unit 1
1/152
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
153 Terms
what two things can a medicine be?
licensed, unlicensed
can a prescriber still make and/or supply an unlicensed medicine?
yes
what do licensed medicines have that unlicensed don’t?
marketing authorisation number
what two ways can an unlicensed medicine be made?
by a manufacturer who holds a manufacturing specials license, under the supervision of a pharmacist
what is formulation science?
science of turning a drug into a medicine
what is the the active pharmaceutical ingredient
substance that has a direct effect in the diagnosis, cure and treatment of disease
what is an excipient
component other than the active substance which is present in a medicinal product
what 3 things does formulating/formulations improve?
patient compliance, API stability, pharmacokinetic profile
formulation cannot give a drug activity which it would not have but a poor formulation can affect?
activity
high drug-excipient interaction causes?
poor release
low drug-excipient interaction causes?
dump dosing
what are the two types of delivery routes?
invasive, non invasive
what is an invasive method?
a method drug delivery that accesses the target via physical trauma
what is non invasive?
a method of drug delivery that accesses the target without physical damage to tissue
what is a systemic delivery
drug enters circulation and is transported around the body
what is a local delivery
drug is administered at or close to target site
what is bioavailability
fraction of unchanged drug reaching the systemic circulation by any route
to get 100% bioavailability the entire dose of an orally delivered drug must:
be completely — from the dosage form
be fully — in the — fluids
be — in the — fluids
pass through the — — without being —
pass through — with being —
released, dissolved, GI, stable, GI, GI barrier, metabolised, liver, metabolised
what shows the varying concentration of a drug in systemic circulation
pharmacokinetic profile
what is Cmax
maximum concentration of the drug in the body following administration of a dose formulation
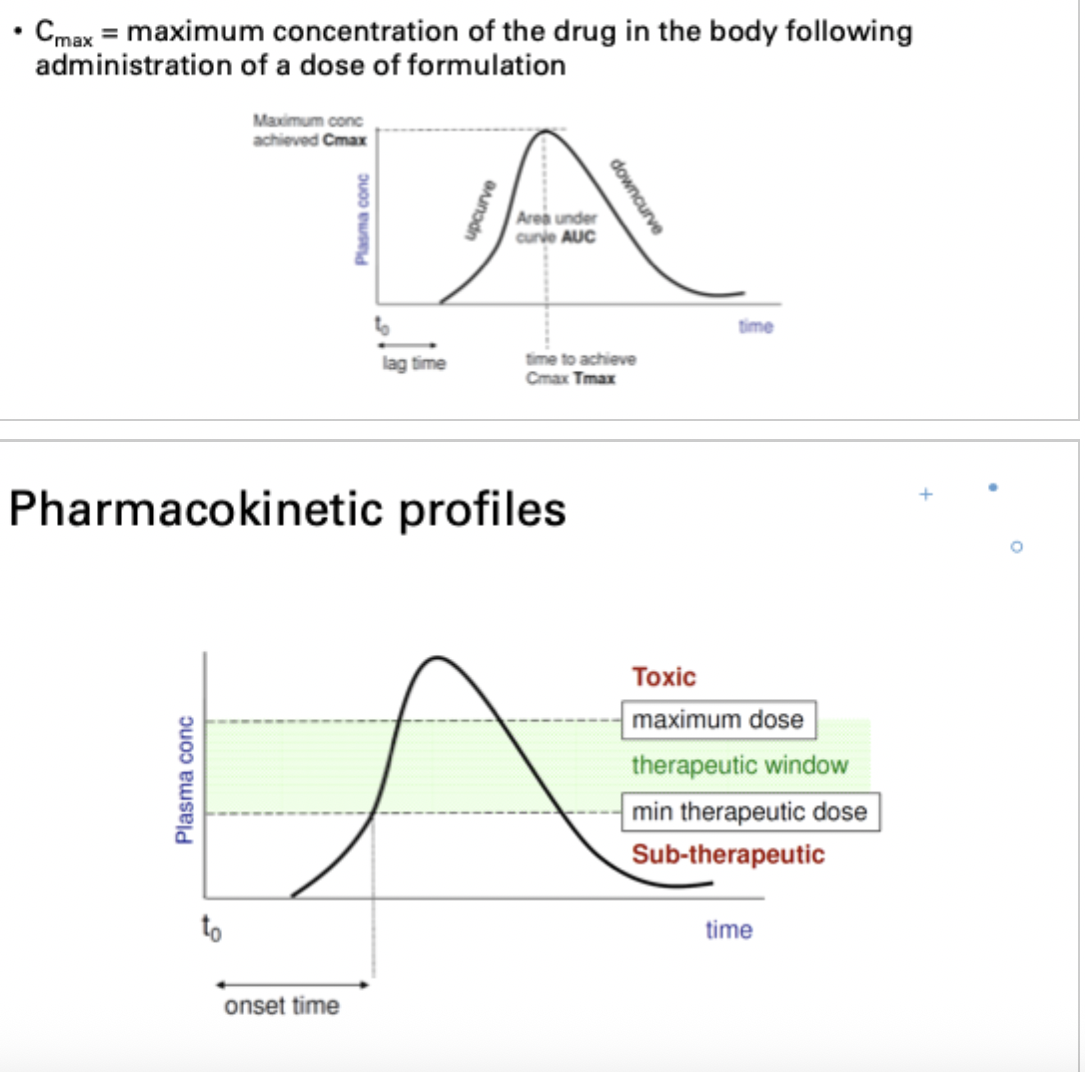
what is the aim of a dosage regimen?
maintain constant therapeutic plasma concentration for duration of therapy
what is a certain formulation that can be used to deliver and maintain consistent drug levels in the circulation (2 answers)
modified release tablets, transdermal patches
what is considered for preparations where they are not for immediate use (also important when water and/or natural products are present)
preservatives
what do preservatives need
high water solubility
COMMON EXCIPIENTS: Density Modifiers
the smaller the density difference between phases-
the less sedimentation will occur
COMMON EXCIPIENTS: do antioxidants prevent oxidation?
no
COMMON EXCIPIENTS: what is the function of buffers?
influence chemical stability, tonicity and physiological compatibility
COMMON EXCIPIENTS: what is the function of organoleptic substances?
mask flavour of drug
COMMON EXCIPIENTS: function of humectants
decrease evaporation of water
what is a solution
mixture of two or more components that form a single homogenous phase
what is the solvent
phase in which the distribution occurs
in liquid formulations the solvent is called the?
vehicle
in solid or semi solid formulations what is it called?
base
what is the solute
component which is dispersed as discrete molecules in the solvent
why do we use a solution?
drug is already in its molecular form: — — of action
drug does not require —: this allows rapid uptake following oral delivery
their — nature means precise tailored doses can be delivered
easy to —
rapid onset, dissolution, homogenous, swallow
DISADVANTAGES OF SOLUTIONS:
some drugs are — when in solution and so would not be logistically feasible to make solutions using them
not all drugs have adequate — — to make a suitably potent solution
liquids are generally — and take up more space than equivalent solid oral dosage forms
— of liquids is also more challenging
unstable, aqueous solubility, heavier, packaging
what is dissolution
transfer of molecules/ions from the solid state into solution
why does this happen?
the forces that bind adjacent molecules of the solute together are overcome by attractive forces of molecules of solvent
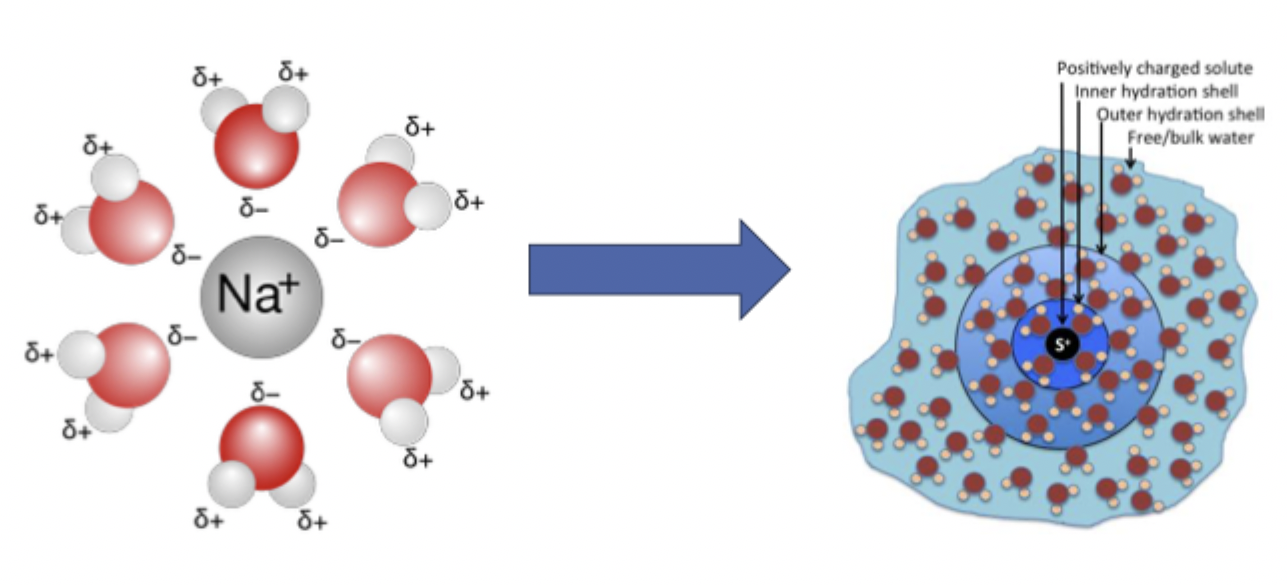
in dissolution, the liberated molecule of the solute becomes —, surrounded by solvent molecules in an arrangement known as a — —
solvated, solvation shell
what 3 things speed up dissolution
heat, kinetic energy, ultrasonication
what is solubility
extent to which dissolution proceeds under a given set of experimental conditions
AKA the amount of a substance that passes into solution when — is established between the — and — (undissolved) substance (forming a — solution)
equilibrium, solution, excess, saturated
what is miscibility
two components forming a solution are either both gases or both liquids
what is a saturated solution
solution in which no more solvent molecules are available to form solvation shells
what is the difference between solubility and dissolution?
solubility is the amount that can be dissolved and dissolution is how quickly solute enters solution
what does logP measure
how lipophilic or hydrophilic a drug molecule is
what is the equation for logP
solubility of drug in non aqueous medium/ solubility of drug in aqueous medium
can ionised drugs exist in isolation?
no
what do they need
counterion
what is an amorphous drug
drug in a random arrangement of molecules
what is the opposite of this?
crystalline drug
some drugs can form more than one crystalline arrangement- what is this described as?
polymorphic
why would more than one co-solvent be employed?
maximise drugs solubility and optimise efficacy and stability
the salt of a drug that is used can determine what?
formulation properties
what can addition of salt increase in a too lipophilic pure drug?
polarity
what is the function of buffers
maintain pH within a given range over a long period of time
what are semi synthetic polysaccharides made up of glucose molecules bound together in a cyclic “bottomless bucket” structure
cyclodextrins
what do cyclodextrins form?
guest host inclusion complexes
what does the inclusion complexation modify?
physical and chemical properties of the guest molecule
mainly by increasing?
aqueous solubility
what does it rely on?
dissociation at target membrane
what is the function of the formation of micelles/liposomes?
solubilise poorly soluble drugs
what are disperse systems?
systems that consist of 2 or more components in a heterogeneous mixture
what are the two different types of dispersion?
colloidal, coarse
which one has smaller particles?
colloidal
in colloidal dispersions, the particles are not visible to the naked eye- what do you require to see them?
electron microscopy
how are they distinguishable from solutions?
has an effect on light
colloidal dispersions differ from coarse dispersions in that their particles are small enough that — does not exert enough of an effect on them to cause them to settle. this means they can only be separated using —
gravity, ultracentrifugation
in colloidal dispersions the particles move by — — — and so will diffuse
random brownian motion
coarse dispersions are commonly referred to as?
suspensions
COARSE:
larger particles are liable to — due to gravitational forces which causes them to settle upon landing
— must be slow enough that a — dose can be obtained following shaking
sedimentation, sedimentation, homogenous
what type of drug may be formulated as a suspension?
drugs susceptible to aqueous degradation

advantages of disperse systems
done

disadvantages of disperse systems
done

oral medicines vs taste buds
done
PROPERTIES OF AN IDEAL DISPERSE SYSTEM MEDICINE:
particles should be — — in the — phase
particles must not — too rapidly
sediment must be easily — —
must easily flow out of the container
particle size should be small and —
evenly distributed, continuous, sediment, re dispersed, uniform
what are the 3 classifications of colloidal systems
lyophobic, lyophilic, amphiphilic
which one is solvent hating
lyophobic
which one is solvent loving?
lyophilic
which one is both loving
amphiphilic
which system is the most thermodynamically unstable
lyophobic
what happens to the particles in lyophobic colloids
aggregate to lower their surface energy
in lyophilic colloids the particles have an affinity for the dispersant and this leads to —. this protective dispersant “coat” prevents — so the dispersion is inherently —
solvation, coagulation, stable
what is the critical micelle concentration?
molecules will arrange at the surface of the continuous phase until there is no space left
what happens after this point?
amphiphiles form micelles
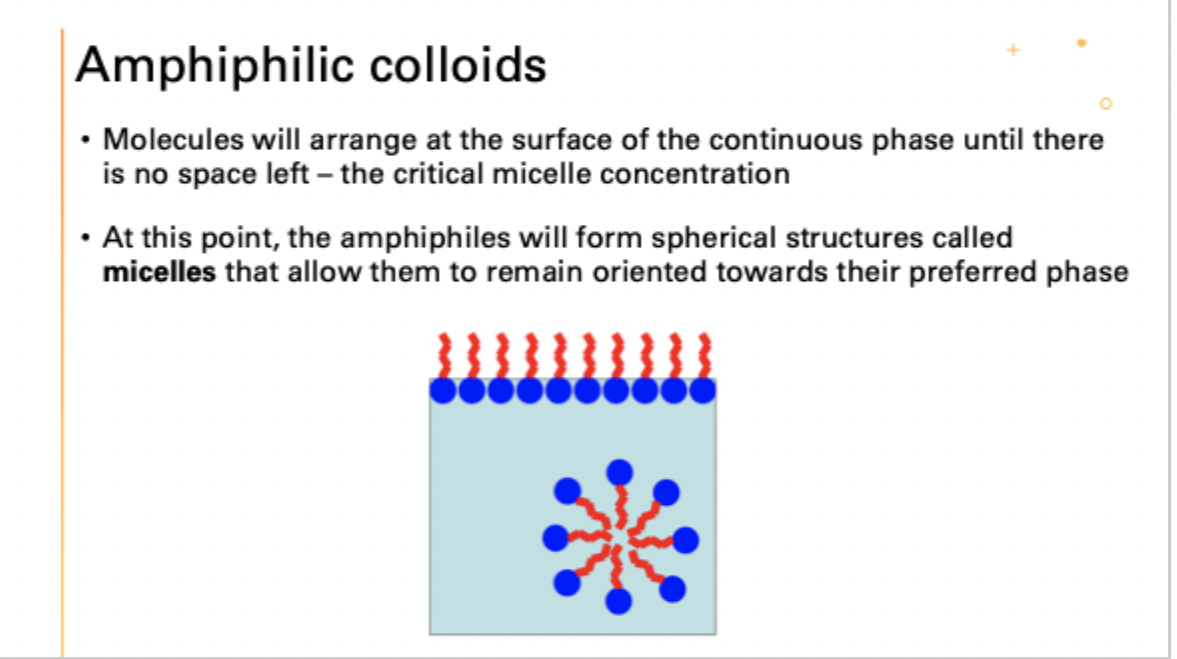
what is a bilayer amphiphile structure?
liposomes
liposomes have a pocket of — phase within them which can be used to encapsulate — materials to control — and protect them from — degradation
— drugs can be dissolved within the middle of the micelle or the bilayer of the liposome and this is a means of utilising colloidal systems to solubilise poorly soluble drugs
continuous, lyophilic, delivery, enzymatic, lyophobic
what are association colloids?
micelles and liposomes constantly disassociate and reassociate with one another to form new structures
what is the name of the effect that occurs when light is shone through a colloidal dispersion?
tyndall effect

optical properties of colloids
done

tyndall effect
done
this light scattering property can be utilised to measure what within a disperse system?
particle size
suspensions are good for remaining oily globules after injection
this is a positive because it reduces — — in contact with physiological fluids and therefore slowing the rate of —
surface area, absorption
what is the definition of wetting?
ability of a solid particle to stay in contact with a liquid
what type of solid is more easily wetted?
diffusible solids
why are indiffusible solids not as easily wetted?
show some hydrophobicity
what do they need in addition to be wetted?
wetting agent
why are finely divided substances particularly difficult to wet?
presence of an absorbed layer of air around the particles
what is the function of a wetting agent?
decrease interfacial tension and help dispersion

interfacial tension slide
done