Acids, Alkalis and Titrations (2c)
1/10
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
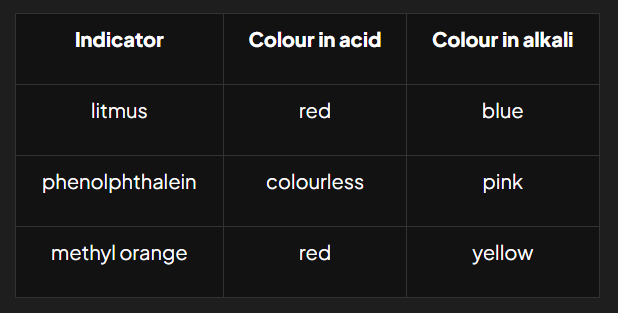
Litmus
comes from lichen → natural
not suitable for titration as colour change is not sharp
there’s a purple transition colour in neutral solutions → endpoint is difficult to determine
Synthetic Indicators
organic compounds that are sensitive to changes in acidity and appear different colours in acids and alkali
ex: phenolphthalein and methyl orange
used to show endpoint in titrations as they have very sharp change of colour when an acid has been neutralised by an alkali and vice versa
Two Colour Indicators
Two colours indicators are used to distinguish between acids and alkalis
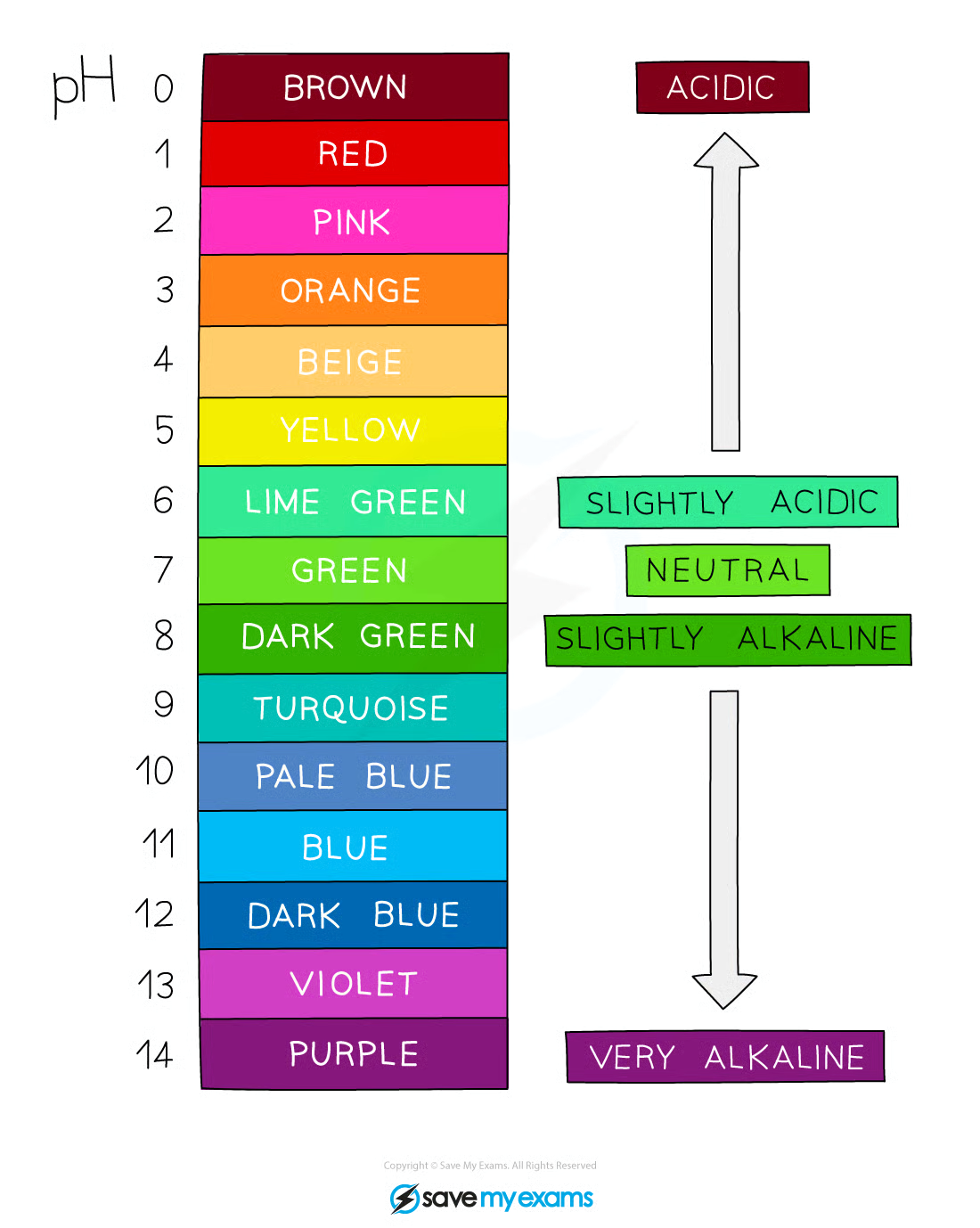
pH Scale
0-14
acids = <7, alkalis = >7
0-3 = strong acid
extremely acidic = below 1
4-6 = weak acid
8-10 = weak alkali
11-14 = strong alkali
7 = neutral
Universal Indicator
wide range indicator
only approx. value for pH
mixture of different plant indicators which operate across a broad range and useful for estimating the pH of an unknown solution
few drops are added to the chart which indicates the pH match with specific colours
charts vary by manufacturer so charts are provided for specific indicator formula
Acids
when added to water, produce H+ ions
these ions make a solution acidic
Alkali
when added to water, produce OH- ions
these ions make an aqueous solution alkaline
pH Scale Measures
numerical scale measuring how acidic or alkaline a substance is
measure of the amount hydrogen ions
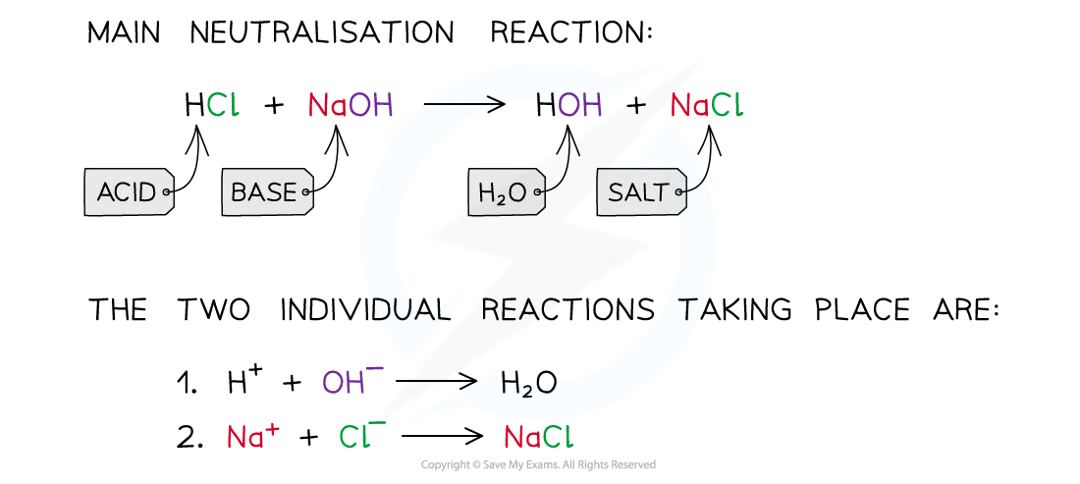
Neutralisation
reaction occurs when an acid reacts with an alkali
when these substances react, the H+ ions react with the OH– ions to produce water
not all acid reactions are neutralisations
important for treatment of soils to raise the pH as some crops can’t tolerate pH levels below 7 → achieved by adding bases like limestone and quicklime
Titrations
method of analysing conc. of solutions
acid-base titrations determine how much alkali is needed to neutralise an acid
can also be used to prepare salts
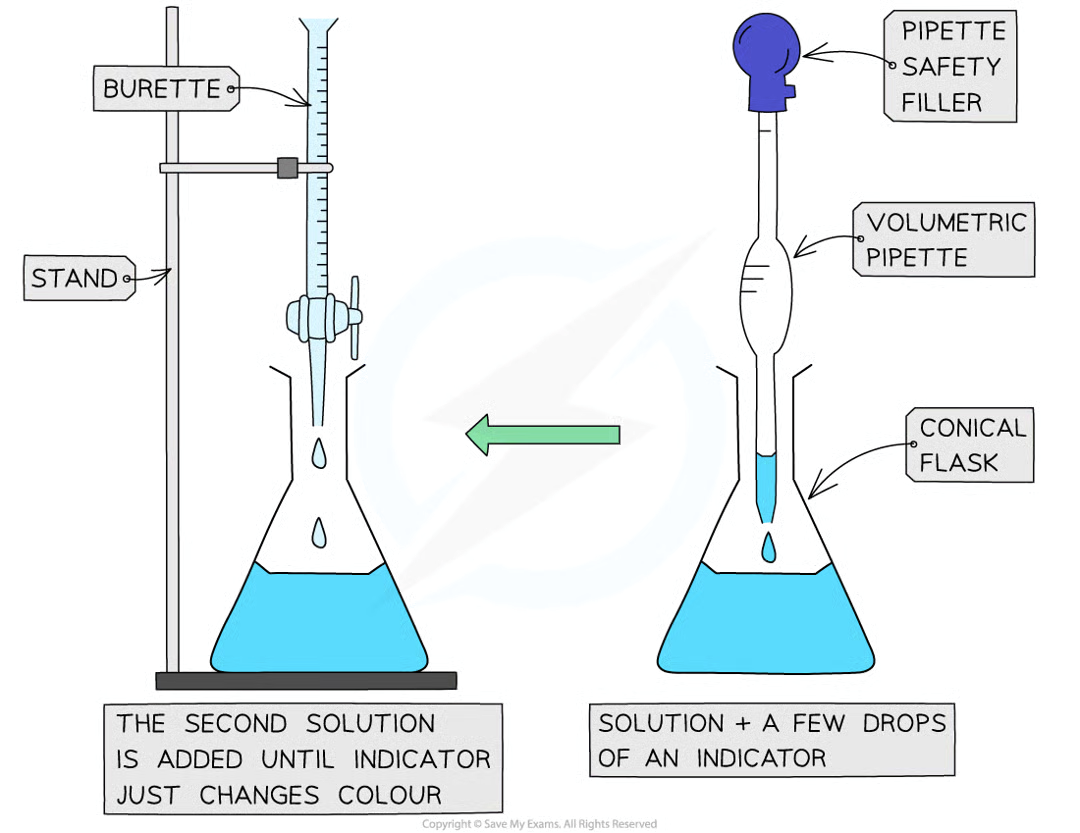
Practical (Acid-Base Titration): Method
use pipette and pipette filler and place exactly 25cc of NaOH in the conical flask
fill burette with HCl, place an empty beaker under the tap
run small portion of acid through burette to remove any air bubbles
record starting point on burette to nearest 0.05cm3
place conical flask on white tile so burette tip is inside the flask
add few drops of indicator to the solution in the conical flask
perform rough titration by taking burette reading and running the solution in 1-3 cc portions, while swirling vigorously
quickly close tap when end point is reached (sharp colour change) and record volume, placing eye level with meniscus
repeat with fresh NaOH
as rough end-point is approached, add solution from burette drop by drop until indicator changes colour
record volume to nearest 0.05 cm3
repeat until you have 2 concordant results (within 0.1cc of each other) to increase accuracy