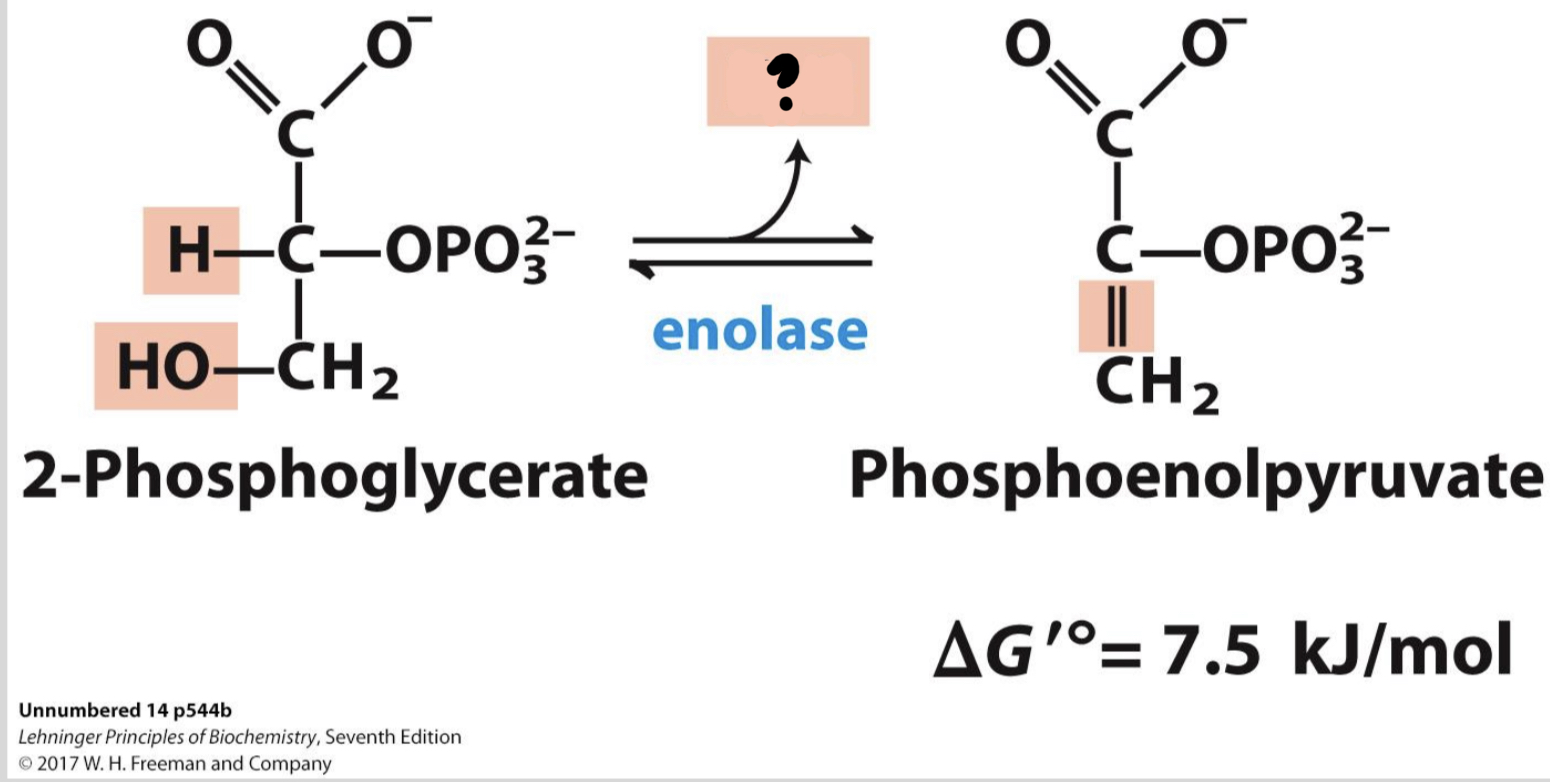
31
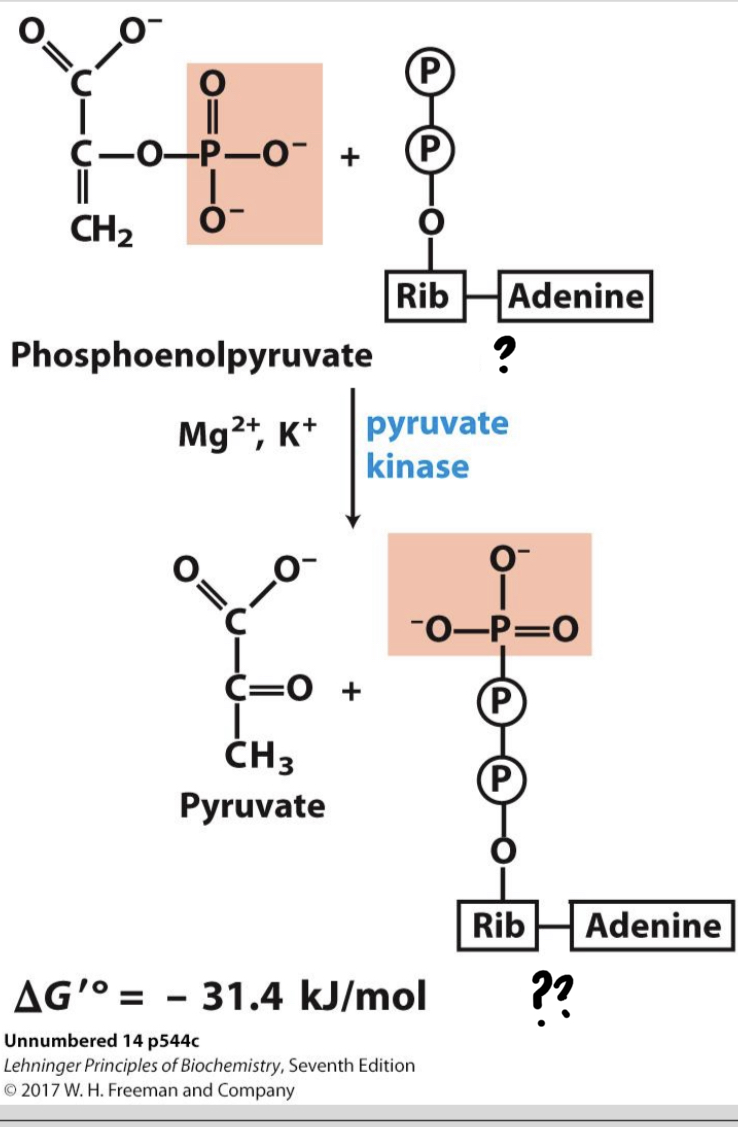
New cards
\*Step 10
^^? Pyruvate kinase^^
? Pyruvate
^^? Pyruvate kinase^^
? Pyruvate
* The phosphate group is transferred from PEP to ADP (a second example of substrate-level phosphorylation), forming ________
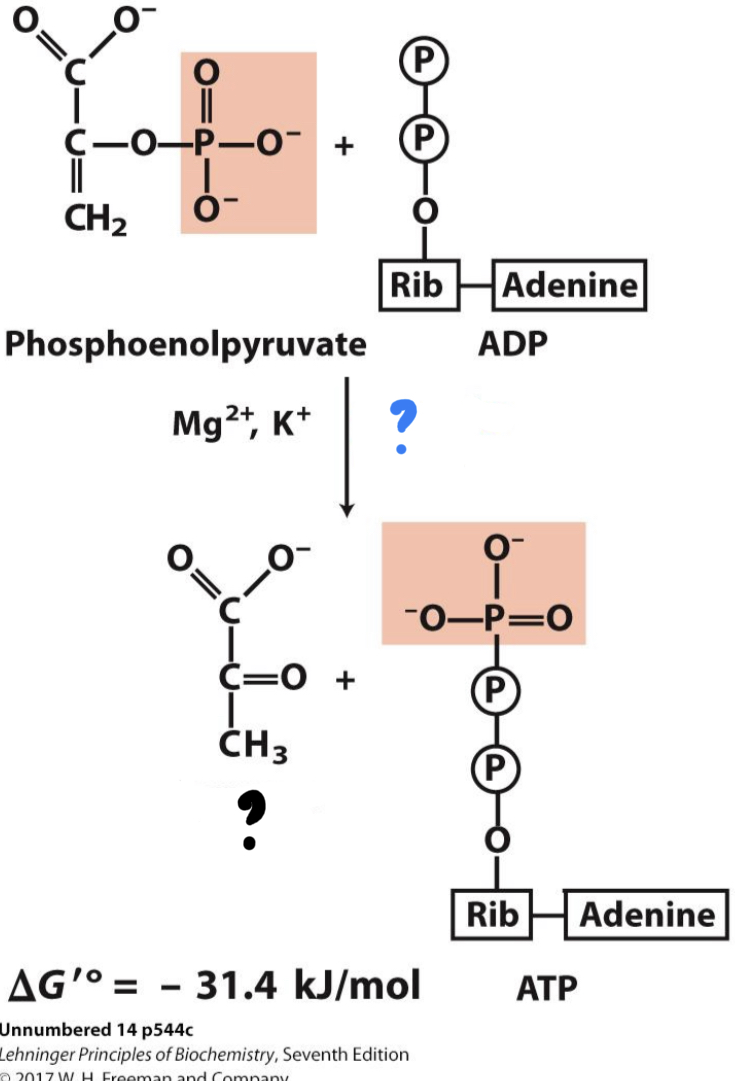
32
New cards
? ADP(x2)
?? ATP(x2)
?? ATP(x2)
33
New cards
Pyruvate Oxidation
* performed by _______ dehydrogenase complex
* Occurs in the **mitochondrial matrix** in eukaryotes
* Occurs at the **plasma membrane** in prokaryotes
* Where pyruvate is converted to acetyl Coenzyme A(acetyl CoA)
* Occurs in the **mitochondrial matrix** in eukaryotes
* Occurs at the **plasma membrane** in prokaryotes
* Where pyruvate is converted to acetyl Coenzyme A(acetyl CoA)
34
New cards
Pyruvate(3 carbons) to Acetyl(2 carbons) + CO2(waste)
* First step of pyruvate oxidation where the 1st enzyme catalyzes ***decarboxylation,*** so a carboxyl group is removed from pyruvate
? 1st Enzyme: pyruvate dehydrogenase
? 1st Enzyme: pyruvate dehydrogenase
35
New cards
Acetyl is __attached to Coenzyme A__ generating **Acetyl-CoA**
* Second step of pyruvate oxidation where a 2nd enzyme __grabs Acetyl to transfer it between reaction sites__
? 2nd Enzyme: Dihydrolipoyl transacetylase
? 2nd Enzyme: Dihydrolipoyl transacetylase
36
New cards
NAD+ is reduced to NADH + CO2
* Third step of pyruvate oxidation where *2 electrons from pyruvate oxidation are passed to coenzyme NADH* catalyzed by the 3rd enzyme
? Enzyme: Dihydrolipoyl dehydrogenase
? Enzyme: Dihydrolipoyl dehydrogenase
37
New cards
Krebs Cycle/Citric Acid Cycle
* Energy remains in bonds of acetyl-CoA
* completes the breakdown of pyruvate to CO2
* oxidizes organic fuel derived from pyruvate, generating 1 ATP, 3 NADH, and 1 FADH2 per turn
* Occurs:
* In the **cytoplasm** of prokaryotes
* In the **mitochondrial matrix** of eukaryotes
* completes the breakdown of pyruvate to CO2
* oxidizes organic fuel derived from pyruvate, generating 1 ATP, 3 NADH, and 1 FADH2 per turn
* Occurs:
* In the **cytoplasm** of prokaryotes
* In the **mitochondrial matrix** of eukaryotes
38
New cards
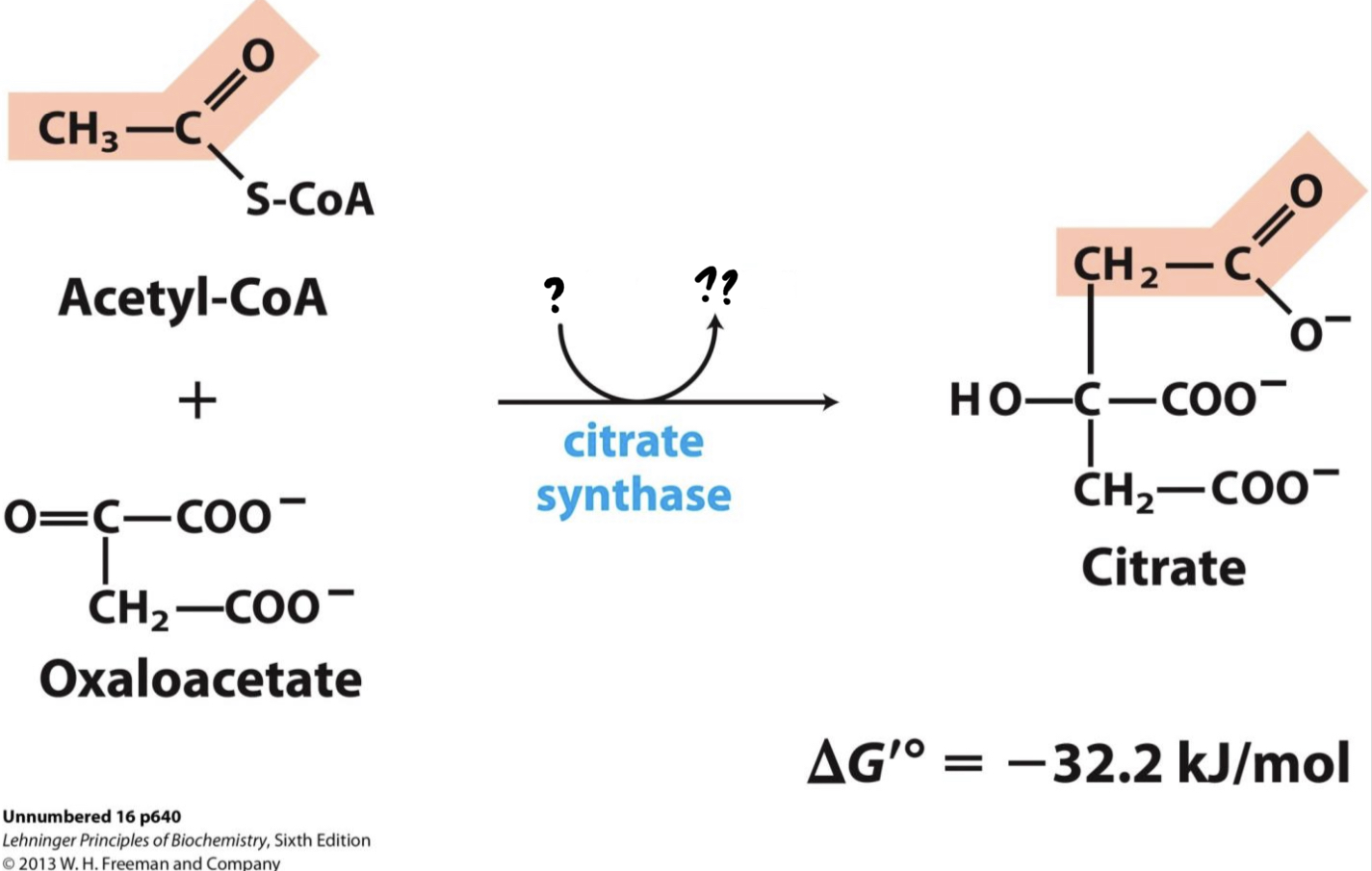
\*Step 1
^^? Citrate synthase^^
? Citrate(6 carbons)
^^? Citrate synthase^^
? Citrate(6 carbons)
* Acetyl CoA (from pyruvate oxidation) adds its __two-carbon acetyl group to four-carbon oxaloacetate__, producing _______.
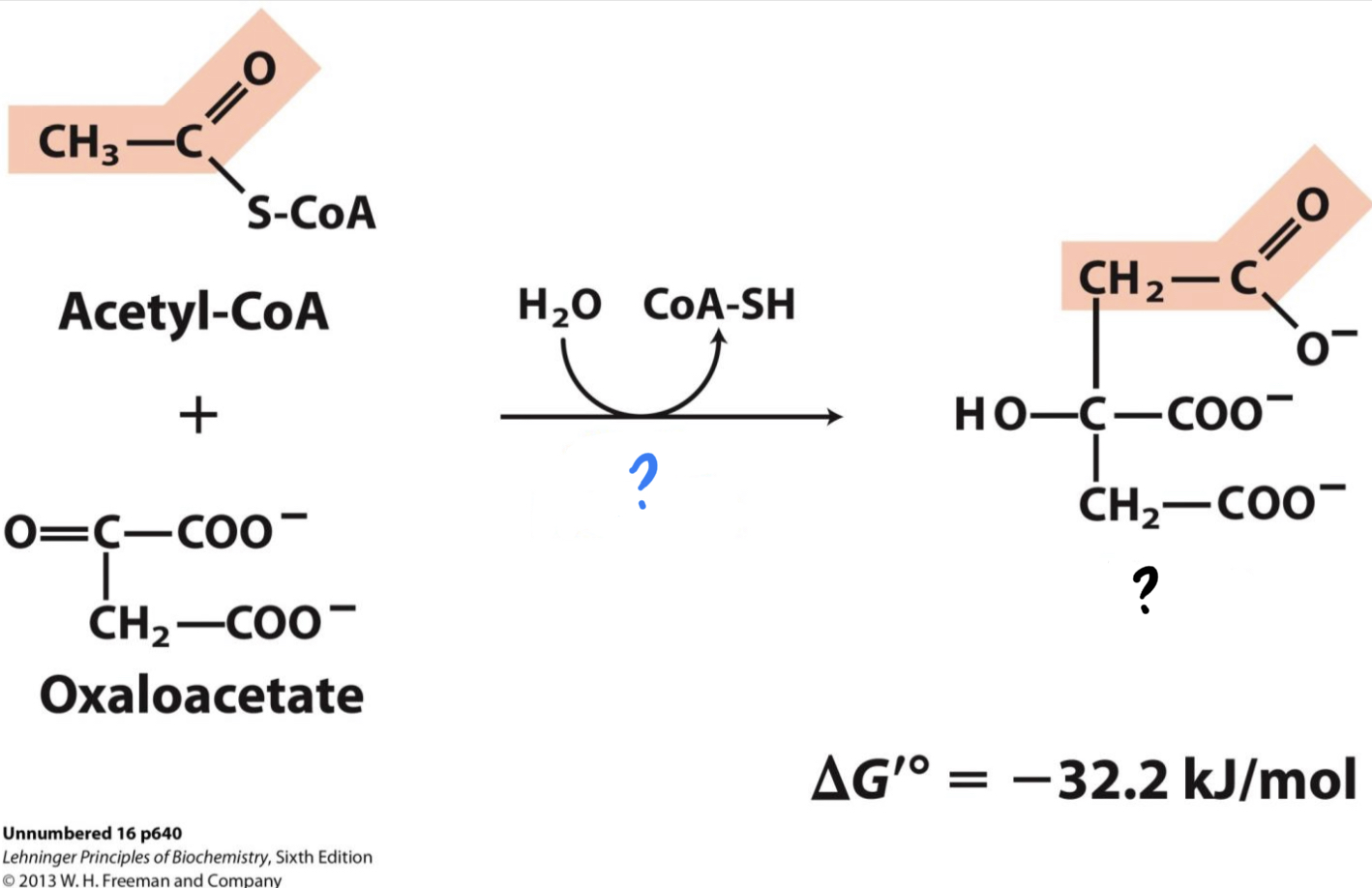
39
New cards
? H2O
?? CoA-SH
?? CoA-SH
\
40
New cards
\*Step 2
? ^^Aconitase^^
? Isocitrate(6 carbons)
? ^^Aconitase^^
? Isocitrate(6 carbons)
* Citrate is converted to its isomer, isocitrate, by removal of one water molecule and addition of another. This reaction does not happen immediately however, as it is converted to cis-^^_________^^ first.
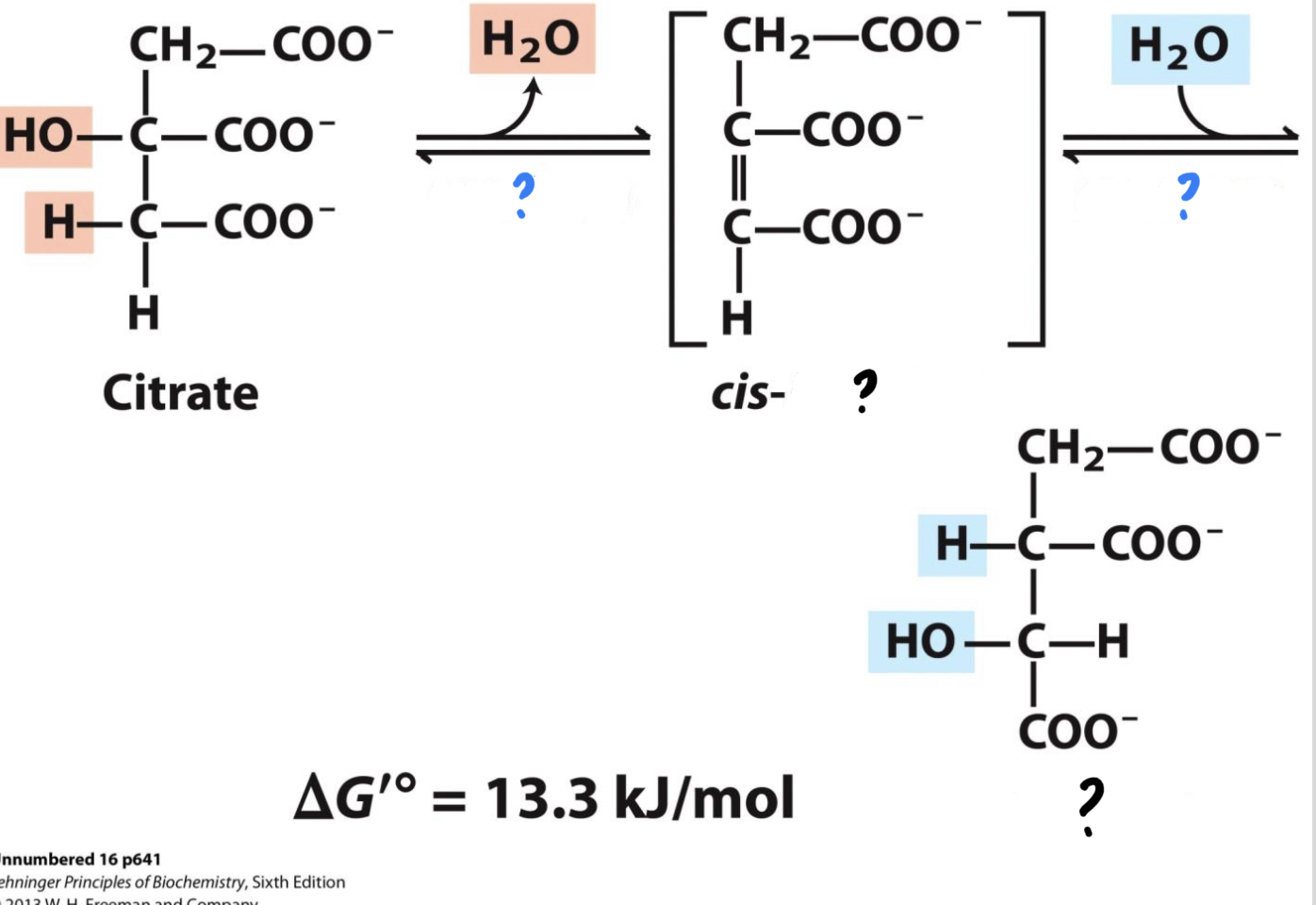
41
New cards
\*Step 3
^^? Isocitrate dehydrogenase^^
? a-Ketoglutarate(5 carbons)
^^? Isocitrate dehydrogenase^^
? a-Ketoglutarate(5 carbons)
* Isocitrate is oxidized, __reducing NAD+__
__to NADH. Then the resulting compound loses a CO2 molecule.__
__to NADH. Then the resulting compound loses a CO2 molecule.__
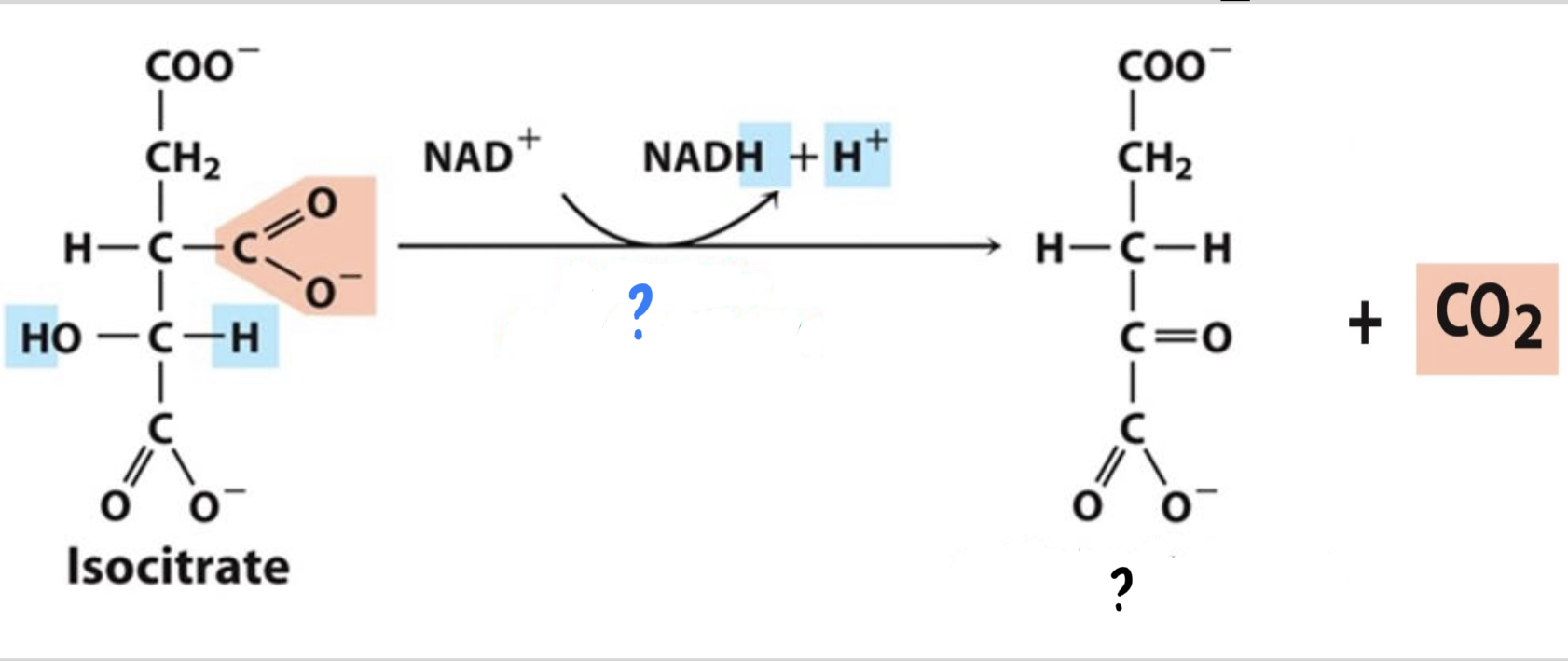
42
New cards
? NAD+
?? NADH & CO2
?? NADH & CO2
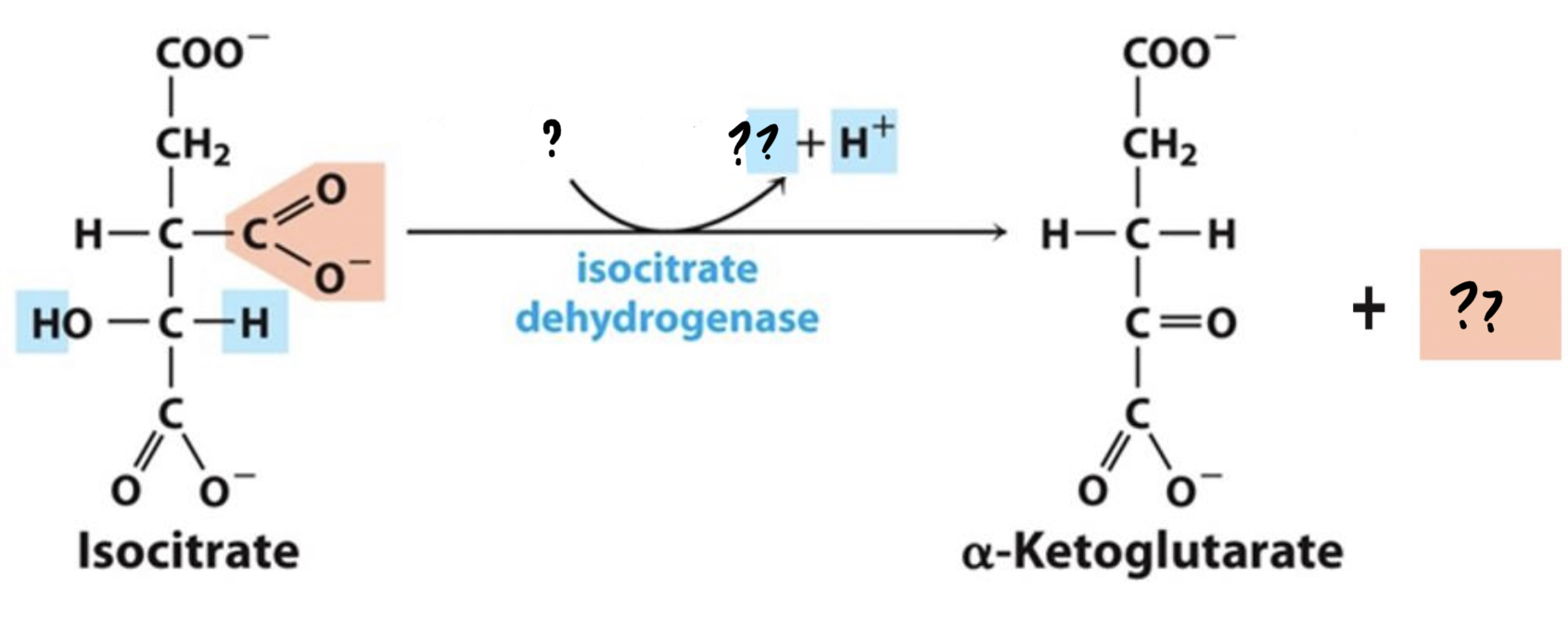
43
New cards
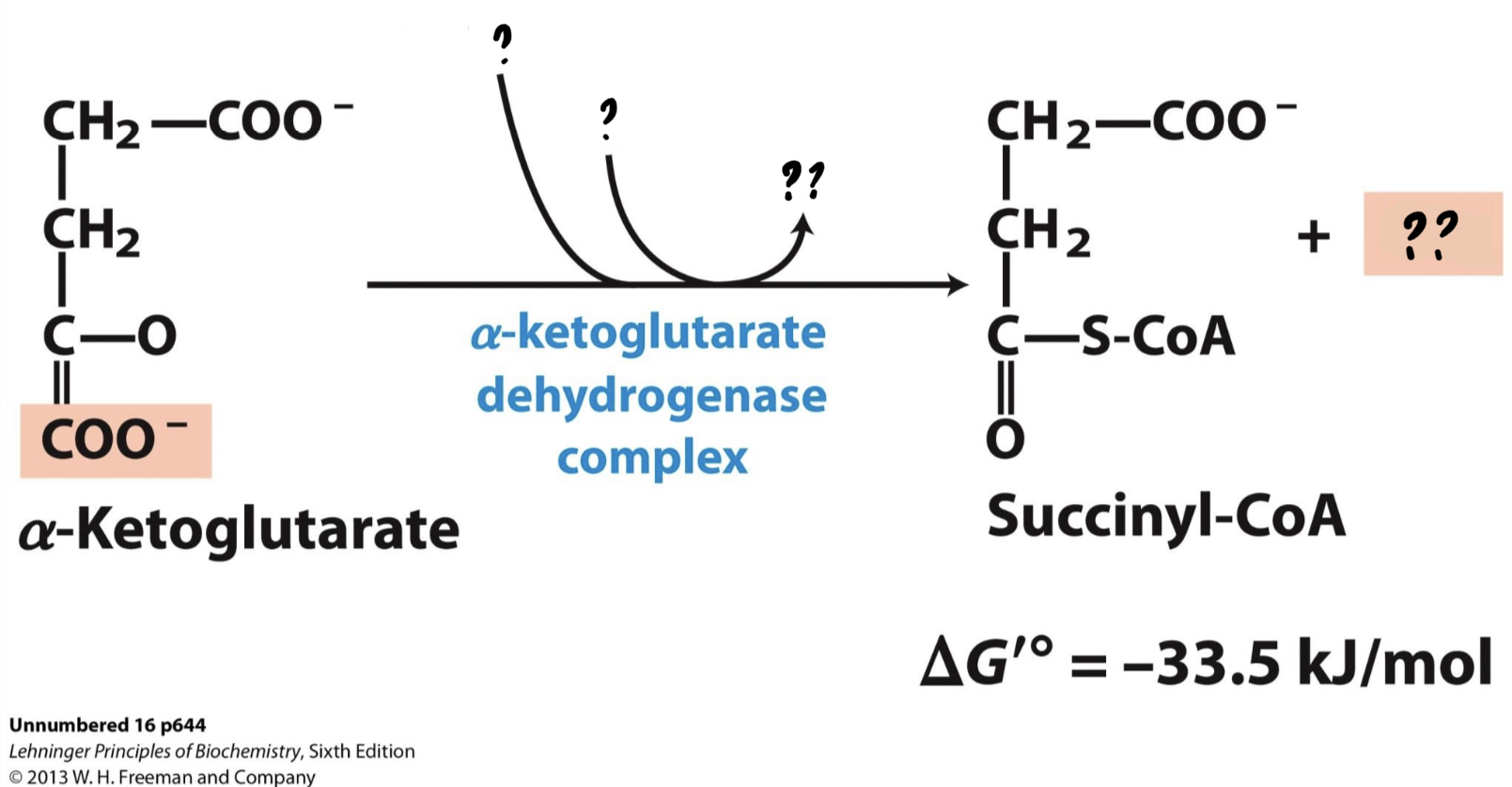
\*Step 4
^^? a-ketoglutarate dehydrogenase complex^^
? Succinyl-CoA(4 carbons)
^^? a-ketoglutarate dehydrogenase complex^^
? Succinyl-CoA(4 carbons)
* __Another CO2 is lost, and the resulting compound is oxidized, reducing NAD+__
__to NADH.__ The remaining molecule is then attached to coenzyme A by an unstable bond.
__to NADH.__ The remaining molecule is then attached to coenzyme A by an unstable bond.
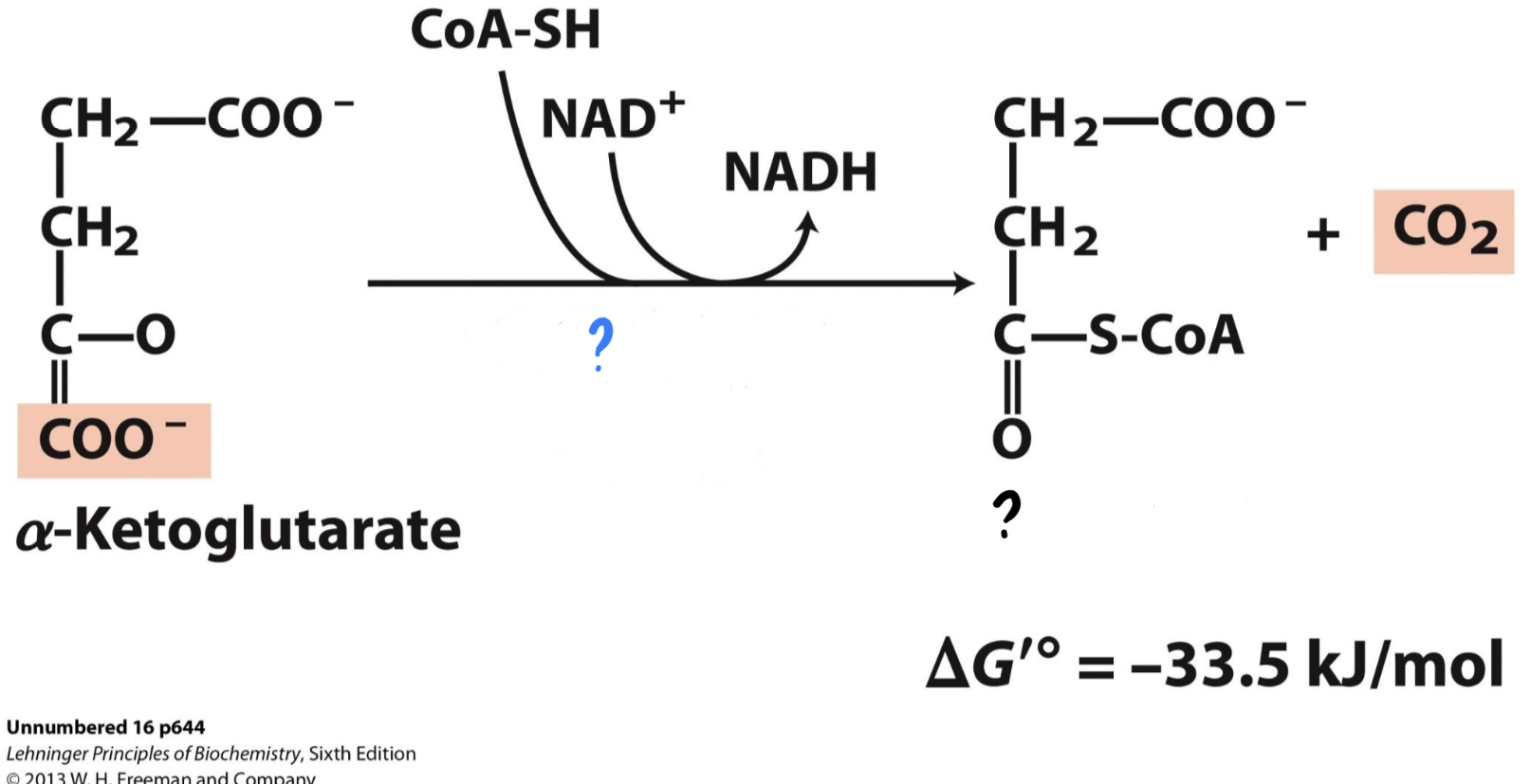
44
New cards
? CoA-SH & NAD+
?? NADH & CO2
?? NADH & CO2
45
New cards
\*Step 5
^^? succinyl-CoA synthetase^^
? Succinate(4 carbons)
^^? succinyl-CoA synthetase^^
? Succinate(4 carbons)
* CoA is displaced by a phosphate group, __which is transferred to GDP, forming GTP, a molecule with functions similar to ATP.__ GTP can also be used, as shown, to generate ATP.
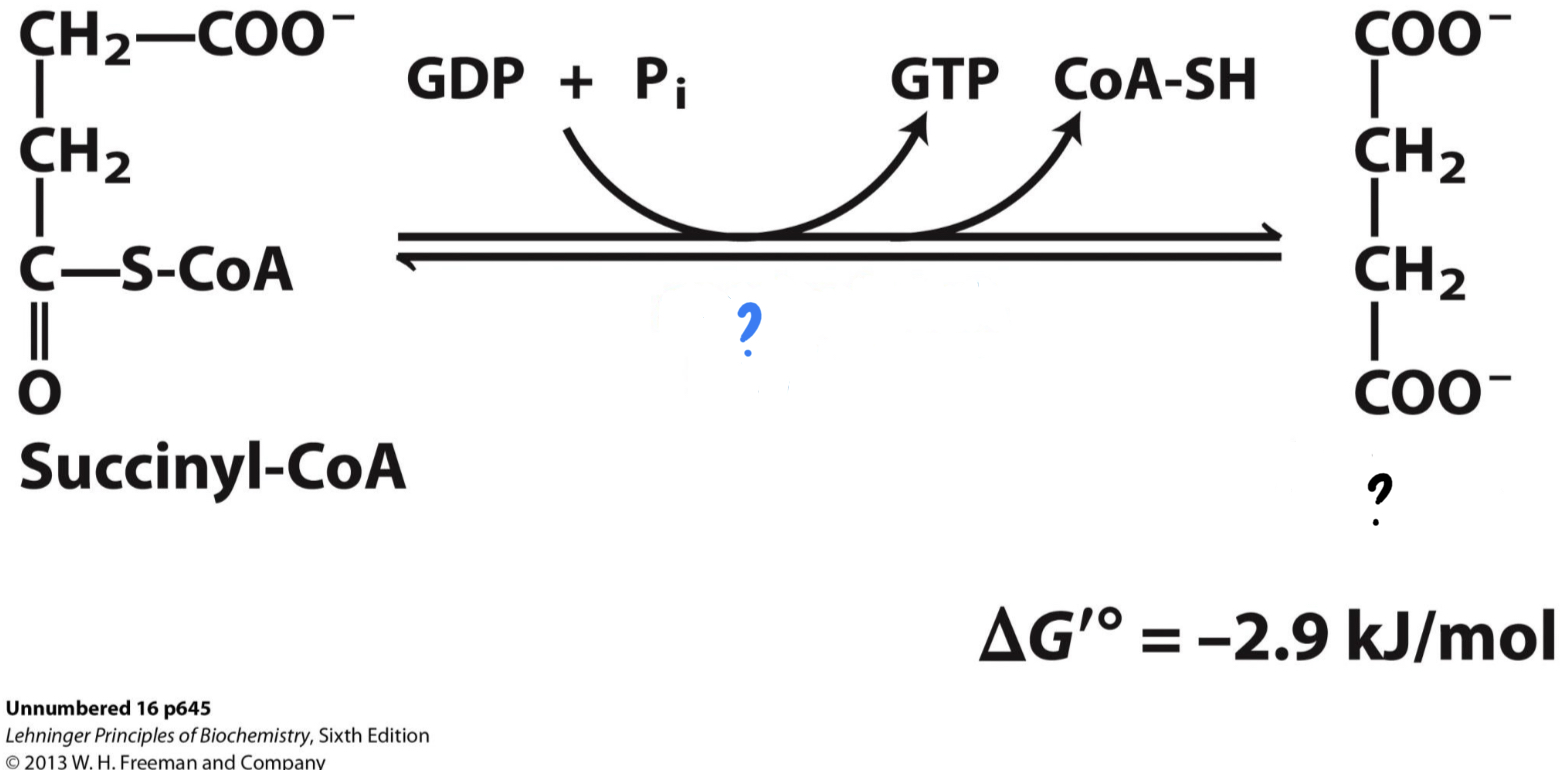
46
New cards
? GDP + P
?? GTP
??? CoA-SH
?? GTP
??? CoA-SH
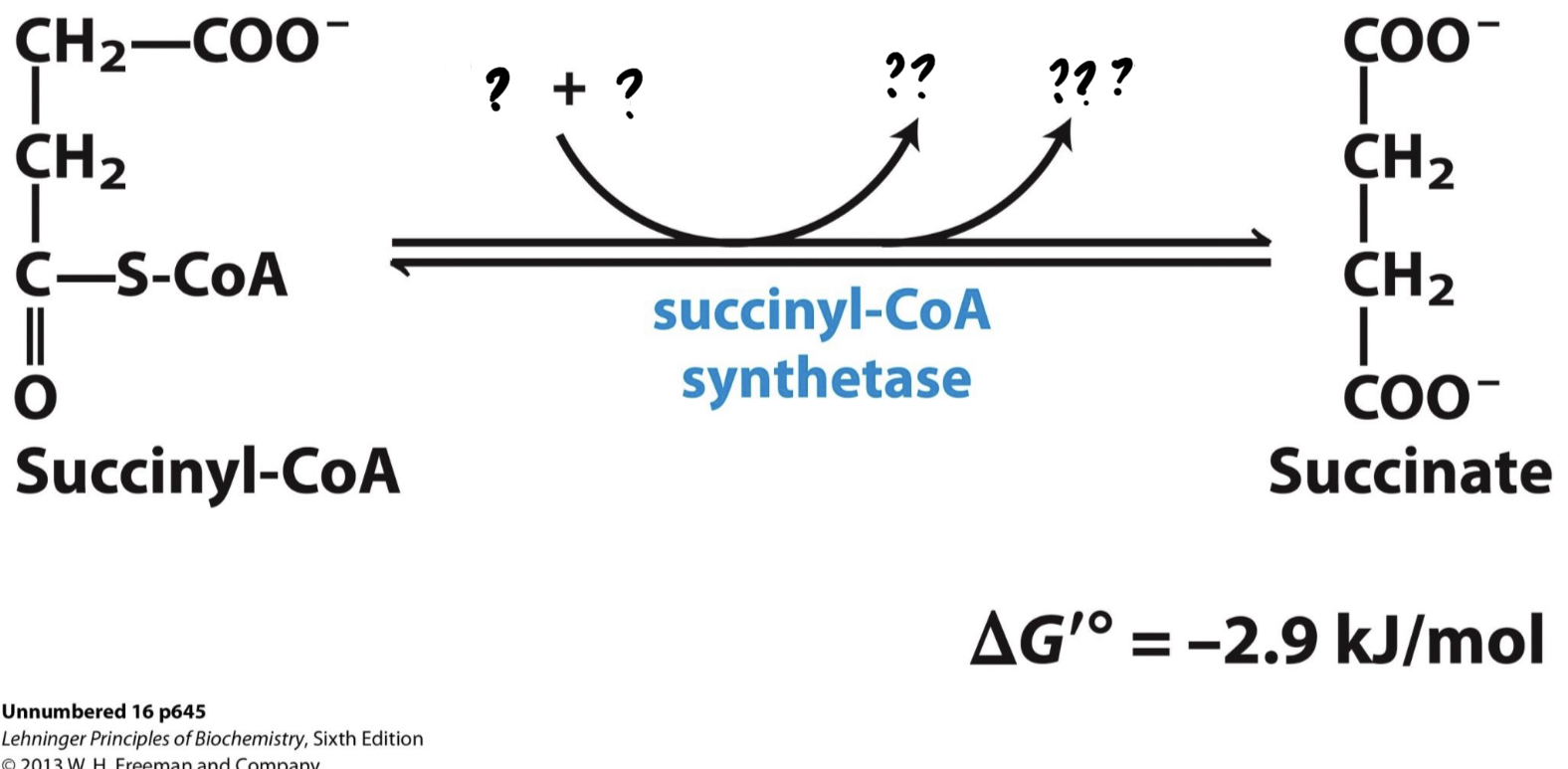
47
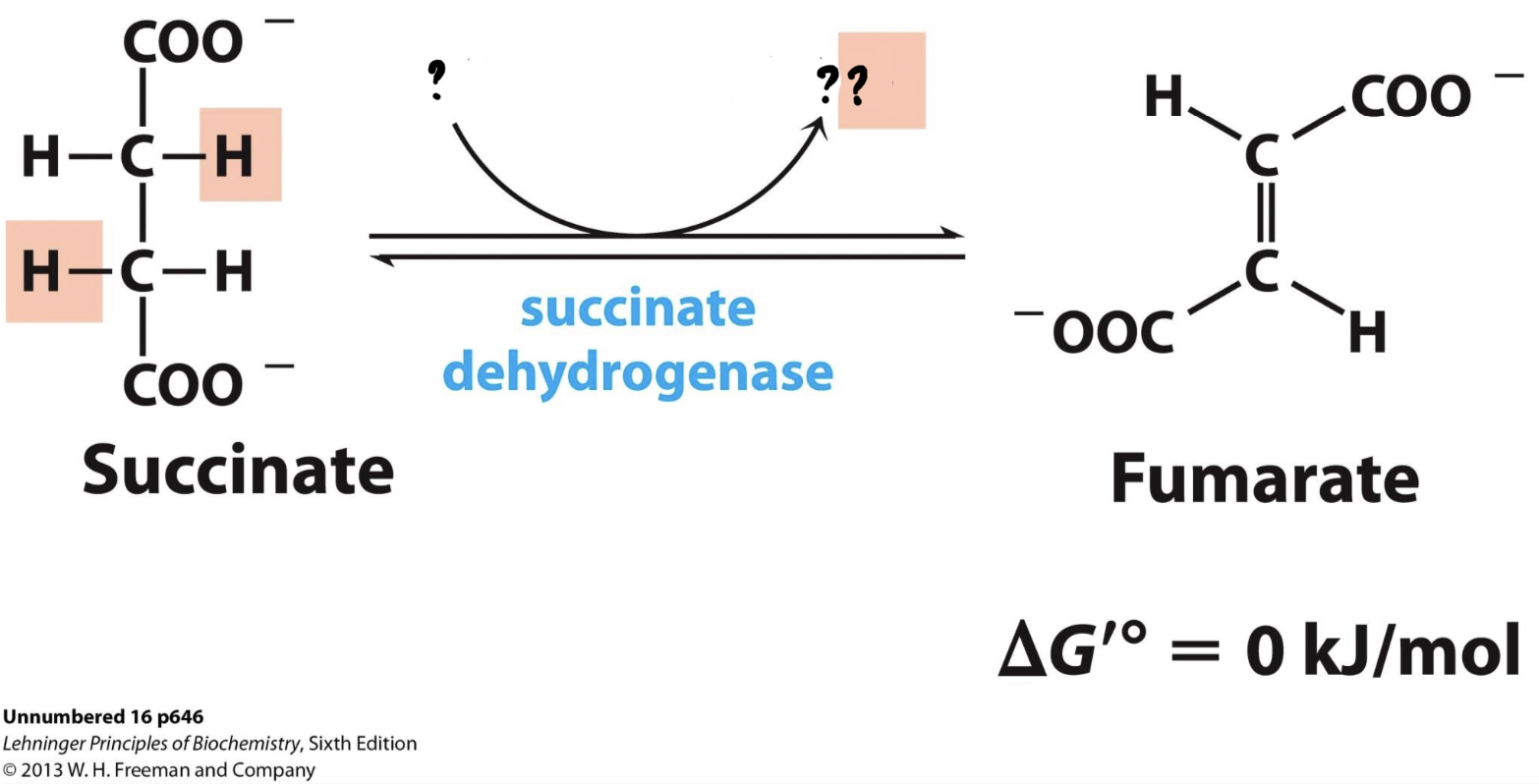
New cards
\*Step 6
^^? Succinate dehydrogenase^^
? Fumarate(4 carbon)
^^? Succinate dehydrogenase^^
? Fumarate(4 carbon)
* Two hydrogens are transferred to FAD, forming FADH2
* The enzyme catalyzes the oxidizing of succinate, __releasing 2 electrons that go to Coenzyme Q__(carries the electrons to nearby enzymes of ETC).
**Uses the enzyme embedded in the inner mitochondrial membrane and known as Complex II of ETC* \*
* The enzyme catalyzes the oxidizing of succinate, __releasing 2 electrons that go to Coenzyme Q__(carries the electrons to nearby enzymes of ETC).
**Uses the enzyme embedded in the inner mitochondrial membrane and known as Complex II of ETC* \*
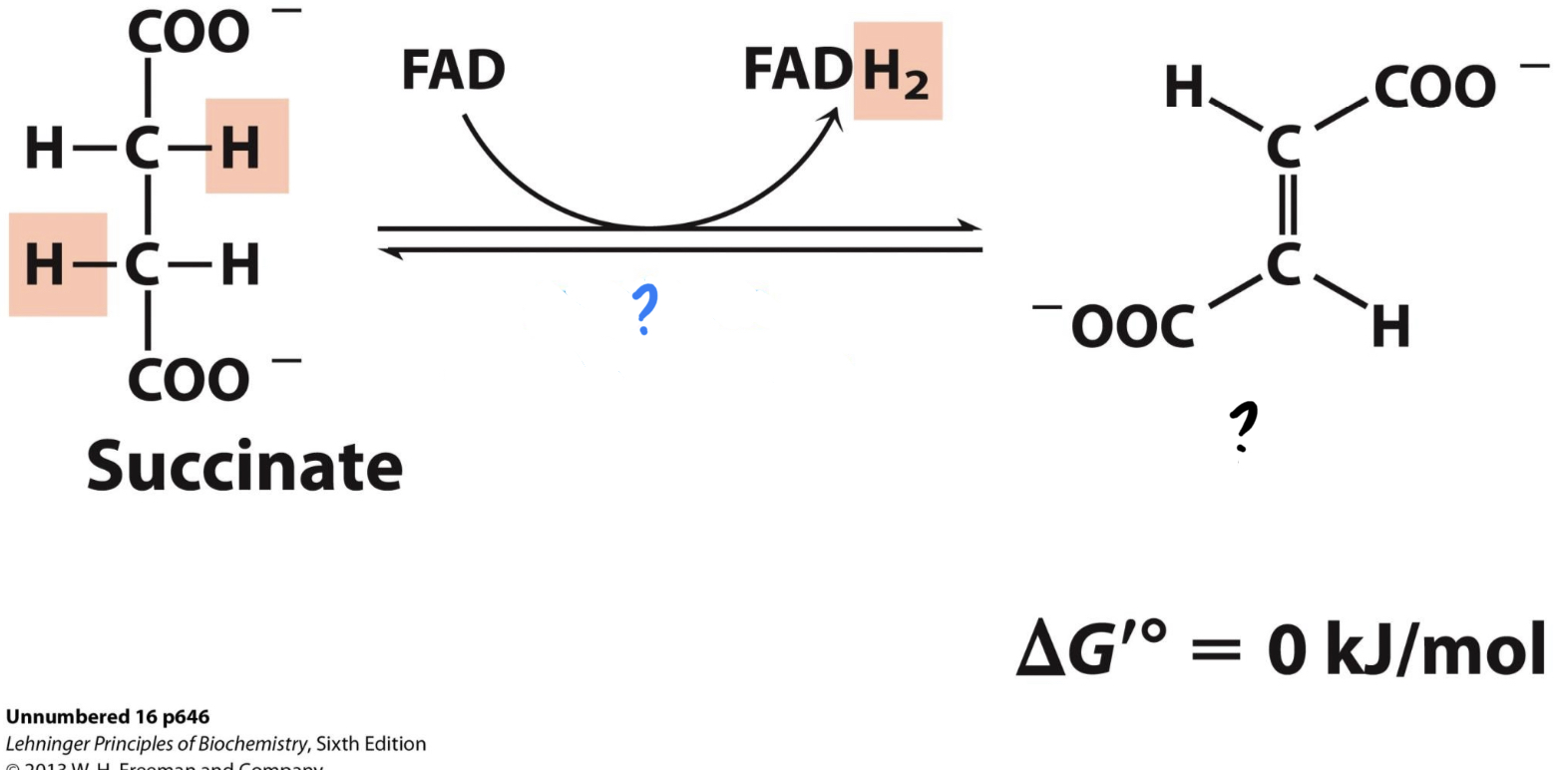
48
New cards
? FAD
?? FADH2
?? FADH2
49
New cards
\*Step 7
^^? Fumarase^^
? Malate(4 carbons)
?? H2O
^^? Fumarase^^
? Malate(4 carbons)
?? H2O
* Addition of a water molecule rearranges bonds in the substrate.
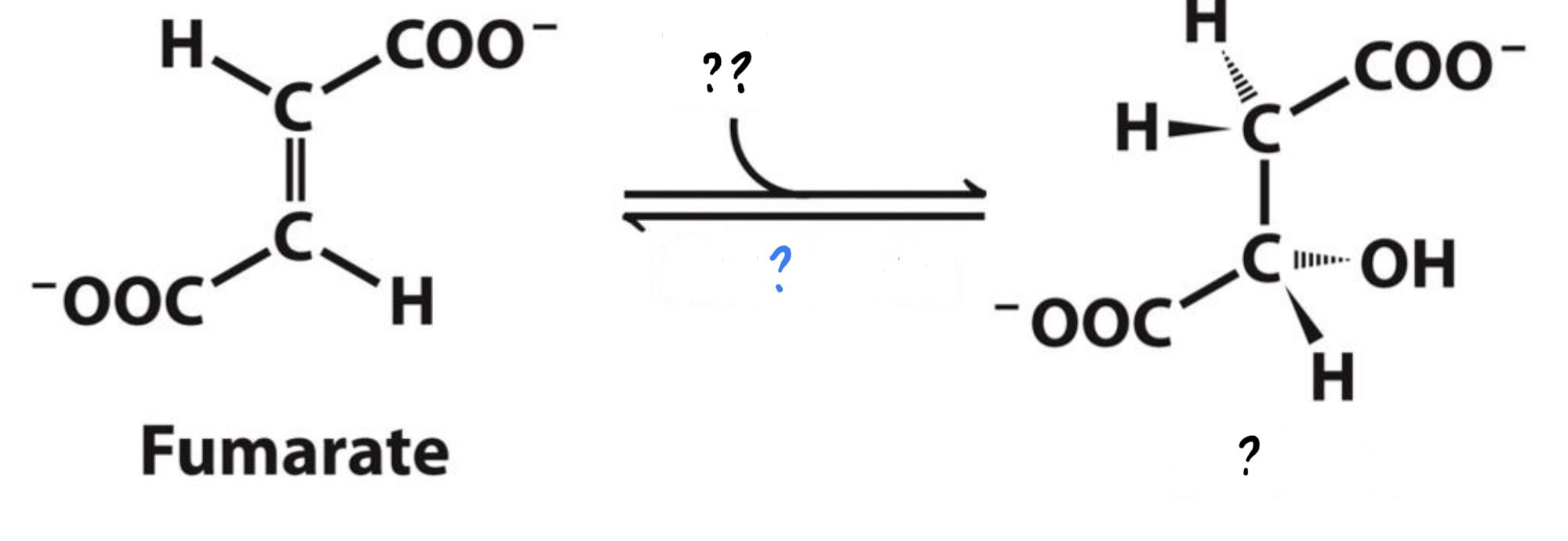
50
New cards
\*Step 8
^^? Malate dehydrogenase^^
? Oxaloacetate(4 carbons)
^^? Malate dehydrogenase^^
? Oxaloacetate(4 carbons)
* The substrate is oxidized, reducing NAD+ to NADH and regenerating ____________.
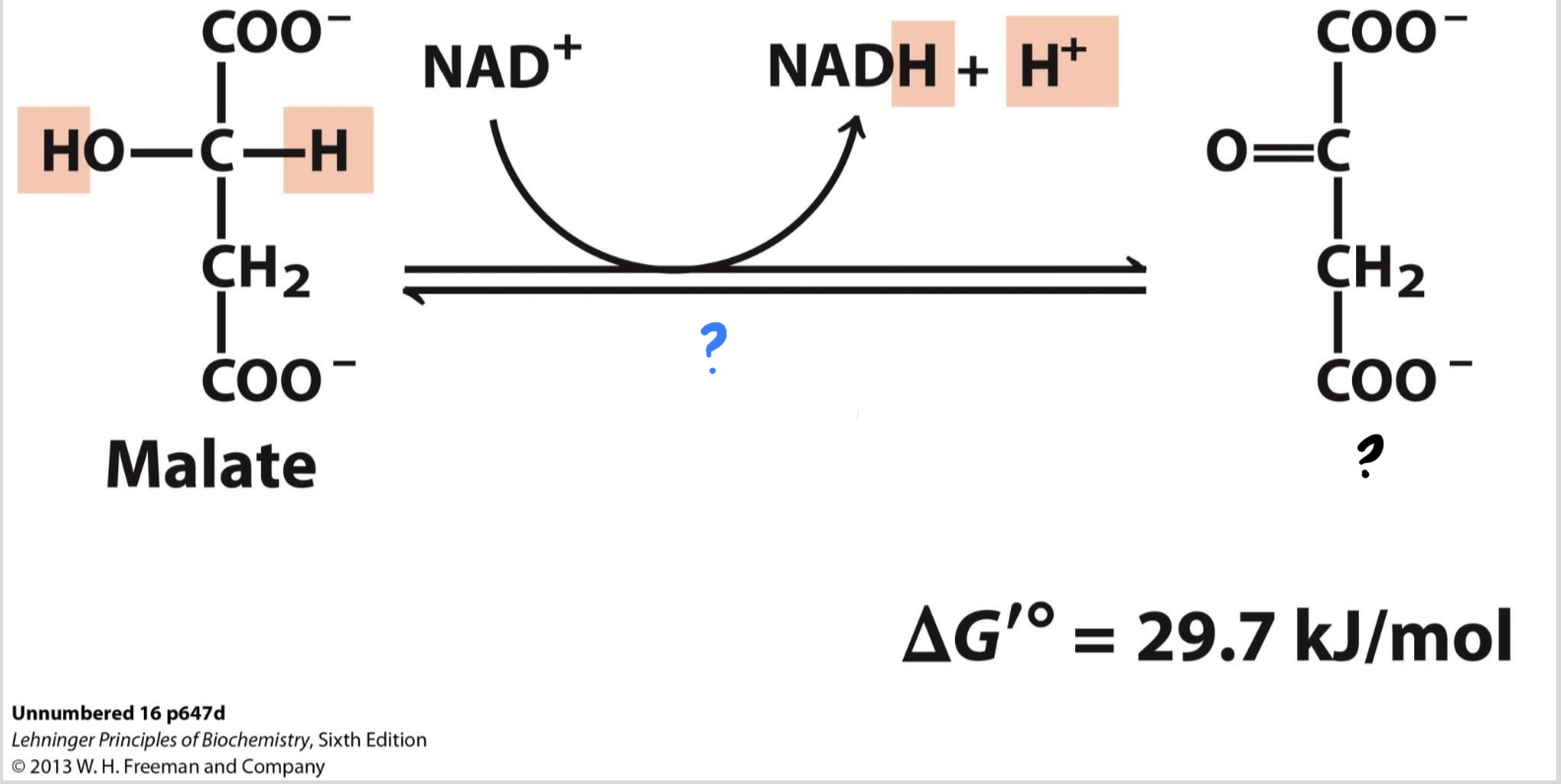
51
New cards
? NAD+
?? NADH + H+
?? NADH + H+
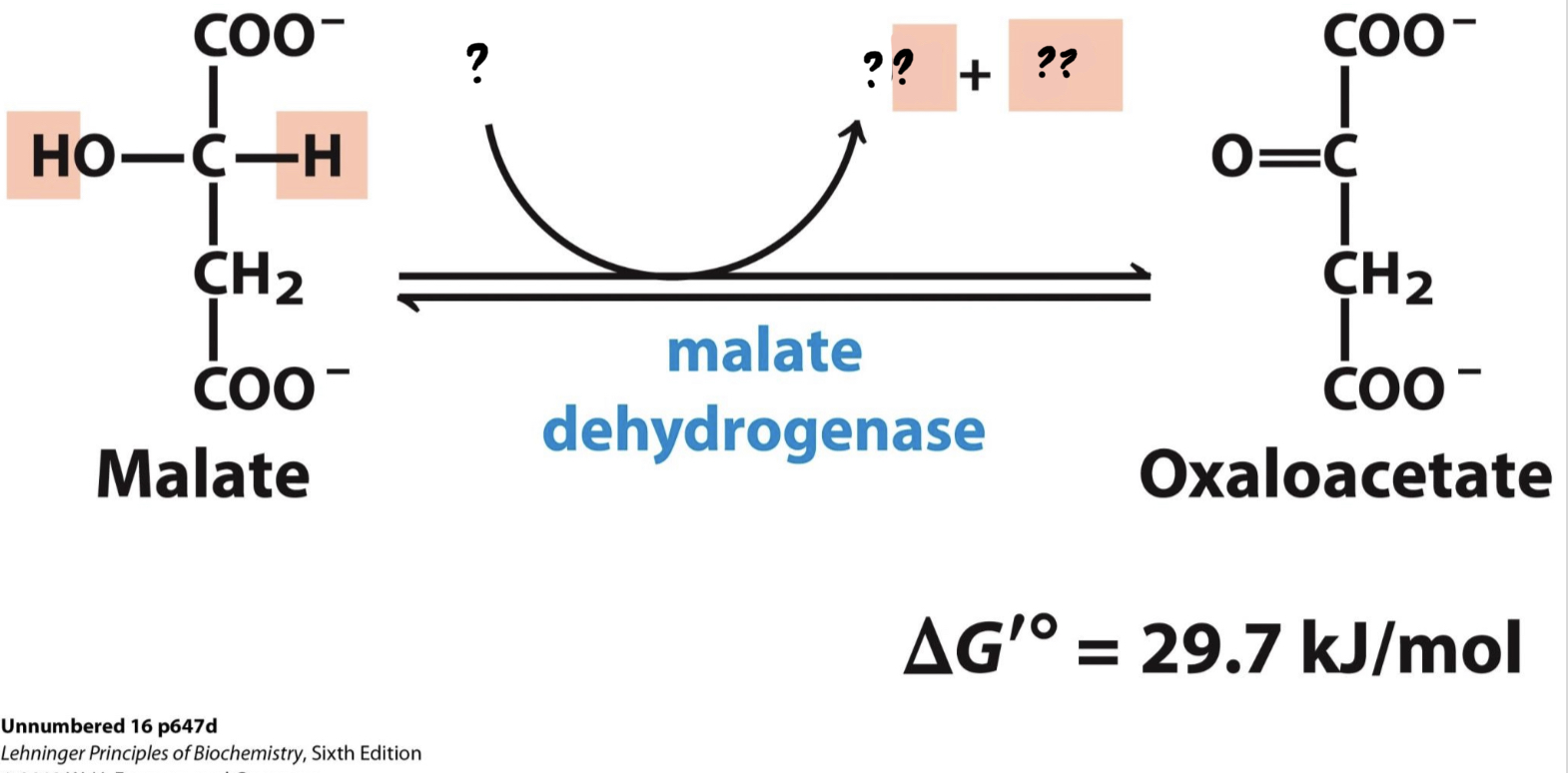
52
New cards
Oxidative phosphorylation
* A mechanism for the synthesis of ATP
* ATP synthase uses energy from a proton gradient
* During ETC and Chemiosmosis
* produces most of the ATP generated from Cellular Respiration
* ATP synthase uses energy from a proton gradient
* During ETC and Chemiosmosis
* produces most of the ATP generated from Cellular Respiration
53
New cards
Electron Transport Chain(ETC)
* is a series of membrane-bound electron carriers
* Embedded in the inner mitochondrial membrane (**cristae**)
* Electrons from NADH and FADH2 are transferred to the four complexes of the ETC
* Each complex
* Has a proton pump creating proton gradient
* Transfers electrons to the next carrier
* Electrons end up in Oxygen -> The final electron acceptor ◦
* Oxygen is reduced to water
* Embedded in the inner mitochondrial membrane (**cristae**)
* Electrons from NADH and FADH2 are transferred to the four complexes of the ETC
* Each complex
* Has a proton pump creating proton gradient
* Transfers electrons to the next carrier
* Electrons end up in Oxygen -> The final electron acceptor ◦
* Oxygen is reduced to water
54
New cards
ETC
* Electron carriers for Glycolysis, Pyruvate Oxidation, and Krebs cycle drop off e- at ETC membrane complexes
* H+ is pumped out, creates a gradient (more H+ outside of inner membrane)
* H+ is pumped out, creates a gradient (more H+ outside of inner membrane)
55
New cards
* NADH dehydrogenase
* Succinate dehydrogenase
* Ubiquinone(Q)-Cytochrome C Reductase
* Cytochrome C Oxidase
* Succinate dehydrogenase
* Ubiquinone(Q)-Cytochrome C Reductase
* Cytochrome C Oxidase
* Complex I
* Complex II (one of the enzymes in the Krebs cycle). This complex does not have proton pumping ability.
* Complex III
* Complex IV
* Complex II (one of the enzymes in the Krebs cycle). This complex does not have proton pumping ability.
* Complex III
* Complex IV
56
New cards
NADH dehydrogenase
* The NADH donated from glycolysis, and the citric acid cycle is oxidized here, transferring 2 electrons from NADH to coenzyme Q
* 1st reaction: NADH is oxidized, __**releasing 2 electrons that go to coenzyme Q(**__which is also reduced) and carry electrons to the next part of ETC
* 2nd reaction: the movement of charged electrons makes Complex I bend in shape, transmit energy and __**pump out 4 protons across the membrane**__
* 1st reaction: NADH is oxidized, __**releasing 2 electrons that go to coenzyme Q(**__which is also reduced) and carry electrons to the next part of ETC
* 2nd reaction: the movement of charged electrons makes Complex I bend in shape, transmit energy and __**pump out 4 protons across the membrane**__
57
New cards
Succinate dehydrogenase
* accepts electrons from succinate (an intermediate in the citric acid cycle) and acts as a second entry point to the ETC.
* When succinate oxidizes to fumarate, 2 electrons are accepted by FAD within complex II. __FADH passes them to coenzyme Q,__ similar to complex I.
* When succinate oxidizes to fumarate, 2 electrons are accepted by FAD within complex II. __FADH passes them to coenzyme Q,__ similar to complex I.
58
New cards
Coenzyme Q(ubiquinone)
* Its purpose is to function as an electron carrier and transfer electrons to complex III.
59
New cards
* Ubiquinone(Q)-Cytochrome C Reductase
* separates electrons from coenzyme Q, passing 1 electron to cytochrome C which is reduced
* __**transports 4 protons across the membrane**__
\
* __**transports 4 protons across the membrane**__
\
60
New cards
reduced Cytochrome C
* carries the electron to the last step of ETC
61
New cards
Cytochrom C Oxidase
* oxidizes cytochrome c and transfers the electrons to **oxygen**, *the final electron carrier in aerobic cellular respiration.*
* a molecule of oxygen is captured, split and reduced, allowing it to accept electrons & pick up protons __**creating 2 molecules of water**__
* The free energy from the electron transfer __**causes 4 protons to move into the intermembrane space**__ contributing to the proton gradient.
* a molecule of oxygen is captured, split and reduced, allowing it to accept electrons & pick up protons __**creating 2 molecules of water**__
* The free energy from the electron transfer __**causes 4 protons to move into the intermembrane space**__ contributing to the proton gradient.
62
New cards
Chemiosmosis
* H+ flows back into the matrix through ATP Synthase, and ATP is created
* Accumulation of protons in the intermembrane space drives protons into the matrix via diffusion
* Since the membrane is relatively impermeable to ions, most protons can only reenter the matrix through ATP synthase
* Uses energy of proton gradient to make ATP from ADP + Pi
* For every 4 H+ in, one ATP is created.
* Accumulation of protons in the intermembrane space drives protons into the matrix via diffusion
* Since the membrane is relatively impermeable to ions, most protons can only reenter the matrix through ATP synthase
* Uses energy of proton gradient to make ATP from ADP + Pi
* For every 4 H+ in, one ATP is created.
63
New cards
64
New cards
* 1 NADH = 10 H+ = 2.5 ATP
* 1 FADH2 = 6 H+ = 1.5 ATP
* 1 FADH2 = 6 H+ = 1.5 ATP
* What is the conversion of NADH and FADH2 to ATP?
65
New cards
Anaerobic respiration
* Couples with glycolysis when there is no O2 for cellular respiration so that the electron transport chain will not cease to operate
* uses an electron transport chain with a final __electron acceptor other than O2__, for example, **sulfate**
* uses an electron transport chain with a final __electron acceptor other than O2__, for example, **sulfate**
66
New cards
Fermentation
* Couples with glycolysis when there is no O2 for cellular respiration so that the electron transport chain will not cease to operate
* uses substrate-level phosphorylation instead of an electron
transport chain to generate ATP
* produces 2 ATP per glucose molecule
* uses substrate-level phosphorylation instead of an electron
transport chain to generate ATP
* produces 2 ATP per glucose molecule
67
New cards
Alcohol fermentation
Type of fermentation:
* pyruvate is converted to __**ethanol in two steps, with the first releasing CO2**__
* _______ fermentation by yeast is used in brewing, winemaking, and baking
* pyruvate is converted to __**ethanol in two steps, with the first releasing CO2**__
* _______ fermentation by yeast is used in brewing, winemaking, and baking
68
New cards
Lactic Acid Fermentation
Type of fermentation:
* pyruvate is reduced to NADH, __**forming lactate as an end product**__, with no release of CO2
* _______ ____ fermentation by some fungi and bacteria is used to make cheese and yogurt
* Human muscle cells use _______ ____ fermentation to generate ATP when O2
is scarce
* pyruvate is reduced to NADH, __**forming lactate as an end product**__, with no release of CO2
* _______ ____ fermentation by some fungi and bacteria is used to make cheese and yogurt
* Human muscle cells use _______ ____ fermentation to generate ATP when O2
is scarce
69
New cards
Obligate anaerobes
* Carry out fermentation or anaerobic respiration and cannot survive in the presence of O2
70
New cards
Facultative anaerobes
* pyruvate is a fork in the metabolic road that leads to two alternative catabolic routes
* able to survive using either fermentation or cellular respiration
Ex. yeast & bacteria
* able to survive using either fermentation or cellular respiration
Ex. yeast & bacteria
71
New cards
Methanogens
Modes of Anaerobic Respiration
* use carbon dioxide (CO2) as the electron acceptor, reducing CO2 to CH4 (methane).
* The hydrogens are derived from organic molecules produced by other organisms.
* Are found in diverse environments, including soil and the digestive systems of ruminants like cows.
* use carbon dioxide (CO2) as the electron acceptor, reducing CO2 to CH4 (methane).
* The hydrogens are derived from organic molecules produced by other organisms.
* Are found in diverse environments, including soil and the digestive systems of ruminants like cows.
72
New cards
Sulfur Bacteria
Modes of Anaerobic Respiration
* In this sulfate respiration, the prokaryotes derive energy from reducing inorganic sulfates (SO4 ) to hydrogen sulfide (H2S).
* The hydrogen atoms are obtained from organic molecules other organisms produce.
* These prokaryotes, thus, are similar to methanogens, but they use SO4 as the oxidizing (that is, electron-accepting) agent in place of CO2.
* In this sulfate respiration, the prokaryotes derive energy from reducing inorganic sulfates (SO4 ) to hydrogen sulfide (H2S).
* The hydrogen atoms are obtained from organic molecules other organisms produce.
* These prokaryotes, thus, are similar to methanogens, but they use SO4 as the oxidizing (that is, electron-accepting) agent in place of CO2.