OZ 8 What is the state of the ozone layer now?
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
Reaction of homolytic fission of haloalkanes when X in alkane group?
XCl + hv → X + Cl
Substitution
reaction in which one atom or group in a molecule is replaced by another atom or group
Nucleophile
a molecule or negatively charged ion with a lone pair of electrons that it can donate to a positively charged atom to form a covalent bond
nucleophilic subsitution
a nucleophile replacing another nucleophile
Nucleophilic substitution of OH to 1-bromobutane
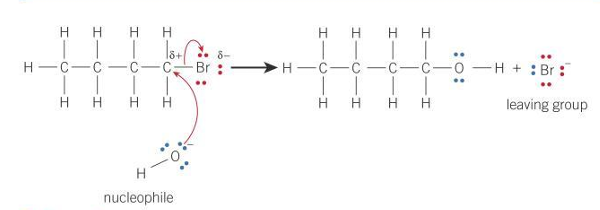
General equation for nucleophilic substitution using Nu- as nucleophile and X as halogen
R— X + Nu⁻ → R — Nu + X⁻
Reaction of water and haloalkane
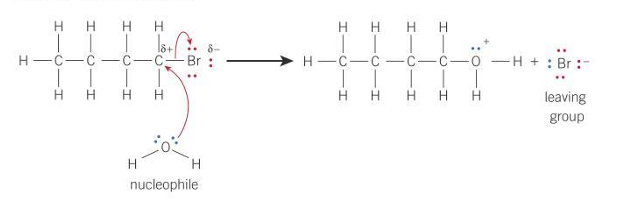
Hydrolysis reaction of water and haloalkane
R — X + H₂O → R — OH + H⁺ + X⁻
General formula of amines
R-NH₂
Why are water and ammonia nucleophiles?
lone pair of electrons on O and N
Conditions for a reverse hydrolysis reaction
strong acid
Describe the steps for a reaction of a halide with an alcohol
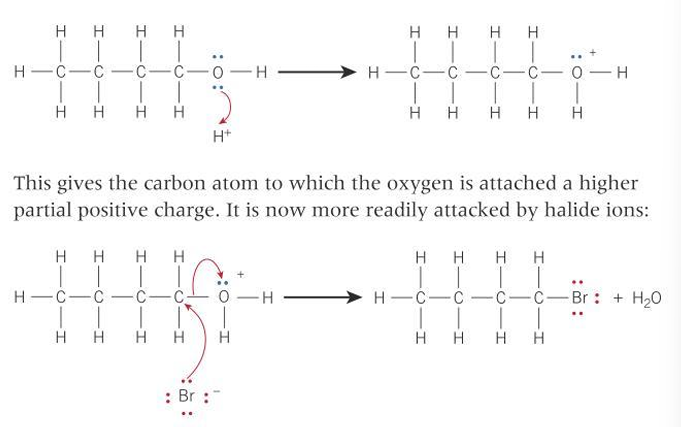
What is the trend in strength of carbon-halogen bond down the group?
weaker
Hydrolysis
nucleophilic substitution reaction, where water or hydroxide replaces the halogen
What is the most important factor in hydrolysis?
bond strength
Why does hydrolysis of carbon-halogen molecules get easier down the group?
bond gets weaker so break more easily
compounds get more reactive
Weaker bonds
faster hydrolysis
Conditions for hydrolysis
reflux
heat