Atomic Stucture, Scientists and Average Atomic Mass Test
1/16
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
17 Terms
Erwin Schrodinger
developed quantum wave equation
James Chadwick
discovered neutrons
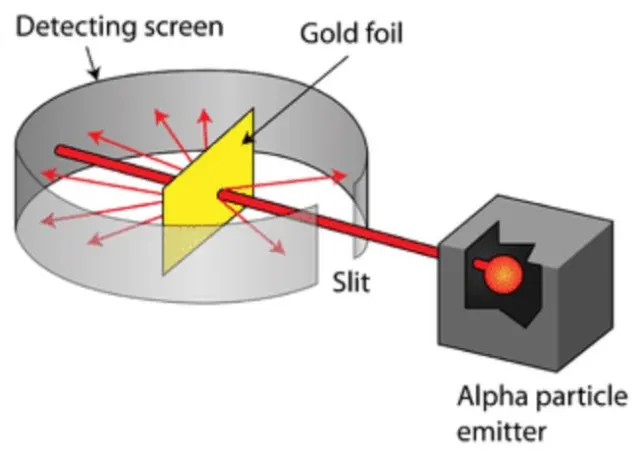
Ernest Rutherford
performed the Gold Foil Experiment that showed atoms were made up of mostly empty space and a nucleus in the middle
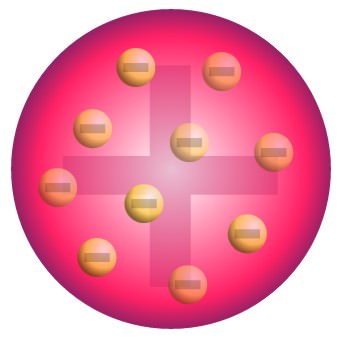
JJ Thomson
proposed Plum Pudding Model - discovered electrons but was incorrect in their location
Neil Bohr
proposed that electrons were in specific orbits around the nucleus
John Dalton
Dalton Atomic Theory
All elements made up of atoms
Atoms of a particular element are identical
Atoms can’t be created or destroyed
Chemical compounds are formed when different kinds of atoms combine
Chemical reactions are formed when atoms are rearranged — changes the way atoms are combined but not the atoms themselves
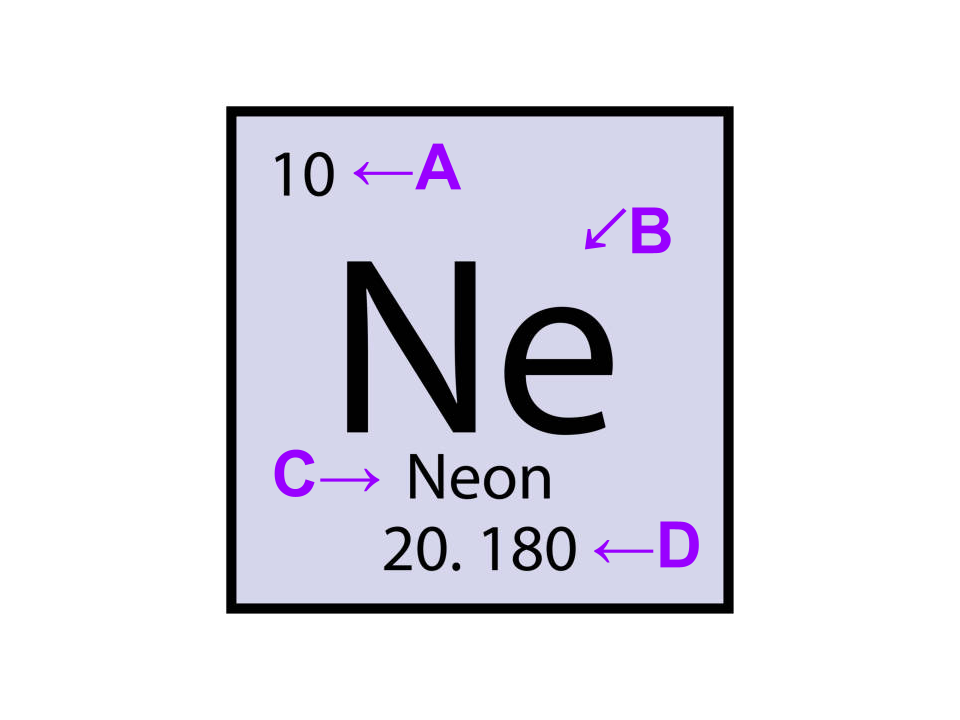
“label” the element
A - atomic number
B - element symbol
C - element name
D - atomic mass
atomic number
the number of protons
atomic mass
number of protons + number of neutrons; average of all atoms of that element in the world
neutral atoms
when the number of protons = number of electrons
ion
when the number of electrons are different than the number of protons
cation
less electrons; more protons
anion
more electrons; less protons
isotope
same number of protons, different number of neutrons
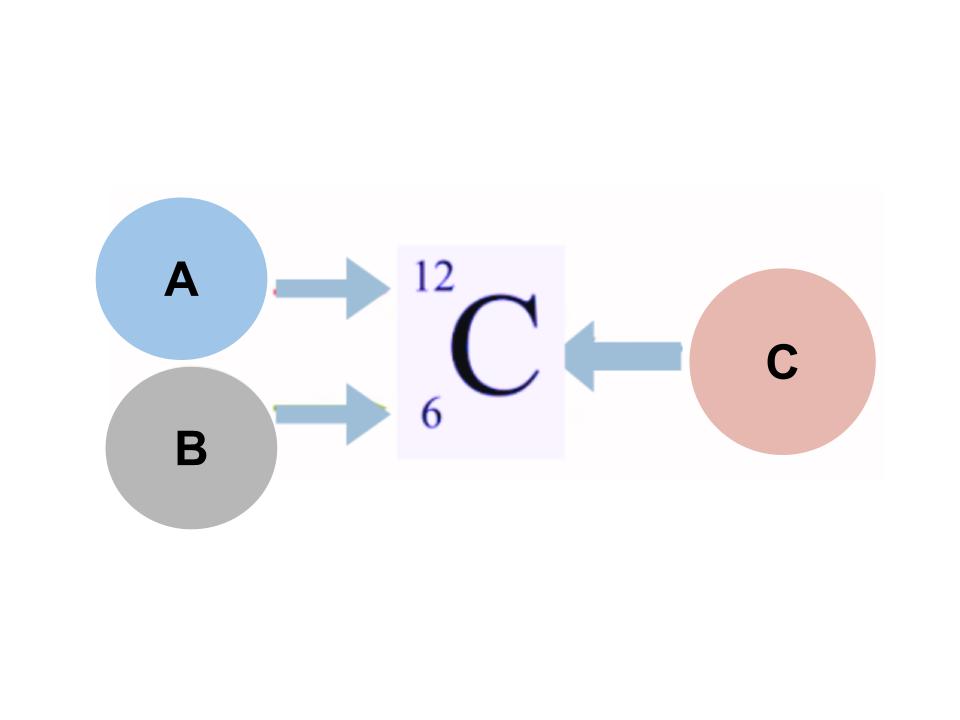
nuclear notation?
A - atomic mass
B - atomic number
C - element symbol
how to calculate average atomic mass?
sum of (% natural abundance)(atomic mass) for all isotopes given
Copper has two isotopes. Copper-63, which has an atomic mass of 62.93 amu and copper-65, which has an atomic mass of 64.93 amu. In any sample of copper atoms, 69.1% will be Copper-63 and 30.9% will be Copper-65. Calculate the average atomic mass of naturally occurring copper.
63.54 amu