ALCOHOLS, ETHERS, THIOLS
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
glycols
2 hydroxyl groups w 2 adjacent carbons
alcohol polarity
polar, bc co and oh are polar covalent
how do alcohols associate in liquid state
hydrogen bond
acidity of alcohols
same pKa as water
phenols acidity
weak acids, reacts wit NaOH/strong bases to form water soluble salts
describe dehydration reactions
elimination of h2o from carbon = alkene, happens when alcohol is heated with h2so4 or h3po4
dehydration of primary alcohols
most difficult, requires high temp
dehydration of secondary alcohols
acid catalyzed dehydrations at lower temps
dehydration of tertiary alcohols
acid catalyzed dehydrations at above room temp
what happens when isomeric alkenes are obtained from dehydration
alkene w greater # alkyl groups predominates
oxidation of primary alcohols
becomes aldehyde/carboxylic acid (using potassium dichromate)
oxidation of secondary alcohol
becomes ketone.
tertiary alcohol reactions
resistant to oxidation.
using acid catalyst they are prone to dehydration
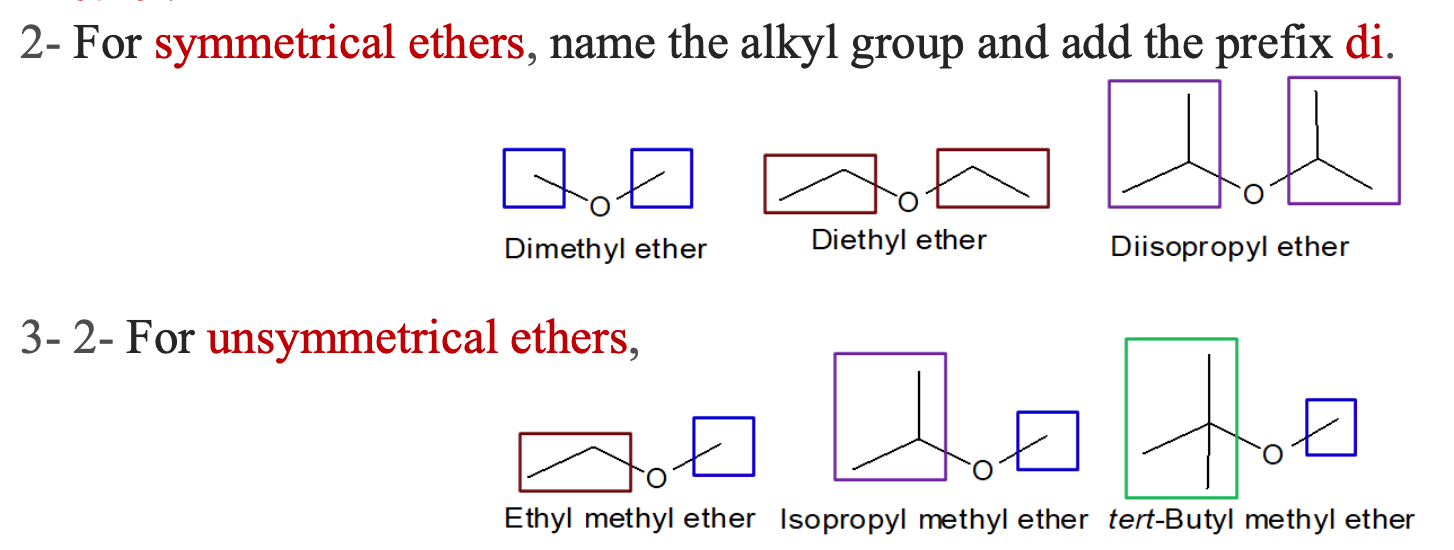
ether
oxygen atom bound to 2 carbon atoms
simplest either
dimethyl ether
most common ether
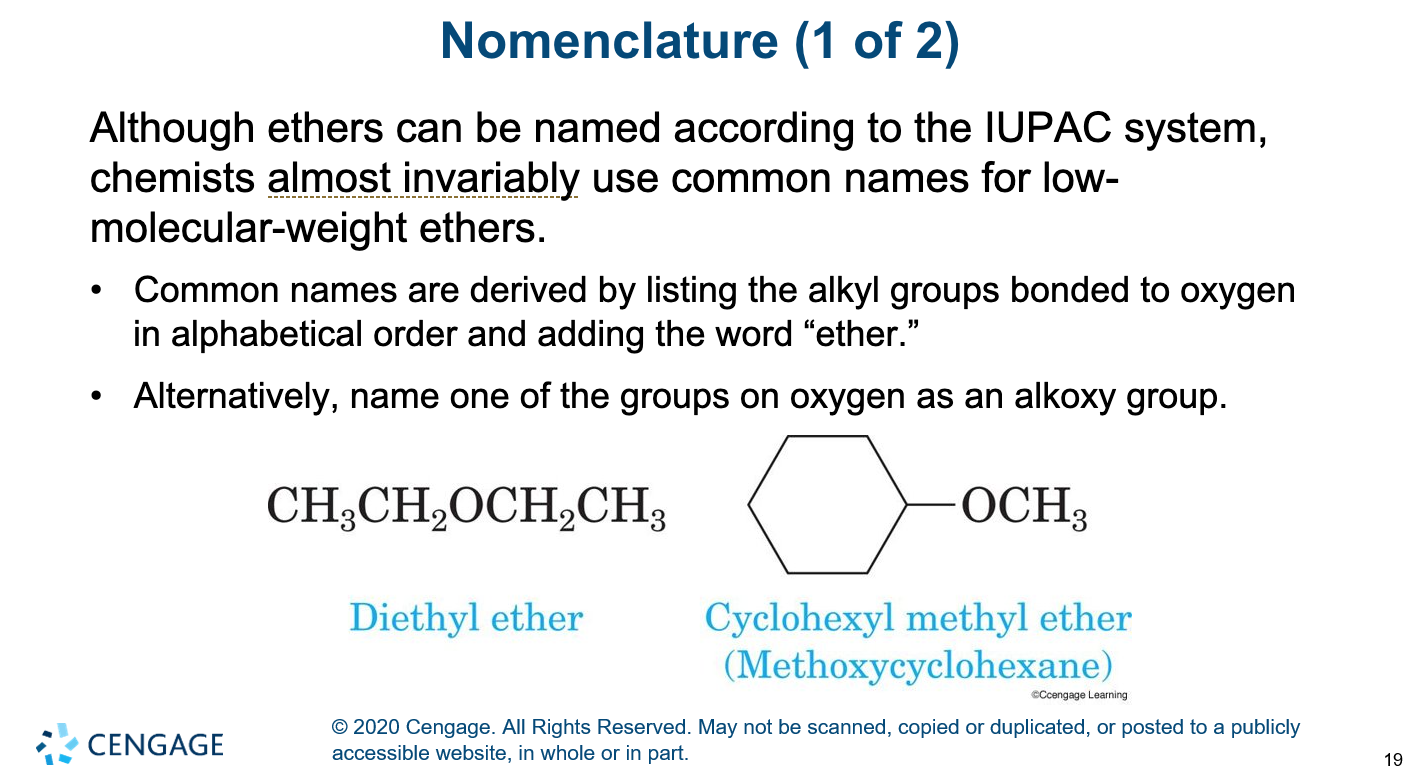
diethyl ether
cyclic ether
one of atoms is oxygen
thiols
contains sulfhydryl group (SH), smelly
common name of thiols
adding word ‘mercaptan’
polarity of thiols
nonpolar covalent, little association of hydrogen bonding, low bp, low solubility
acidity of thiols
weak acid (high pKa)
describe oxidation of thiols
oxidation to disulfides, susceptible to oxidation
important alcohols
ethanol, methanol, isopropyl alcohol, and glycerol, propene (raw material base)
nomenclature of ethers
physical properties of ethers
1. polar
2. weak forces
3. bp close to hydrocarbons of same molecular weight but lower than alcohols with same molecular formula
nomenclature of thiols
