Lecture 3 BIOT 511
1/50
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
51 Terms
What is the central dogma of biological sciences?
where DNA transcribes into mRNA which translated into protein
(exceptions to retroviruses HIV)
RNA → DNA → RNA → Protein (reverse transcriptase)
Non-coding RNA
Self-replicating proteins
How do you mass produce a protein?
getting a consensus protein sequence
obtaining the average sequences in mRNA (that is agreed upon)
And the overall gene expression systems
What is a gene clone?
a molecule, cell, or organisms that was produced from another single entity
What are the influences from restriction enzymes?
its a DNA cutting enzymes (molecular scissors) → possible from bacteria has been able to be discovered
What are plasmid DNA vectors?
circular form of self- replicating DNA (are outside bacteria)
they can be manipulated to carry and clone other pieces of DNA
can be inserted into other cells & protein of interest
move DNA form host to host (like uhaul)
Visual of Restriction Enzyme:
What are some visuals from restriction enzymes?
What are the main common restriction enzymes?
What type of restriction enzyme is EcoR1?
type 2 → cleaves and is within short distance
Which type of restriction enzymes are most used in industry?
Type 2 is the most common
What is plasmid DNA?
small circular pieces of DNA found primarily in bacteria
considered extrachrmosla DNA since they are in the cytoplasm in addition to bacterial chromosome
small (1- 4 kb)
Can be used as VECTORS (pieces of DNA that can accept, carry and replicate other pieces of DNA
What is a visual of a restriction enzyme?
What is a technique that can transform a bacterial cells?
CaCl2 Cold Shock (a slower process/ not so efficient)
a process that inserts foreign DNA into bacteria
treats bacterial cells w/ calcium chloride
adds plasmid DNA to cells chilled on ice
heats the cell & DNA mixture
plasmid DNA enters bacterial cells and is replicated & express their genes
What is a another technique that can transform a bacterial cells?
Electroporation (punches holes in bacterium)
applies a brief pulse of high voltage electricity to create tiny holes in the bacteria cell wall that allows the DNA to enter
What is the visual of stages of electroporation?
What is selection?
is a process that is designed to facilitate the identification of recombinant bacteria while preventing the growth of non-transformed bacteria and bacteria that contain plasmid w/o foreign DNA
selects but its resistant
selection of recombinant bacteria after transformation
What is are the a type of selection?
Antibiotic selection- plates that contain transformed cells on plates containing different antibiotics to identify recombinant bacteria and non-transformed bacteria
doesn’t select for plasmid containing foreign DNA vs. recircurlarized plasmid
What is another type of selection?
Blue-White selection - DNA is cloned into the restriction site in the lacZ gene
DNA is cloned into the restriction site in the lacZ
when is it interrupted by an inserted gene the lacZ gene can’t produce functional B gal
Xgal (artificial lactose) that is added to the plate, its functional lacZ is present = blue colony
Non functional lacZ → White colony = clone = genetically identical bacterial cells each containing copies of recomb. plasmid
Review 192 - 193 for the visual of plasmid & gene resistance
How was the insulin created?
The first human protein expressed by recombinant techniques which is insulin → growth hormone
Cloning human insulin (regulatory steps) DNA sequences into a plasmid and the bacterial cells were then used to synthesize the protein product of the cloned gene
not all proteins are small tho
small proteins don’t require alot but are doable
First source of growth hormone was from cattle (pancreas)
What makes a good vector?
size - that are small enough to be separated from chromosomal DNA of host plasmid
Origin of replication (ori) - site for DNA replication that allows plasmids to replicate independently from host chromosome
copy # → # of plasmids in the cells
Multiple Cloning Site (MCS) - recognition sites for several restriction enzymes in which an insert is cloned into
Selectable marker genes- are allowed to select for transformed colonies
RNA polymerase promoter sequences - used for transcriptions in vitro and in vivo
DNA sequencing primers
Table of vectors etc.
What are the types of vectors?
Bacterial plasmid vectors - can clone inserts that are smaller than 7kb (express eukaryotic proteins from genes poorly)
Bacteriophage vectors
Cosmid vectors
Expression vectors
Bacterial Artificial Chromosomes (BAC)
Yeast Artificial Chromosomes (YAC)
Ti vector
What is important about bacteriophage vectors?
they are advatageous, because they can clone up to 25kb (lambda genome) that is linear and 49kb
Cloned DNA is inserted into restriction sites in center of lambda chromosome
Recombinant chromosomes are packaged into viral particles in vitro
phages then infect a lawn of E. Coli cells
At the end of each lambda are 12bp sites = COS
which base pair together when they infect bacteria and circularize and replicate
Obtain plaques that are zones of dead bacteria which contain millions of recombinant phage particles
What is the visual of Bacteriophage Vectors:
What do cosmid vectors contain?
COS ends of lambda DNA
Plasmid origin of replication
Gene for antibiotic resistance
DNA is cloned into restriction site
Cosmids are packaged into viral particles and used to infect E coli cells @ a low copy number
*Advantage clone fragments between 20 - 40 kb
What is important about bacterial expression vecotrs?
can allow high levels of protein expression in bacterial cells because they have a prokaryotic promter sites nect the the MCS
Bacterial RNA polymerase can bind to the promoter and transcribe the insert’s sequence which is then translated into protein
Protein is then purified → biochem technique
*Disadvantage: bacterial ribosomes can’t translate a eukaryote sequence or protein isn’t folded correctly → bacteria fon’t have oragenelles for processing. to use thi system w/ eulaytoie tenomic DNA
What is important about bacterial artificial chromosomes?
can obtain large low copy plasmids
contains genes that encode the F factor (unit of genes controlling bacterial replication)
accept large sizes of DNA inserts ranging from 100 - 300 kb
Used during human genome project → clone and sequence large pieces of chromosomes
What is important to yeast artificial chromosomes?
smaller version of eukaryotic chromosomes contains an ori or rep
two telomeres, selectable markers, centromere
allows the replication of YAC & segregation of daughter cells
best option for cloning very large DNA from 200 kb to 2 megabases
used for the human genome project
small plasmids grown grow in E. Coli & introduced to yeast cells (S. crevisiae)
What is important about TI vectors?
are naturally occurring plasmids that are isolated from the bacterium that is in soil plant pathogens → causes diseases in plants
When bacteria infects plant cells, T DNA from the Ti plasmid inserts into the host chromosome
T DNA codes for auxin hormones that weakens plant cell wall, an infected plant divides and enlarges to form a tumor (gall)
Scientists can use Ti vectors to deliver gene to plants by removing toxic gene for auxin
What is the main information of creating DNA libraries?
will be collections of cloned DNA fragments from a particular organisms contained within bacteria or viruses as the host
Screens to pick out different genes of interest
What are the two types of libraries?
Genomic DNA libraries
Complementary DNA libraries (cDNA libraries)
Visual of Genomic library preparation:
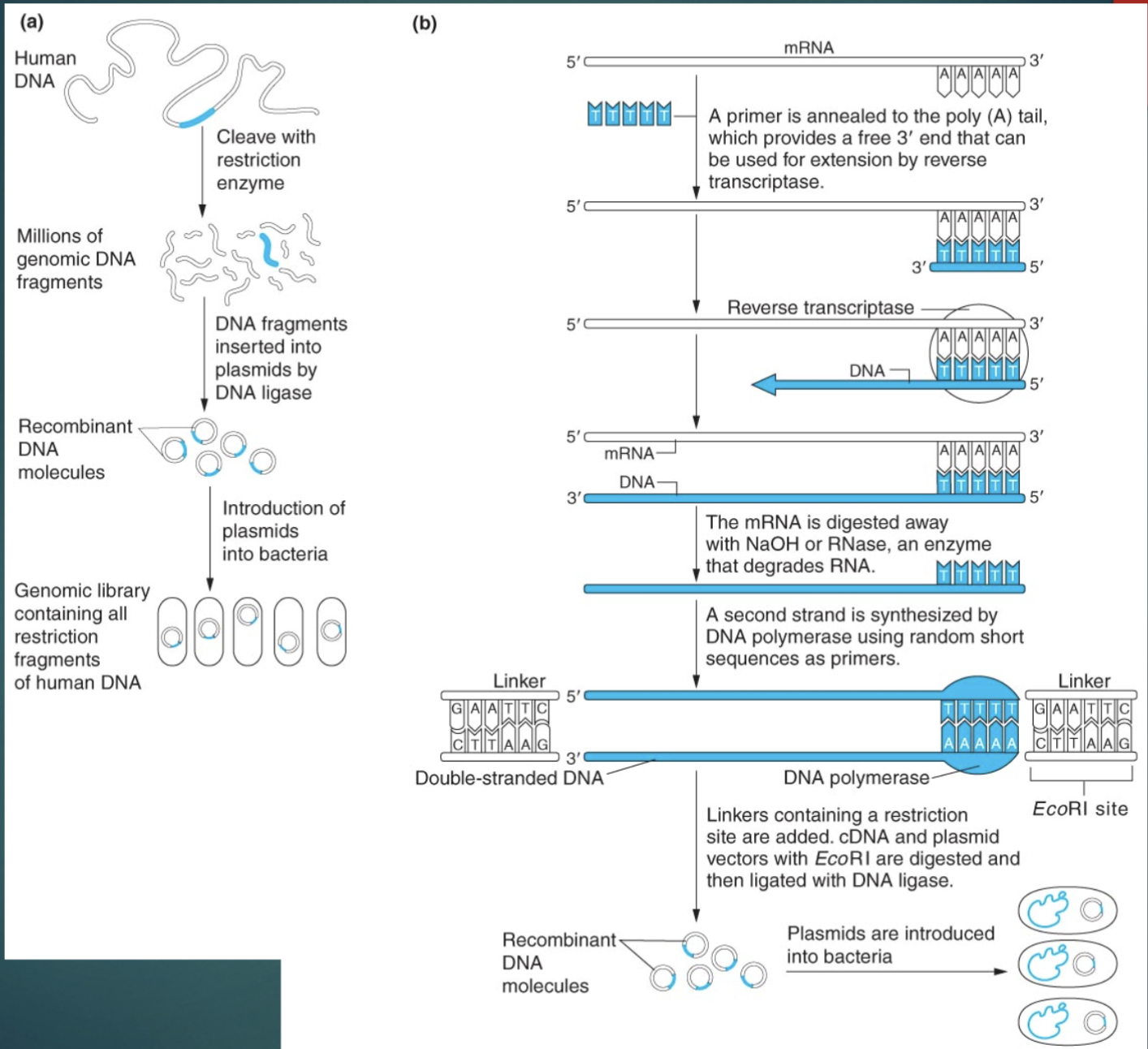
/What are some of the important components of genomic libraries?
chromosomal DNA from tissues of interests are isolated and digested w/ restriction enzyme which produces many fragments that include the entire genome
vectors are digested w/ the same enzyme
DNA ligase is used to ligate genomic DNA fragments and vector DNA
Recombinant vectors are used to transform bacteria and theoretically each bacteria will contain a recombinant plasmid
What are some disadvantages of genomic libraries?
introns are cloned in addition to exons
majority of genomic DNA contains introns in eukaryotes
majority of the library will contain non-coding pieces of DNA
many organisms have large genomes
searching gene of interests becomes difficult
time consuming
What are the important components of cDNA libraries?
mRNA from tissue is isolated
How to make ds DNA from mRNA
using reverse transcriptase catalyzes synthesis of complementary ss DNA from mRNA (complementary cuz of copy of mRNA)
mRNA is degraded either w/ enzyme or alkaline solutions
DNA pol is used to synthesize second strand of DNA to create ds cDNA
Short linkers ds DNA sequences contain restriction enzyme recognition sites that are added to the ends of the cDNA
Cut it with restriction enzyme, cut vector w/ same enzyme, ligate fragments to create recombinant vectors
Transforms bacteria w/ recombinant vector
What are some advantages of genomic libraries (cDNA)?
collection of actively expressed genes in the cells or tissues from which the mRNA was isolated
introns are not cloned
can be created and screened to isolate genes that are primarily expressed only under certain condition in a tissue
SCENARIO: Assume that a gene involved in increased muscle mass is
expressed when the muscle cells are exposed to growth
hormone. What would be the source of the cDNA library:
muscle cells or muscle exposed to growth hormone? Work in
groups to explain your answer.
DISADVANTAGE:
can be difficult to make the cDNA library if a source tissue with an abundant amount of mRNA for the gene is not available.
Genomic Libraries:
can library screen to identify the gene of interests
Colony hybridization
bacterial colonies contain recombinant DNA that are grown on an agar plate
either nylon or nitrocellulose filters are places over the plate and some of the bacterial colonies stick to the filter at the exact location they were on the plate
Treat filter w/ alkaline solution to lyse the cells and denature the DNA
Denatured DNA binds to the fitler as a ss DNA
Filter is then incubated w/ a probe that is tagged w/ a radioactive nucleotide or fluorescent dye
DNA fragments are complementary to the gene of interest
Probe binds by the hydrogen bonding to complementary sequences on the filter = hybridization
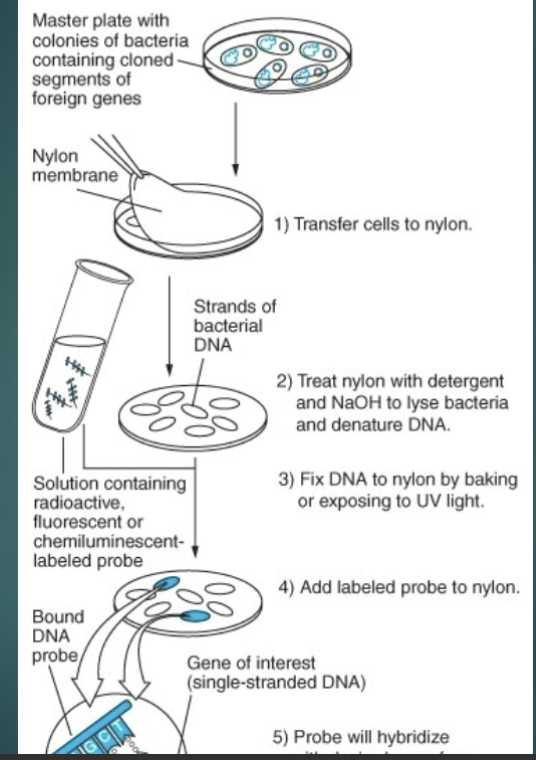
Go Back to probe section of lecture 3 to hear breakdow (Visual)
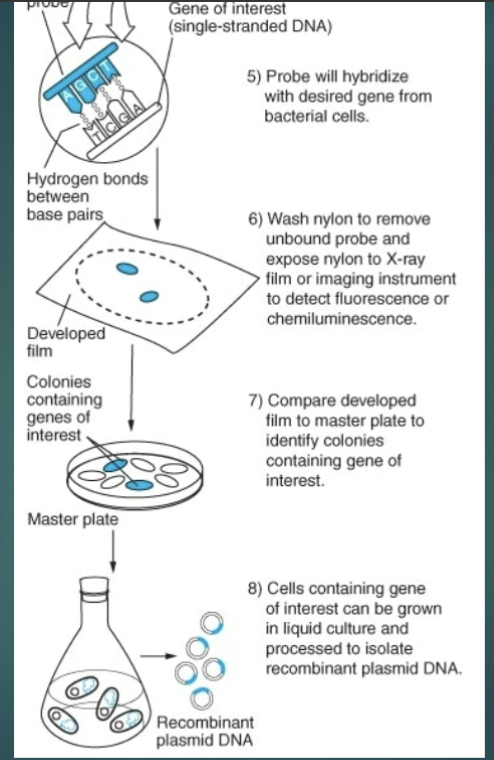
Colony Hybridization:
the type of probe that is used depends on what is already known about the gene of interest
Ex. using a mouse or rate probe to screen a human library because many genes between these species are similar
IF gene sequences has NOT been cloned in another species that something is kown about the protein, what can be done?
Library screening rarely results in the cloning of the full-length gene
Usually get small pieces of the gene; pieces are sequences and scientists look for overlapping sequences
Look for start and stop codon to know when the full length of the gene is obtained
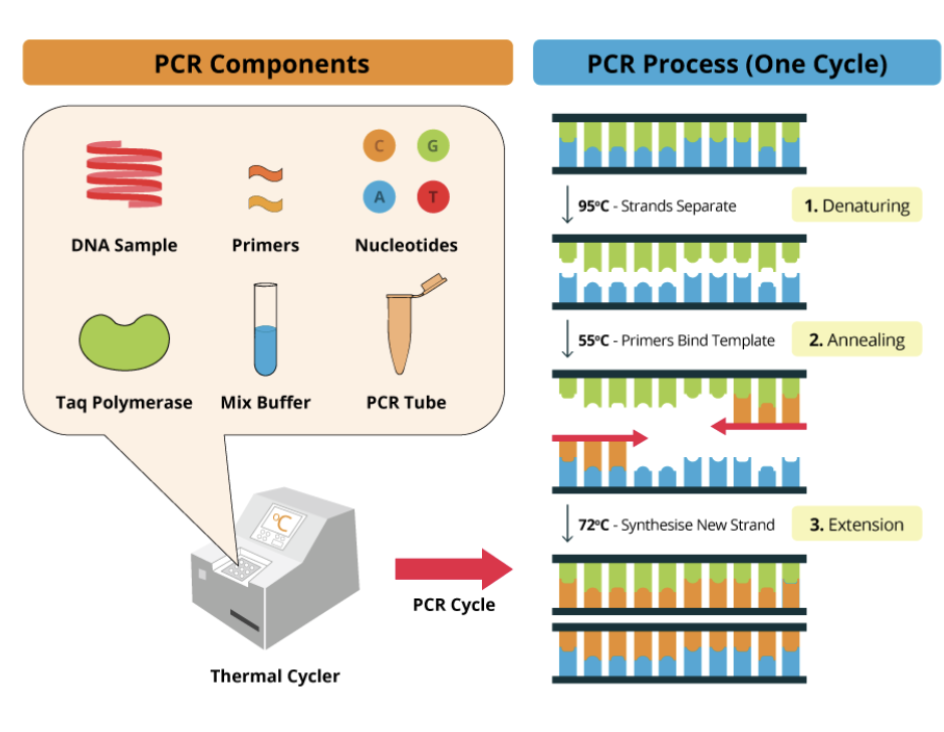
What is PCR?
polymerase chain reaction
developed mid 1980s → Kary Mullis
technique for making copies, amplifying, a specific sequences of DNA in a short period of time.
Good to know:
is a combination of tertiary and quartenary structure
is hydrophobic
contains alpha helices (configurations) and beta sheet amino acid sequences.
What is the overall process of PCR?
Target DNA is amplified to be added to a tube, mixed w/ nucleotides (dATP, dGTP, dTTP), buffer, and DNA polymerase
Paired set of forward and reverse primers are added short single stranded DNA oligonucleotides (20-30bp long)
primers are complementary to nucleotides flanking opposite ends of target DNA
Reactin tube is places in a thermocycler → to run reaction
The thermocycler will take DNA through a series of reactions called a PCR cycle
Each cycle consists of three stages
1. Denaturation heat to 94 to 96 celsius
2. Annealing (hybridization) - which primers H bond (with complementary bases at the opposite ends of target sequences at 55 celsius to 65 celsius
3. Extension (elongation) - DNA pol copies target DNA at 70 to 75 celsius
at the end of one cycle the amount of DNA has doubled
Cycle is then repeated to 20-30 times
What are the advantages of PCR?
can amplify million of copies of target DNA from short small amount of starting material in short period of tie
1. molecule of DNA will yield 2^n copies
N represents the number of PCR cycles
scenario in lect (can do idk)
Type of DNA pol used → very important
Taq DNA polymerase → is isolated from species known as Themus aquaticus thrives in hot springs
What are some application that PCR is used in?
Making DNA probes
Studying gene expression
Detection of viral and bacterial infections
Diagnosis of genetic conditions
Detection of trace amounts of DNA from tissues found at crime scene
Detection of DNA from fossilized dinosaur tissue
Visual of gene of intersts (230)
In a primer design what should you aim for:
primers should have a GC content between 40 and 60% with the 3’ end of a primer ending in G or C to promote binding.
Having too much GC content means there is a higher hydrogen bonding that can make the primer bind too tightly that can cause a primer-dimer formation (not ideal).
An ideal primer design is around 18-30 bases (aiming for short primers) making this a more efficient bind to the target gene.
The ideal melting temperatures of primers are between 65℃ and 75℃ (within 5 ℃ of each other) because this makes Tm dependent from the base temperature.
232 on ipad on tipes for PCR
How do you calculate T-melt (233- 234 on ipad)
Cloning PCR products:
is rapid and effective compared to using DNA libraries
Disadvantage: need to know something about DNA sequences that flanks the gene of interests to design primers