Exam 2 Kinetics multiple choice practice
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
What is kinetics?
the study of how fast a chemical reaction happens and how the reaction happens
How do we determine the rate of a reaction?
by seeing how concentration changes over time
What is the equation for measuring the rate of a reaction?
rate = —∆[reactant]/∆t or rate= —∆[product]/∆t
![<p>rate = —∆[reactant]/∆t or rate= —∆[product]/∆t </p>](https://assets.knowt.com/user-attachments/efd680a1-f6cf-450f-a8fb-84b70db70a76.png)
What affects the rate of a reaction?
Surface area, reactant concentration, temperature, catalysts and pressure
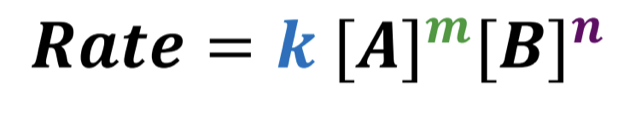
What is a differential rate law?
an equation that relates the reaction rate to the concentrations of reactants
How do you find the differential rate law from experimental data?
Compare trials where only one reactant concentration changes at a time. See how the rate changes to determine each reactant’s exponent, then solve for k.
What is reaction order?
tells how the rate depends on reactant concentration. It is the exponent on each reactant in the rate law. The overall order is the sum of all exponents.
What are integrated rate laws used for?
To find how much reactant or product is present after a certain time, or how long a reaction takes to reach a certain point.
What is the integrated rate law for a zero-order reaction?
[A]t=—kt+[A]0
What graph is linear for a zero-order reaction?
A plot of [A] versus time is linear.
What is the slope for a zero-order plot?
−k.
What is the integrated rate law for a first-order reaction?
ln[A]t = ln[A]0 − kt
What graph is linear for a first-order reaction?
A plot of ln[A] versus time is linear
What is the slope for a first-order plot?
−k
What is the integrated rate law for a second-order reaction?
1/[A]t = 1/[A]0 + kt
What graph is linear for a second-order reaction?
A plot of 1/[A] versus time is linear.
What is the slope for a second-order plot?
+k
What is half-life?
the time it takes for the concentration of a reactant to decrease to half of its original value.
What is the half-life formula for a zero-order reaction?
t1/2 = [A]0 / 2k
What is the half-life formula for a first-order reaction
t1/2 = 0.693 / k
What is the half-life formula for a second-order reaction
t1/2 = 1/k[A]0
How does the rate constant depend on temperature?
It increases exponentially as temperature increases
What equation shows how rate constant depends on temperature?
The Arrhenius equation: k = Ae (−Ea/RT)
What do the symbols in the Arrhenius equation mean?
k=rate constant, A=frequency factor, R=gas constant, T=temperature in Kelvin, Ea=activation energy
What is activation energy?
energy to overcome electron repulsion and form the transition state;
determines the rate constant of the reaction
Front: What is the transition state?
the unstable, high-energy arrangement of atoms that occurs during a chemical reaction
What are the points of interest on a reaction diagram?
reactants, products, activation energy, transition state, and the overall energy change, ΔWhat are the points of interest on a reaction diagram?
How does activation energy affect the rate constant?
A higher activation energy gives a smaller rate constant and a slower reaction. A lower activation energy gives a larger rate constant and a faster reaction.
What is a catalyst?
a substance that speeds up a reaction without being permanently used up by reducing the activation energy required.
What are the two types of catalysts discussed in class?
Homogeneous catalysts and heterogeneous catalysts
What is a homogeneous catalyst?
A substance that speeds up a chemical reaction while remaining in the same phase (solid, liquid, or gas) as the reactants
What is a heterogeneous catalyst?
A substance that speeds up a chemical reaction while existing in a different physical phase than the reactants.