A2 Units 3.1 & 3.2 Redox Reactions & Electrochemistry
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
Redox

Oxidation numbers

Deciding whether reduction or oxidation occurred

Ion-Electron Half Equations
Rules for working out:
Elements - check they are balanced
Oxygens - balance by adding H20
Hydrogens - balance by adding H+
Charge - balance by adding e-
(Every One Hates Charlie)
practice questions on pg. 6

Using half-reactions to obtain the equation for a redox reaction - Example
(practice questions on pg. 8)
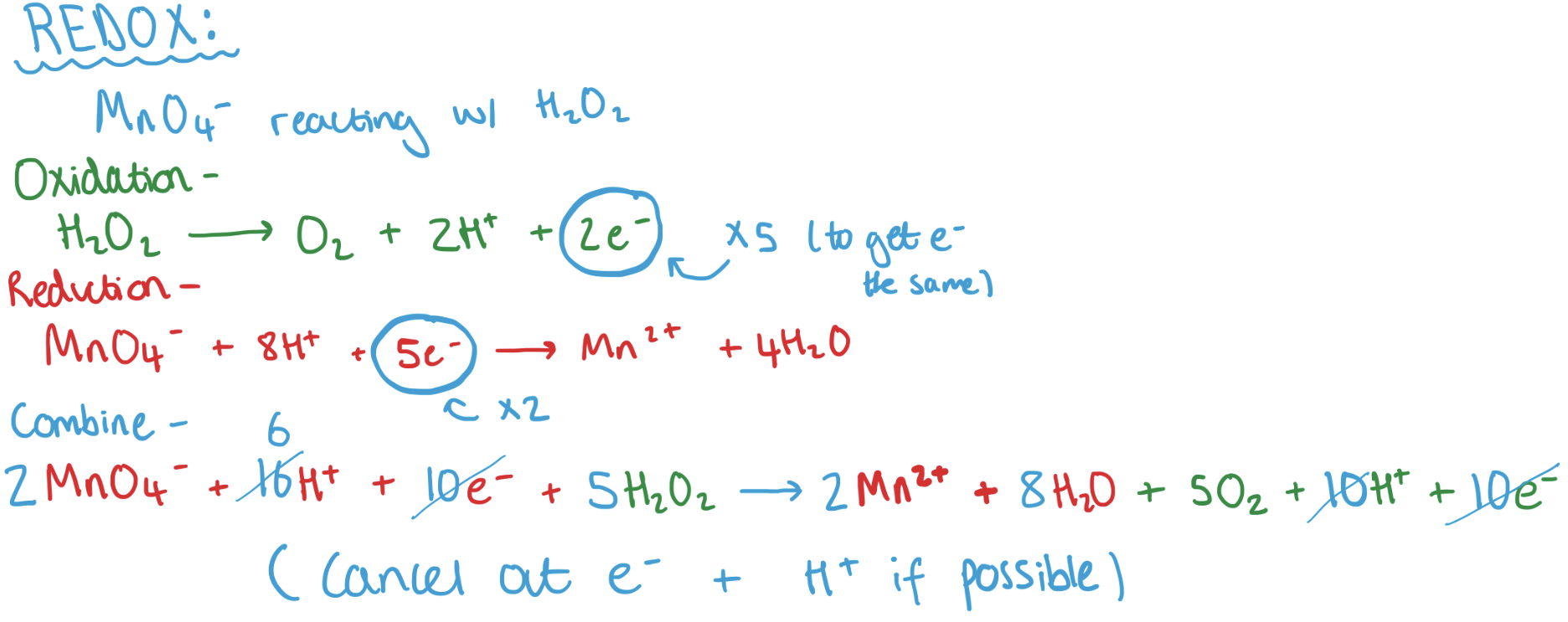
n = v x c questioned based on redox
(practice questions on pg. 17)

Redox reaction between Cu2+ and I- and determination of the liberated iodine with S2O3(2-)

Example

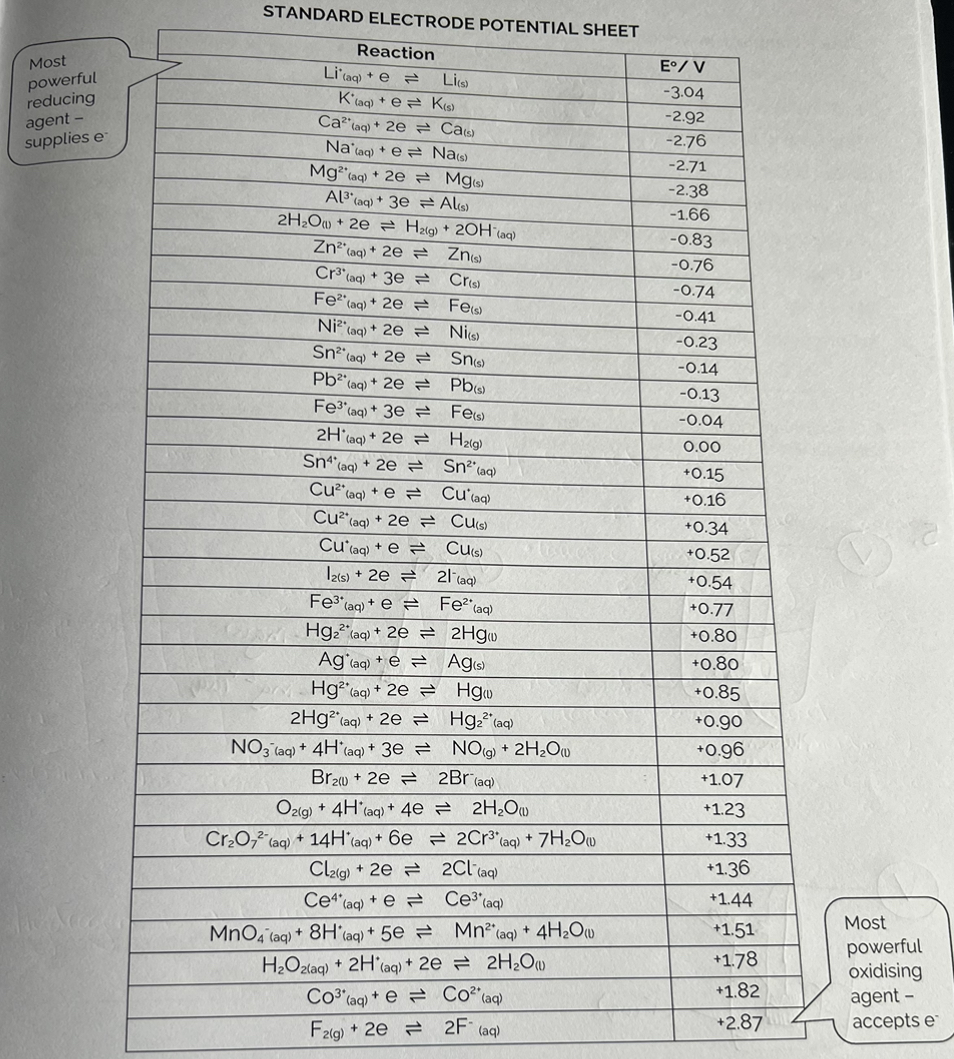
Standard Electrode Potentials
The electrode potential values range from +2.88 volts (F2/F-) to -3.04 volts (Li+/Li). Therefore, E* has a sign.
Strong oxidising systems have a large positive values of E°. (accepts / attracts e-)
Strong reducing systems have large negative values of E°. (donates / repels e-)
You do not need to remember E° values. See sheet which contains most systems used in the exam.
The convention is to write the symbol equation as reductions and use the convention that:

Measuring Standard Electrode Potentials
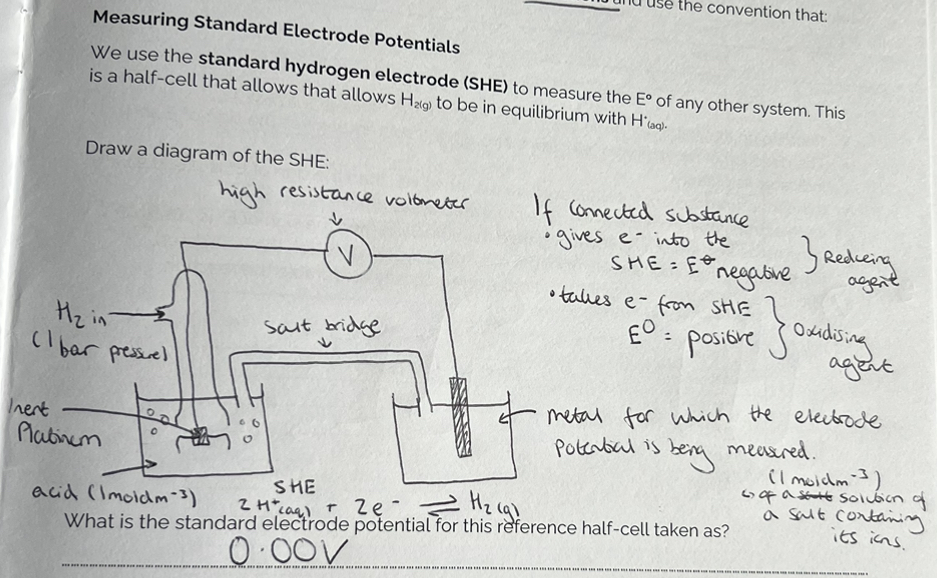
Why is Platinum Foil used?

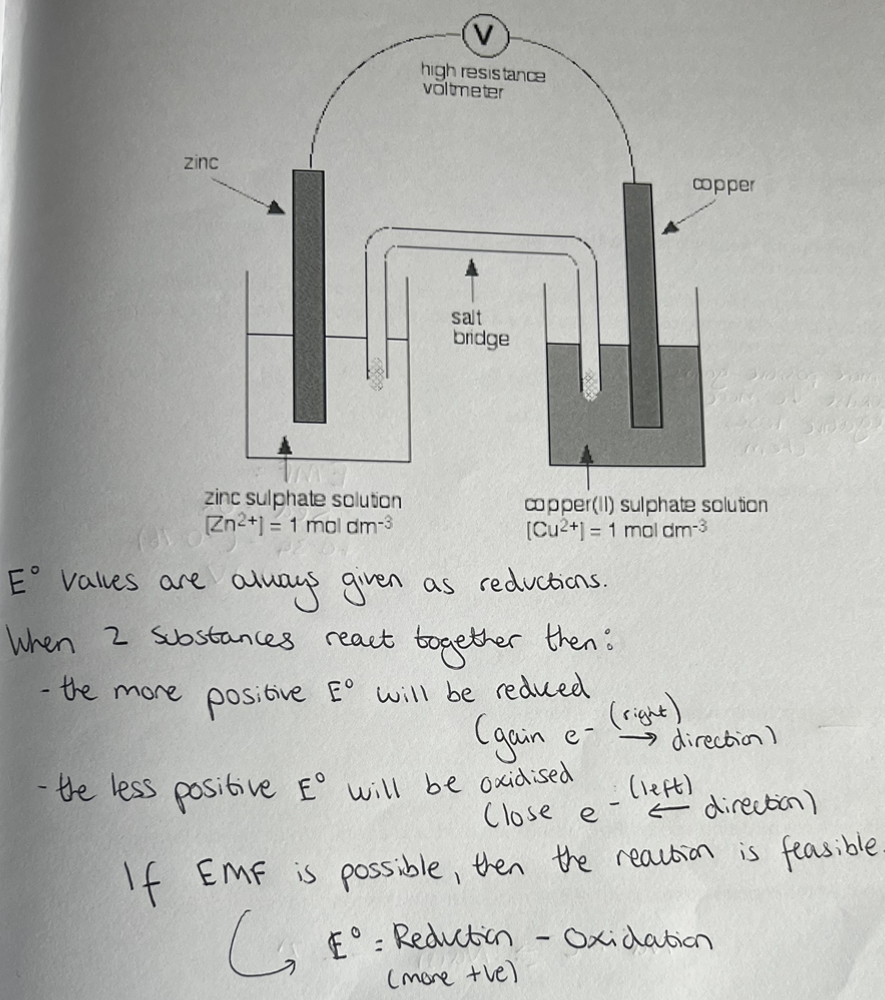
E* Measurements & The Salt Bridge
Reading on the voltmeter gives the VALUE of E° for the measured system.
The direction of movement of electrons through the voltmeter gives the sign of E and indicates its polarity
If the measured system is MORE positive than the SHE, then the sign of E° is Positive (i.e. electrons flow from the SHE to the measured system)
If the measured system is MORE negative than the SHE, then the sign of E° is negative (i.e. electrons flow into the SHE from the measured system through the voltmeter).
The more positive the value of E°, the more energetically favourable the reaction.

(Questions on pg. 25)
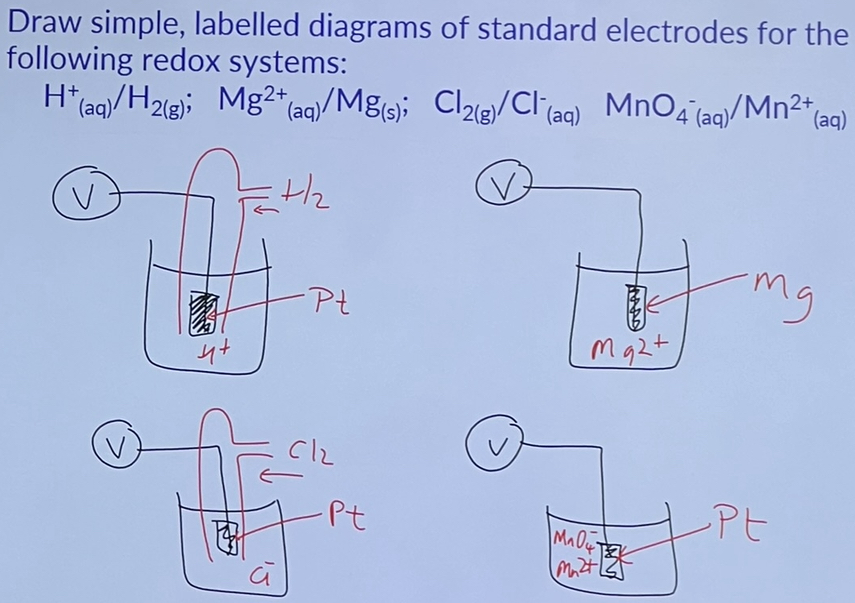
Putting half cells together + Standard cell potential
Calculate the emf (voltage) of the cell between Cr2072- /Cr and a Zn2+ / Zn electrode:
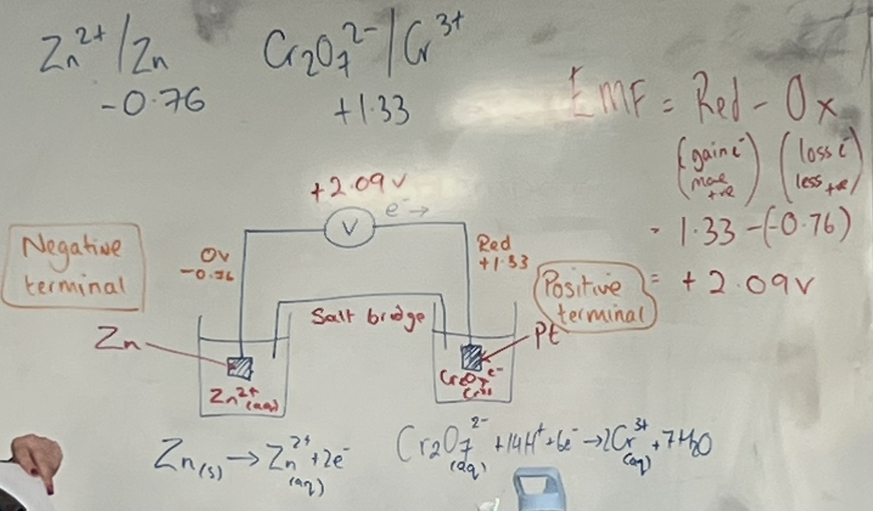
Electron Flow

Examples
Formula: EMF = Red - Ox
Red = EMF + Ox
Ox = EMF - Red

Daniel Cell

SHE
If E* is positive, the reaction is feasible.
Predicting the redox reactions and their feasibility
By studying standard electrode potentials, predictions can be made about the likely feasibility of a reaction. In general, a process is feasible if the overall cell potential is positive. i.e. the equilibrium will lie to the RIGHT

Standard Electrode Potential Sheet
Displacement & Questions


Electrochemical Series

(practice questions on pg. 34)

Hydrogen Fuel Cell
A fuel cell is an electrochemical cell. A fuel cell converts the chemicals hydrogen and oxygen into water, and in the process it produces electricity.
The other electrochemical device that we are all familiar with is the battery. A battery has all of its chemicals stored inside, and it converts those chemicals into electricity too. This means that a battery eventually "goes dead" and you either throw it away or recharge it.
With a fuel cell, chemicals constantly flow into the cell so it never goes dead -- as long as there is a flow of chemicals into the cell, the electricity flows out of the cell.

Electrode Equation

Benefits & Drawbacks of using Hydrogen as an energy source
