5.2 energy
1/58
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
59 Terms
what are the ∆H units
Kj mol⁻¹
what is meant by ∆f H ?
Enthalpy change when elements form 1 mole of a compound under standard conditions
what is meant by ∆at H?
Formation of 1 mol of gaseous atoms
what is meant by first ionisation energy?
energy needed to remove 1 e⁻ from 1 mole of gaseous atoms
what is meant by second ionisation energy
energy needed remove 1 e⁻ from 1 mole of gaseous ion atoms
what is meant by first electron affinity ?
1 mol of gaseous atoms gain 1 mol of e⁻
what is meant by second electron affinity
energy needed to change 1 mole of gaseous 1- ions into gaseous 2- ions ( adding another e⁻ )
what is meant by ∆LE H (lattice enthalpy) ?
Formation of 1 mol of ionic lattice from gaseous ions
what is meant by ∆hyd H (Hydration) ?
dissolving of 1 mol of gaseous ions in water
∆H of hydration general equation
X(g) → X(aq)
what is meant by ∆sol H (solution) ?
dissolving 1 mol of solute
∆sol H general equation
YX(s) → YX(aq)
First e⁻ affinity general equation
X(g) + e⁻ → X⁻(g)
Second e⁻ affinity general equation
X⁻(g) + e⁻ → X²-(g)
bond strength and lattice enthalpy relationship
More negative the lattice enthalpy, the stronger the ionic bonding
What are the factors affecting lattice enthalpy
ionic charge
ionic radius
How does ionic radius affect lattice enthalpy
the smaller the ionic radius of ions → higher charge density → stronger electrostatic force of attraction between ions → lattice enthalpy is more exothermic
How does ionic charge affect lattice enthalpy
higher ionic charge → stronger the electrostatic force of attraction → lattice enthalpy is more exothermic
Ionic radius relationship
Increases down the group
When to use a Born-Haber cycle
Ionic compounds
Born-Haber cycle where do endothermic reactions arrows go
Arrows point upwards → energy increases
Born-Haber cycle where do exothermic reactions arrows go
Arrows point downwards → energy decreases
Born Harbour cycle General structure ( Image)
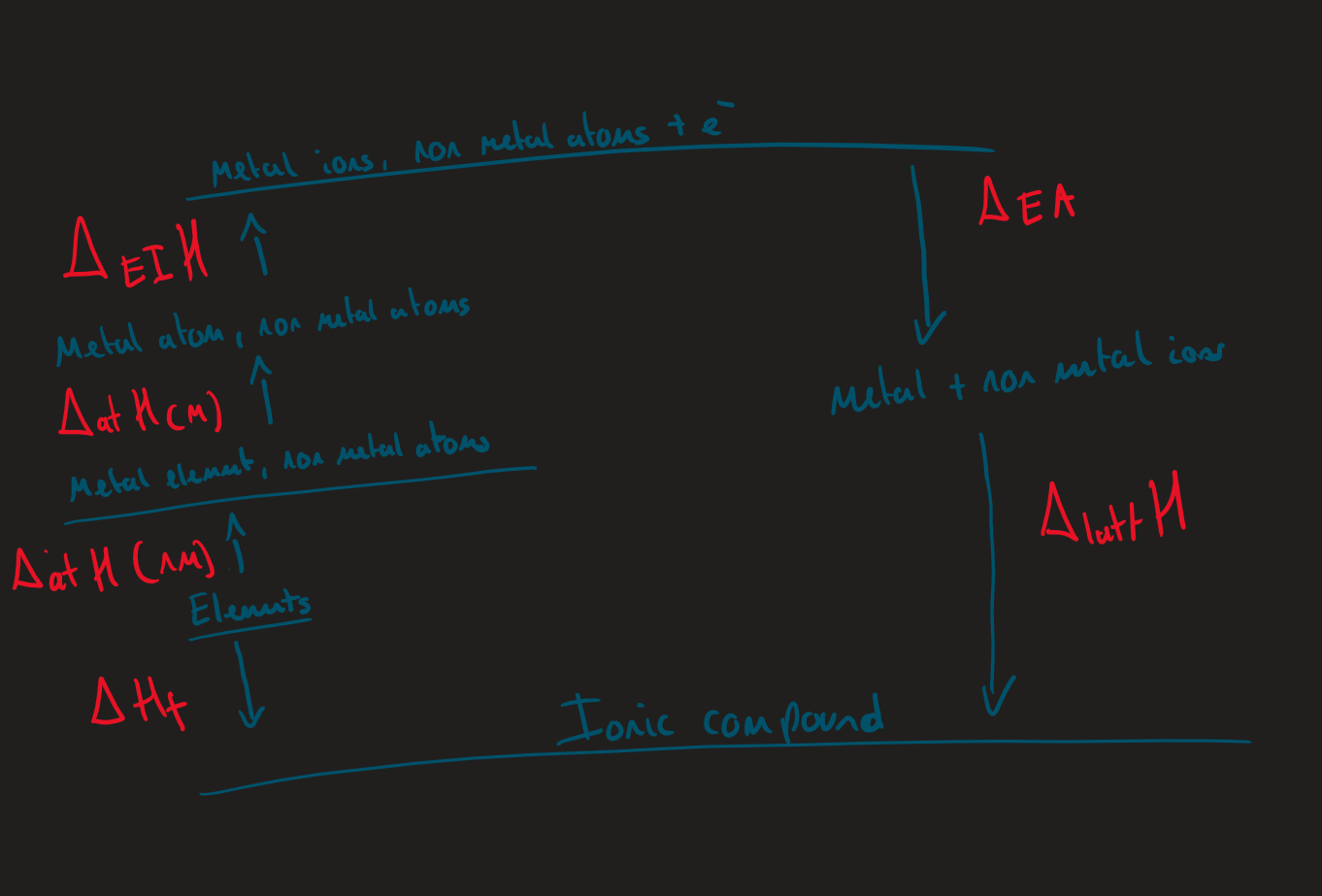
What is the direction of born haber cycles
Atoms → gaseous atoms → gaseous ions → ionic lattice
Why are born cycles used to measure lattice enthalpy
lattice enthalpy can't be measured directly due to gaseous ions
Born-Habour cycle what happens when a diatomic element undergoes ∆Hat
forms 1 gaseous atom
Born-Habour cycle why is ∆Hea2 always endothermic
requires energy to get over the e⁻ / anion repulsion
Born-Habour cycle why is ∆Hea1 always exothermic
e⁻ is added to atom → attracted to positive nucleus
releases energy
born Harbour cycle what to do to ∆Hatt and ∆Hea when forming more then one atom
∆Hatt /∆Hea x by number of atoms / ions formed
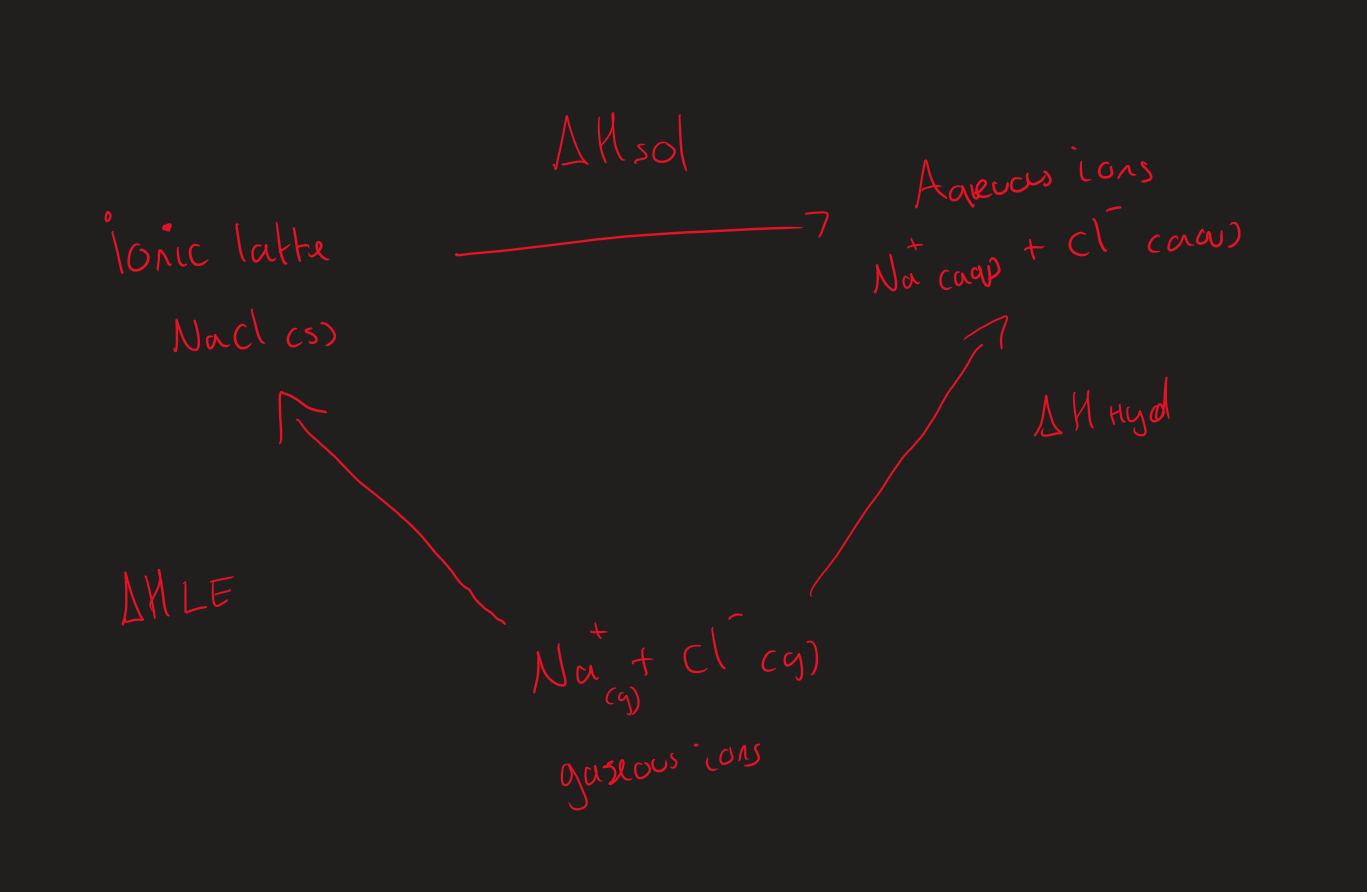
NaCl ∆Hsol reaction image ( Image)
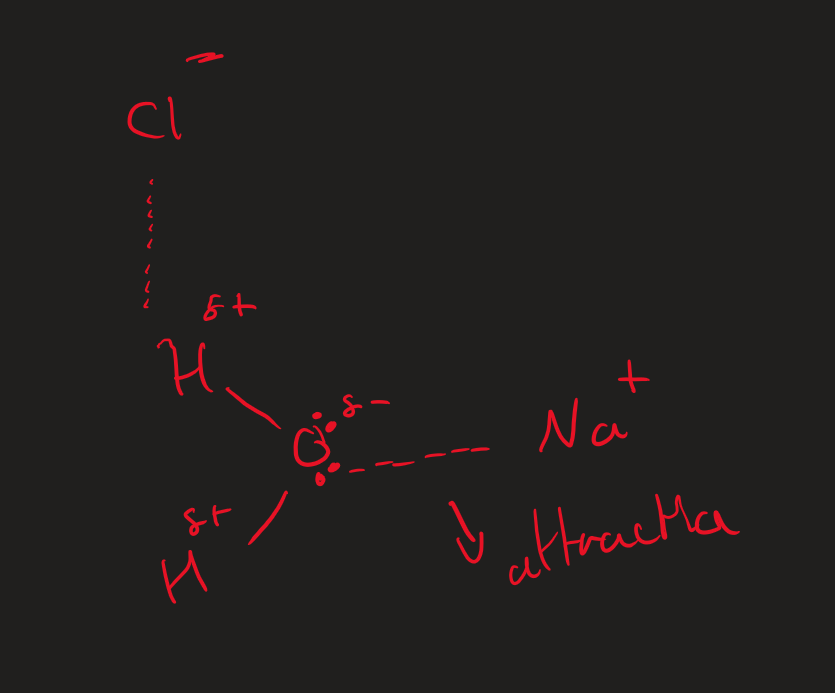
Why is ∆Hhyd always exothermic
Electronegative O in H₂O is attracted to metal cation
attraction → releases energy
What are the ∆H needed to work out ∆Hsol ?
∆Hhyd
∆HLE
∆Hsol
What are the factors effecting ∆Hhyd ?
Ionic charge → greater the ionic charge
Ionic radius → smaller the ion→ more exothermic ∆Hhyd + ∆HLE
Stronger attraction to H₂O
Suggest a reason why ∆Hsol is more exothermic in RbF then KF
K has a smaller ionic radius then Rb
Which ∆H is more significant in group 1 fluorides?
∆HLE is more significant than ∆Hhyd
Which ∆H is more significant in group 1 chlorides?
∆Hhyd is more significant ∆HLE
What is entropy (S)
A measure of the dispersal of energy in particles
disorder and entropy relationship
As disorder increases, entropy increases
What are the units of entropy ( S)
S → JK⁻¹mol⁻¹
State of matter and entropy relationship ?
Gas → random → highest disorder
liquid
solid ( fixed regular pattern) → lowest disorder
How to increase entropy?
Thermal decomposition
dissolving a solid
More moles of Gas in reactants
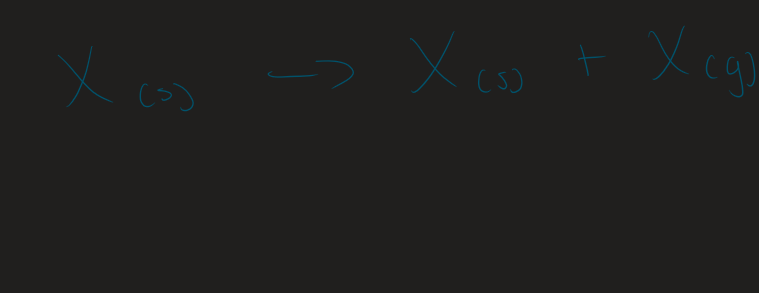
thermal decomposition reaction what is happening to entropy?
Solid → gas
more disorder in reactants
S increases ∆S is positive

Explain the changes in entropy
∆S is negative
S decreases
3 mol of gas to 2 mol of gas
How to calculate ∆S ?

what does a reaction being feasible/ spontaneous mean?
Processes occur without a continuous supply of energy
what does ∆G mean
Gibbs free energy
When are reactions feasible ?
Negative ∆G
∆G is less then 0
reaction is feasible
How to work out ∆G
∆G = ∆H - T∆S
what are the units for T
K
How to convert JK⁻¹mol⁻¹ to KJK⁻¹mol⁻¹
/1000
How to convert KJK⁻¹mol⁻¹ to JK⁻¹mol⁻¹
X 1000
How to work minimum temperature needed for a reaction to be feasible?
T = ∆H / ∆S
∆S / 1000 → get KJK⁻¹mol⁻¹
Limitations of ∆G
Predicts in standard conditions only
Reaction may not occur due to too high EA
Feasibility doesn’t equal rate ( kinetics)
Which ∆H enthalpies always point down ?
∆Hf
∆Hea1
∆HLE ( all exothermic)
Suggest why its difficult to use a born- haber cycle for carbonates / sulfates
Carbonate ions contain more than one type of atom
Cycle would need to include formation of carbonates
Conditions for a reaction to be feasible + effect of increasing temp
Exothermic ( - ∆H)
+∆S
∆G is less than 0 → Reaction is feasible
Increase T → ∆H gets more negative → more feasible
Conditions for a reaction not be feasible + effect of increasing temp
Endothermic ( + ∆H)
-∆S
∆G is more than 0 → not feasible
increase temp → T∆S becomes more positive
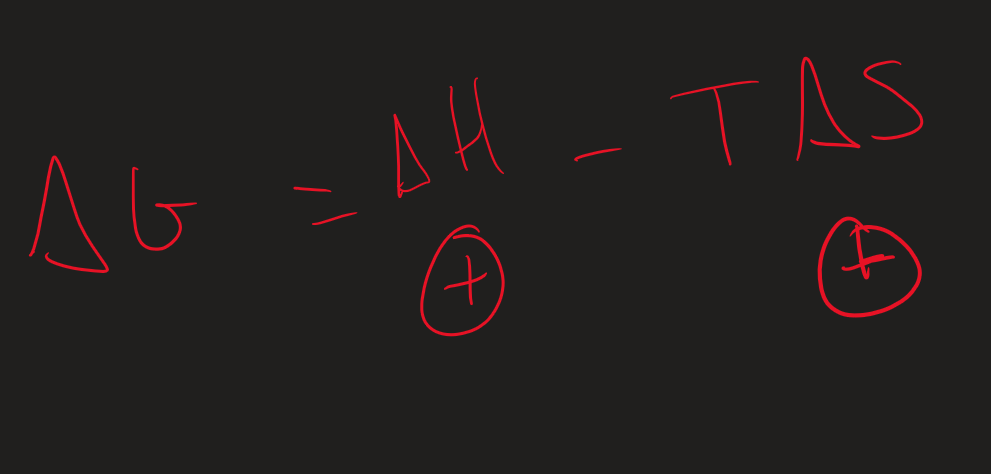
Effect of feasibility + Temperature
More feasible as T increases
∆G gets less positive
Why is the ∆H Hyd of Cl⁻ ions / anions exothermic?
bonds form between the Cl⁻ and H