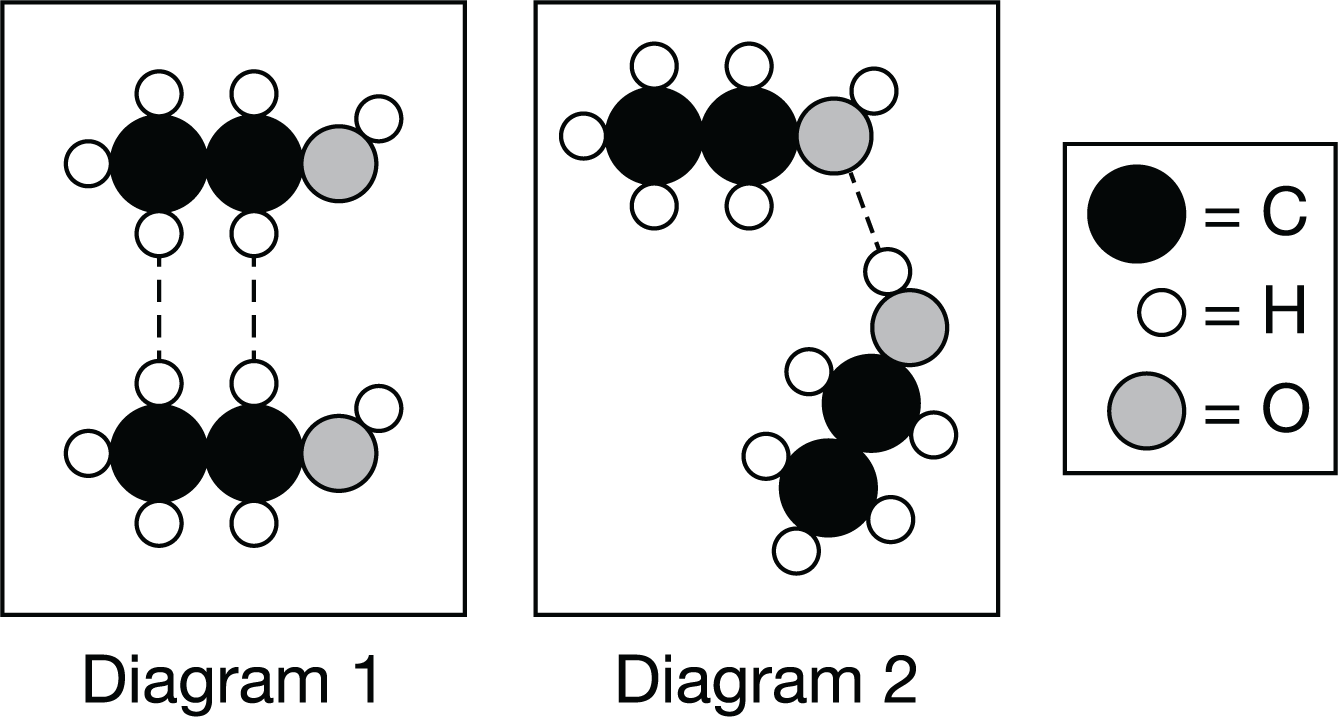
Which particle diagram shown above best represents the strongest intermolecular force between two ethanol, C2H6O, molecules?
Diagram 2, because it shows the formation of a hydrogen bond between an H atom bonded to an O atom with an O atom from anothe
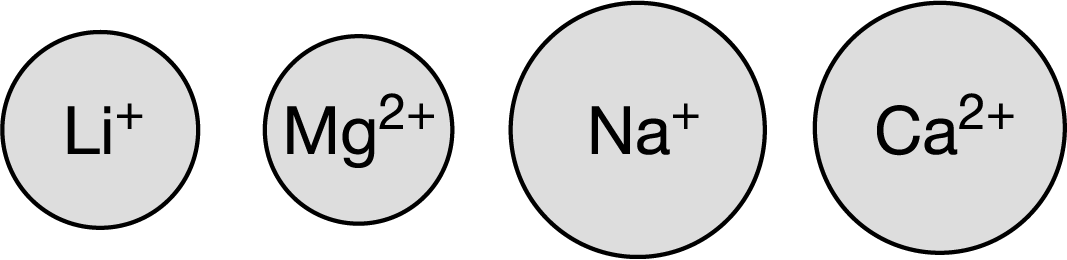
The diagram above represents four cations, all shown to the same scale. Which cation would be predicted by Coulomb’s law to have the strongest ion-dipole attraction to water, and why?
idk
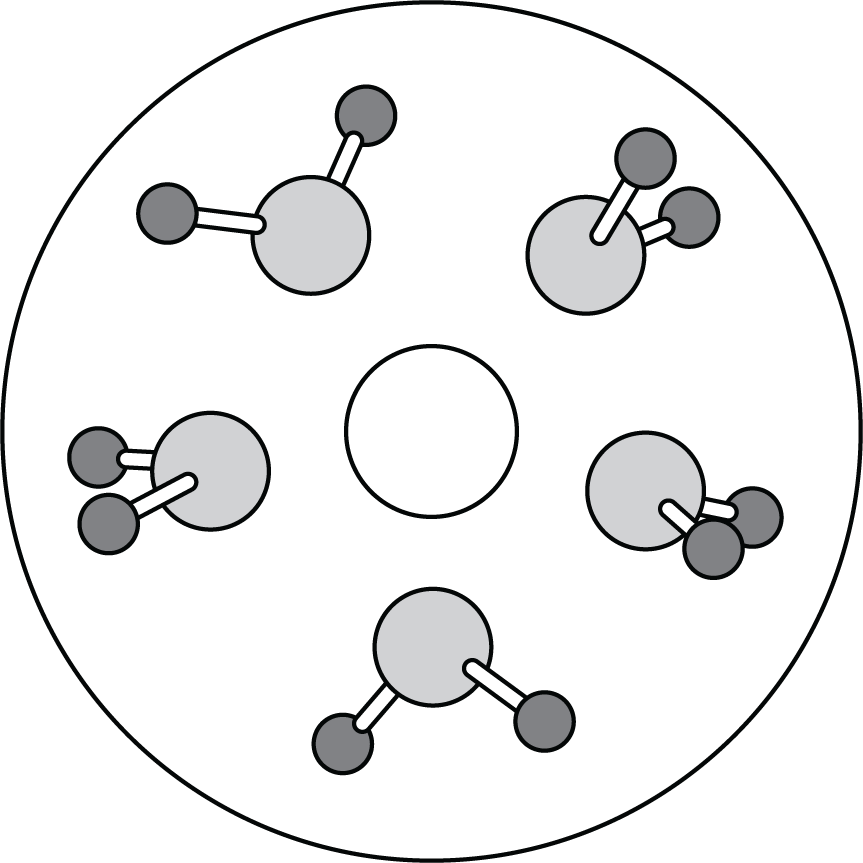
A solid compound of a group 1 (alkali) metal and a group 17 (halogen) element dissolves in water. The diagram above represents one type of solute particles present in the solution. Which of the following identifies the solute particles and best explains how the solute particles interacts with water molecules?
idk
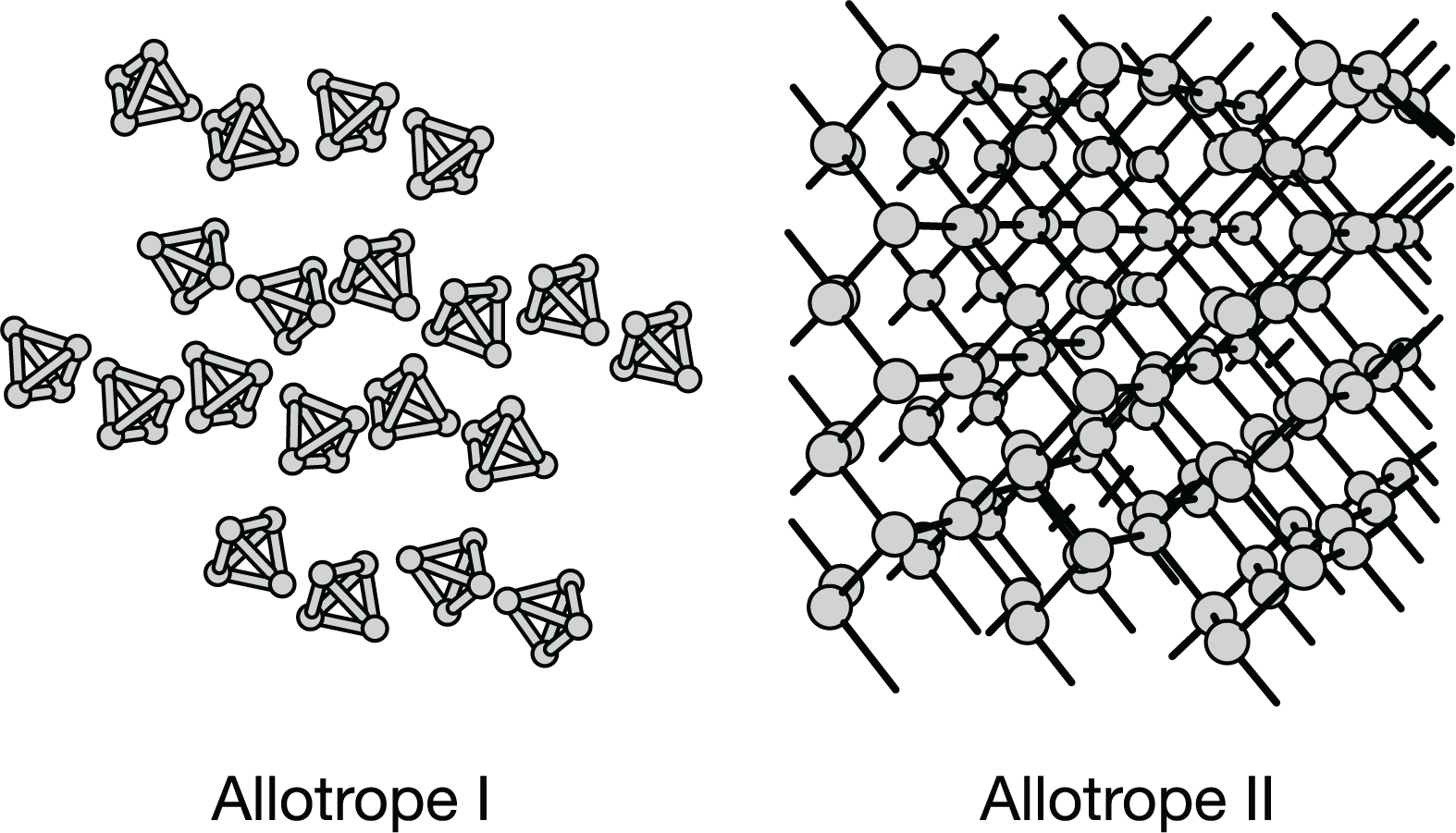
The diagram above represents two allotropes of solid phosphorus. Which of the following correctly identifies the alotrope with the higher melting point and explains why?
idk