BIOL1502: Macromolecules, Organelles, Plasma membrane, Metabolism, Cell Communication
1/213
Earn XP
Description and Tags
for first midterm
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
214 Terms
Organic Molecules
molecules based on carbon
4 major classes of organic molecules
lipids, nucleic acids, proteins, carbohydrates
hydrocarbons
moelcules consisting of carbons only linked to hydrogen
functional groups
small reactive groups of atoms that give the molecule its specific properties, participating in biological reactions, linked by covalent bonds to other atoms in the molecule
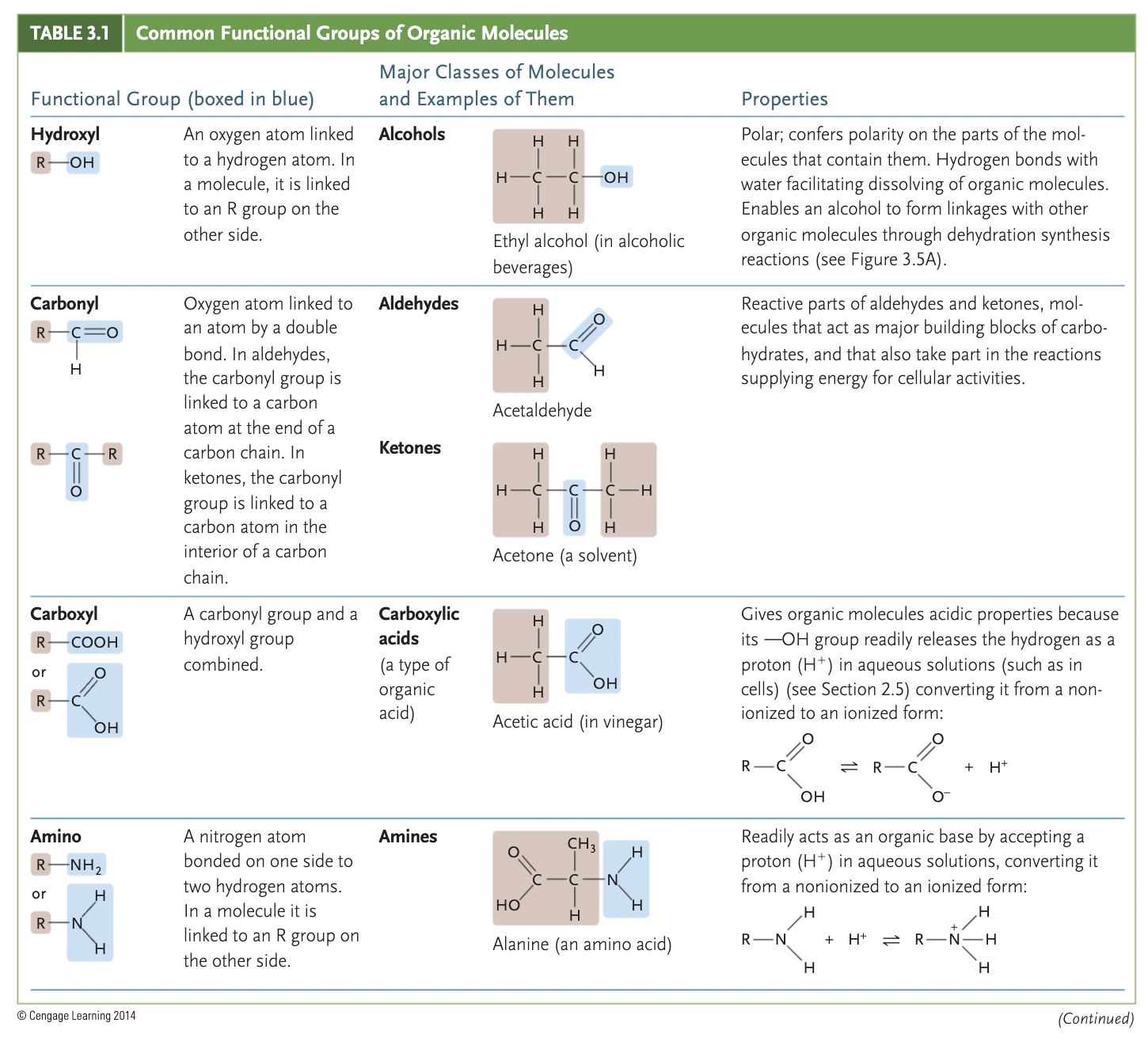
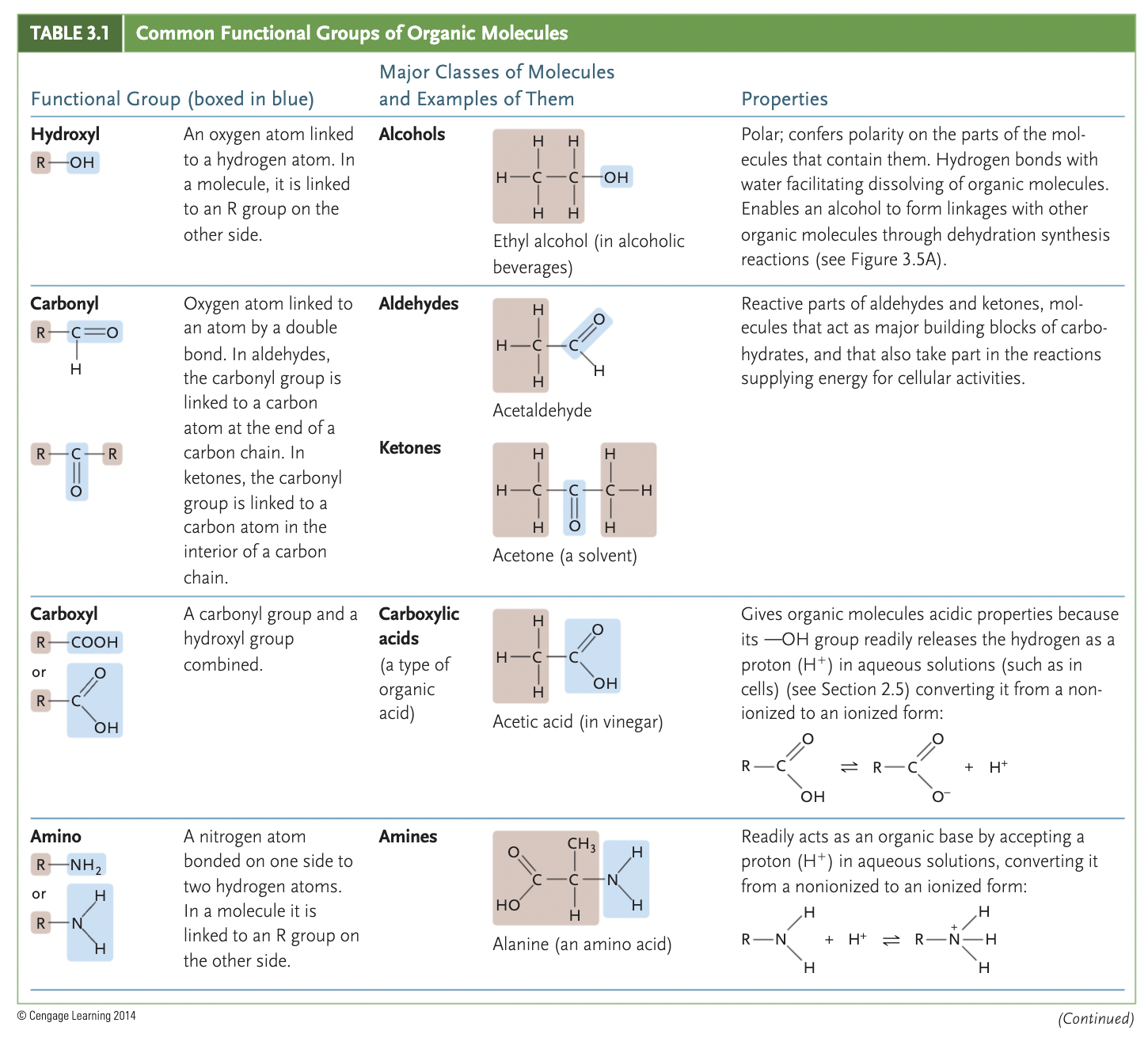
examples of functional groups
amino (-NH2), phosphate (-OPO3²-), hydroxyl (-OH), carbonyl (>C=O), carboxyl(-COOH)
isomers
2+ molecules w/ same chemical formula that have different structures
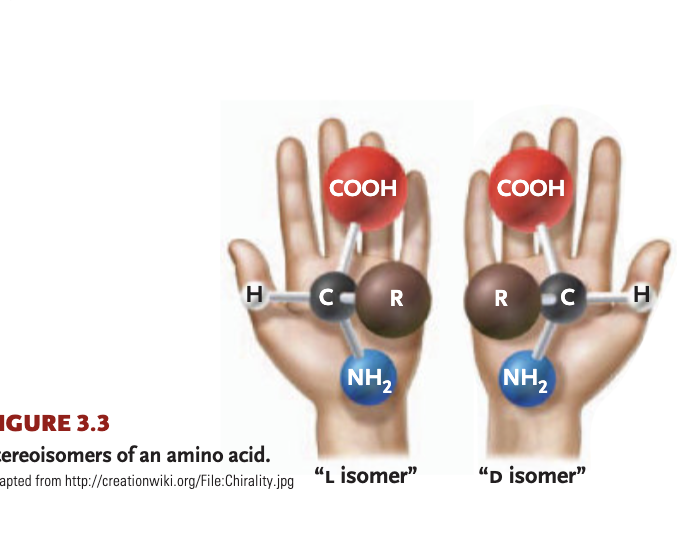
stereoisomers
molecules that are mirror images of each other
asymmetric carbon
1+ carbon atom linking to 4 different atoms/functional groups
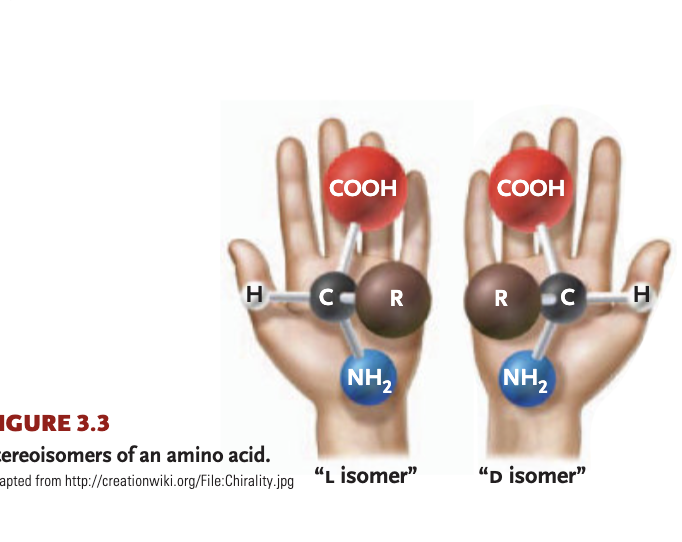
L+D isomers
one form typically enters into cellular reactions more readily, as enzymes best fit one
structural isomers
2 molecules w/ same chemical formula but atoms are connected differently
polymer
a molecule assemble from subunit molecules (monomers) into a chain by covalen bondsp
polymerization
dehydration synthesis reaction that form polymers
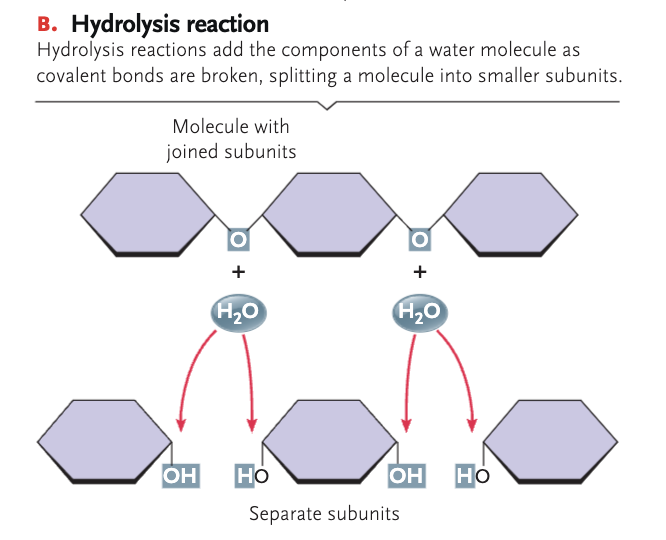
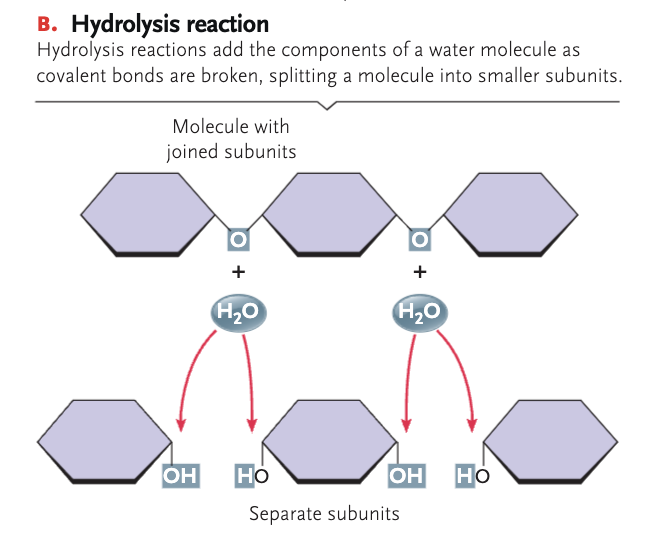
hydrolysis
where water molecules break down chemical compounds into smaller molecules by adding a hydrogen ion H+ and a hydroxyl group OH-
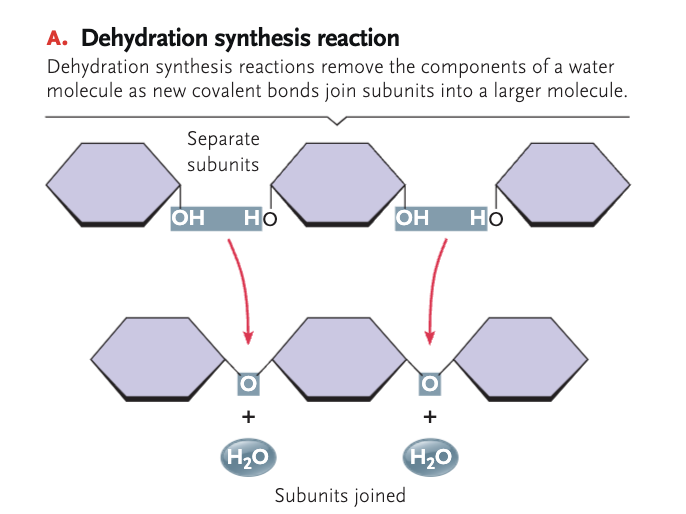
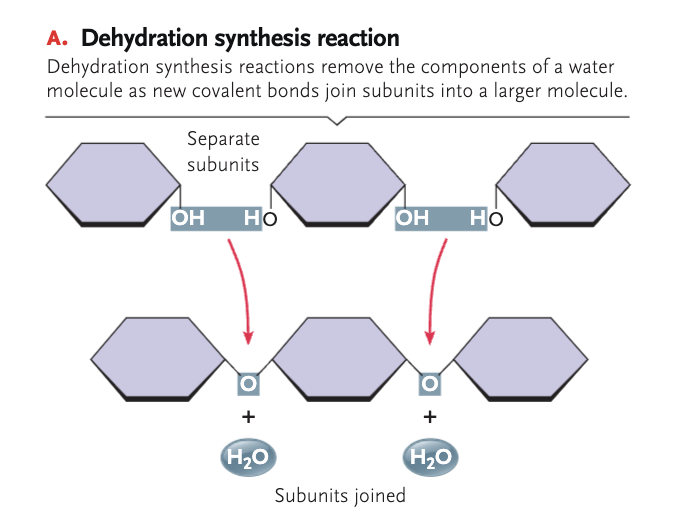
dehydration synthesis
a chemical reaction that involves the loss of an H₂O from the reacting molecule or ion
monomer
a molecule that can react together with other ______ molecules to form a larger polymer chain
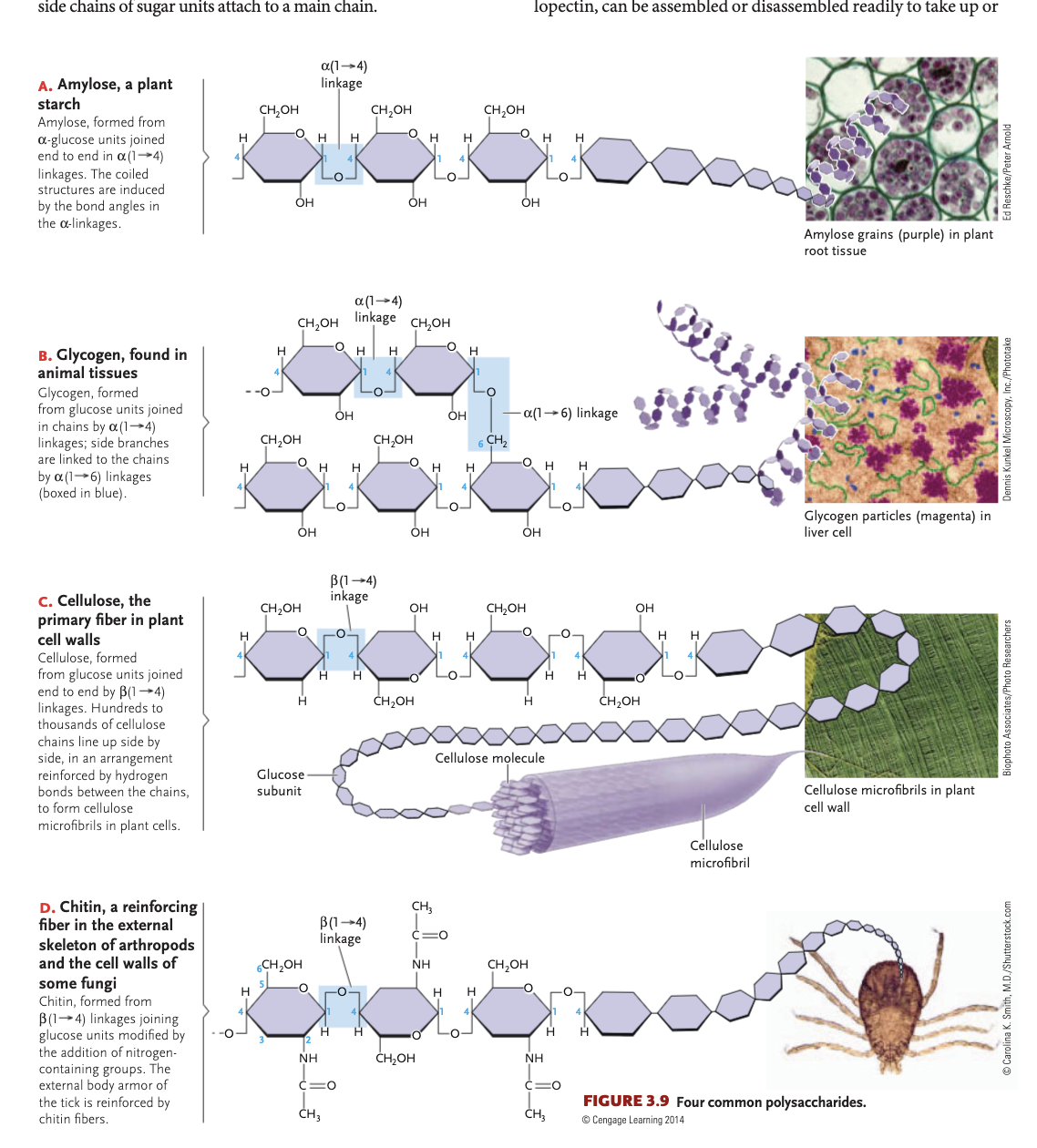
energy providing carbs
starch (plants) glycogen (animals)
carbohydrate monomer
monosaccharides (3-7 carbon atoms)
disaccharide
2 monosaccharides - linked by dehydration synthesis
glycosidic bonds/linkage
bonds between 1’ and 4’ carbon w/ oxygen as a bridge
carbohydrate polymers
polysaccharides: can be a continuous chain, can be branched, can be bonded in different configurations (many different polysaccharide types)
trioses
3 carbons
5 carbons
pentoses
6 carbons
hexoses
monosaccharide form
can occur in the linear or ring form
linear: each carbon in the chain (except 1) will have an -H and -OH attached
ring form: possible with monosaccharides with 4+ carbons, occurs through a reaction between 2 functional groups in the same monosaccharide
favored for stability, allows for switching betwen alpha and beta configuration
ring form occurs with attraction between hydroxyl and carboxyl in linear form, twists around

alpha configuration for monosaccharides
-OH group is pointing below the plane of ring, assembled into starches —> biologically reactive polysaccharides easily digested by animals

beta configuration in monosaccharides
-OH group is pointing above the plane of the ring, makes up cellulose, relatively unreaction and indigestible to animals
Lipids
water insoluble, primarily non-polar, composed mainly out of hydrocarbons, can be stored in cells as an energy source
Neutral lipids
found in cells as energy storage molecules (meaning insoluble, non-polar with no charged groups)
oils and fats
oils are liquid when fats are semisolid at biological temperatures
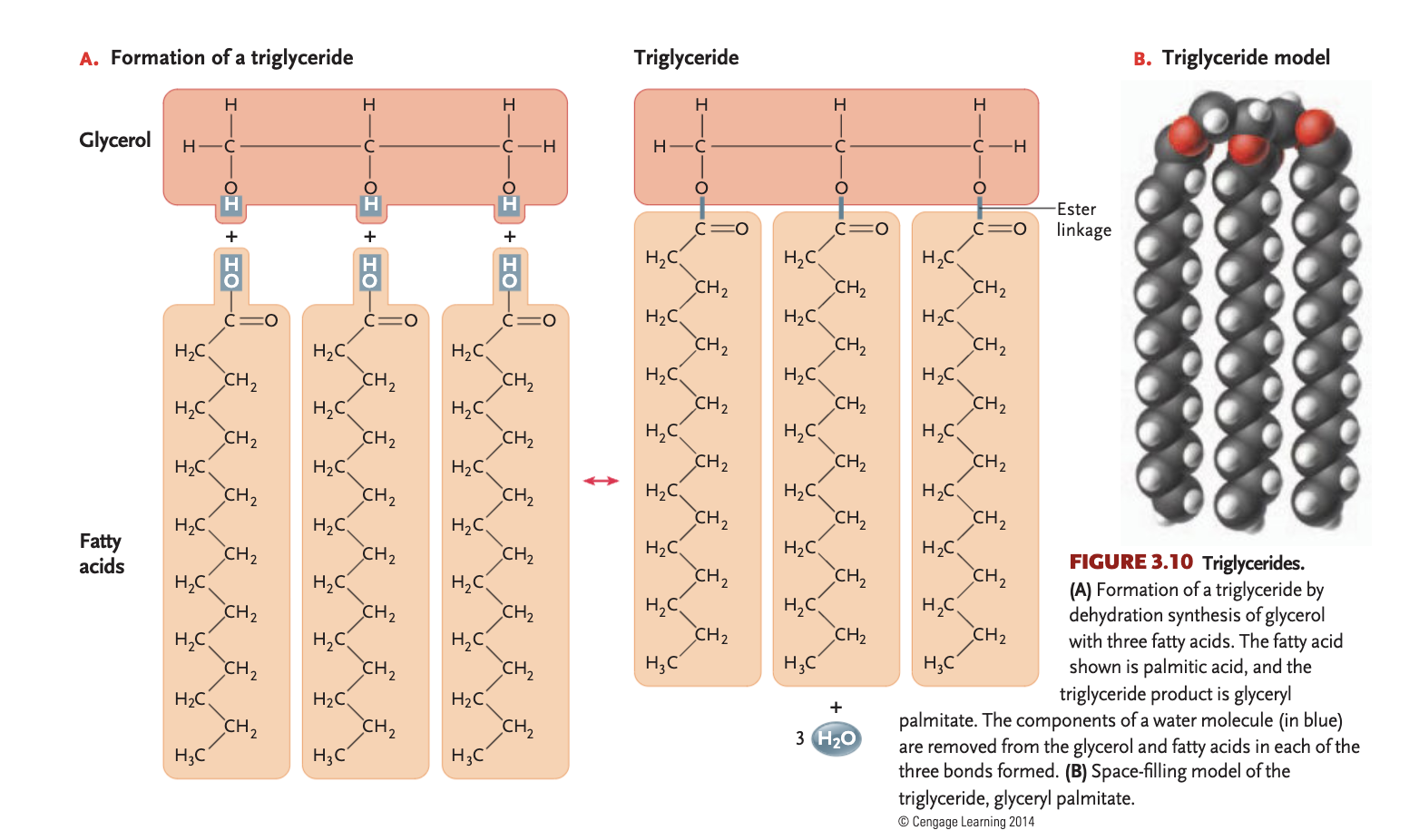
triglyceride
glycerol + 3 fatty acids (glycerol backbone)
synthesized by carboxyl of fatty acid + 1 hydroxyl group of glycerol
amphipathic
molecule having both hydrophobic and hydrophillic parts
glycerol
3 carbon alcohol w/ an -OH attached to each carbon, would be a polar sweet-tasting water soluble molecule
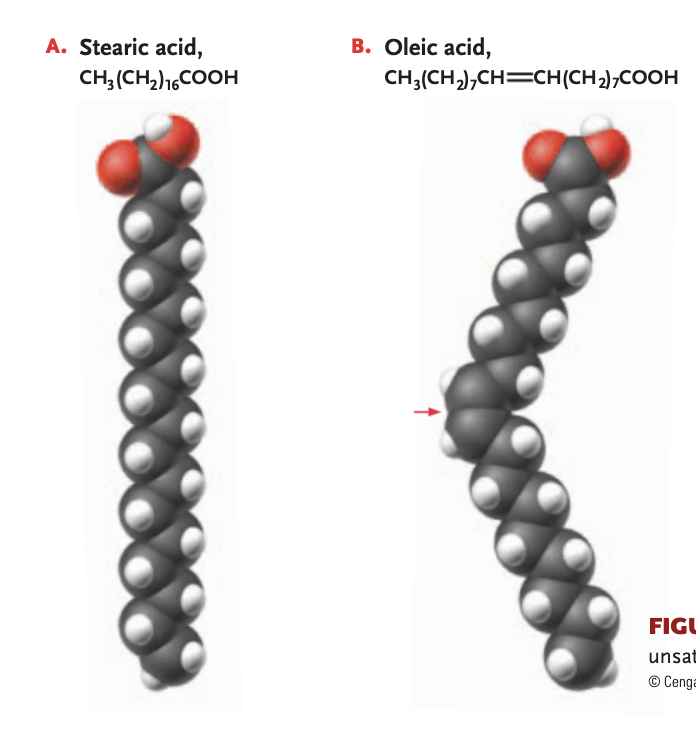
fatty acid
contains a singles hydrocarbon chain with -COOH (carboxyl) at the end
ester linkage
covalent bond between carboxyl (COOH) + hydroxyl (-OH) in lipids, essential for energy storage
formed by dehydration synthesis
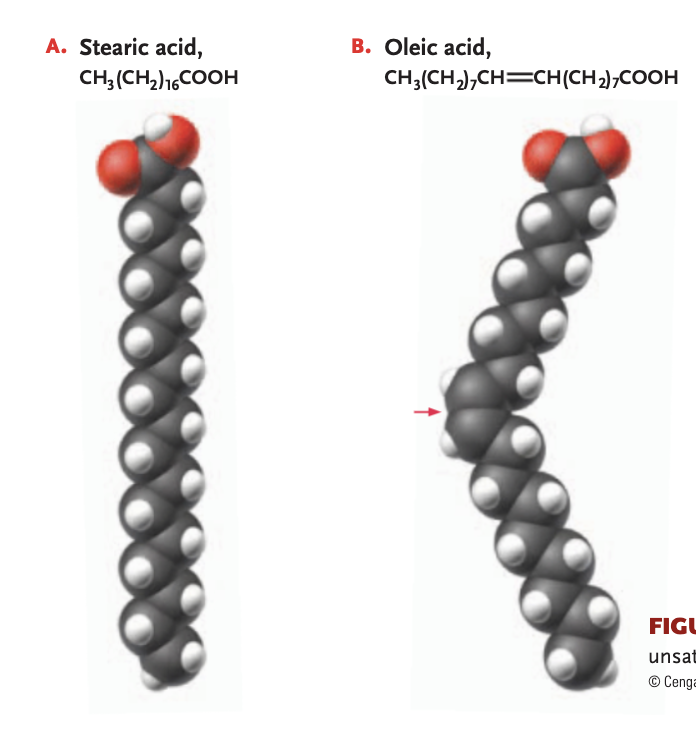
saturated fat
hydrocarbon chain of fatty acid bonds to max # of hydrogen atoms (only single bonds link to carbon atoms)
ethane
can compact closely together at room temperature
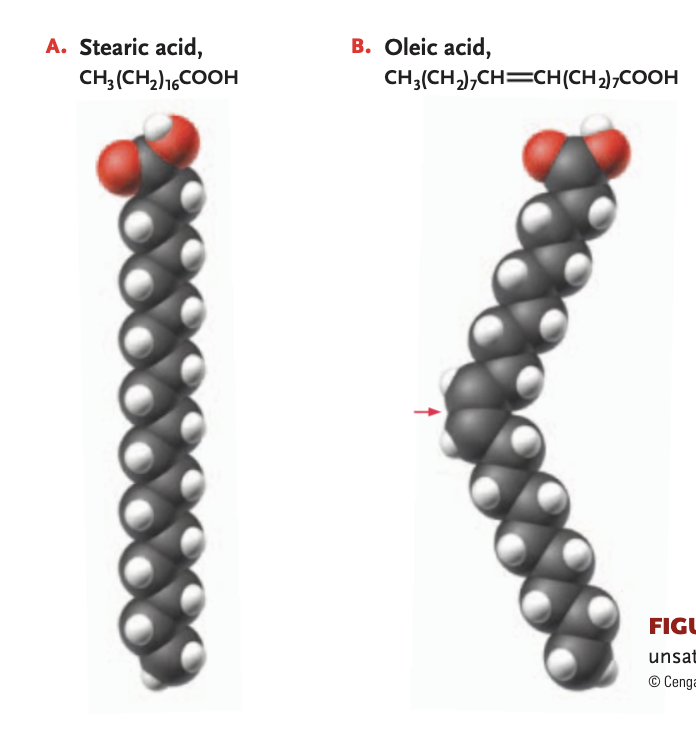
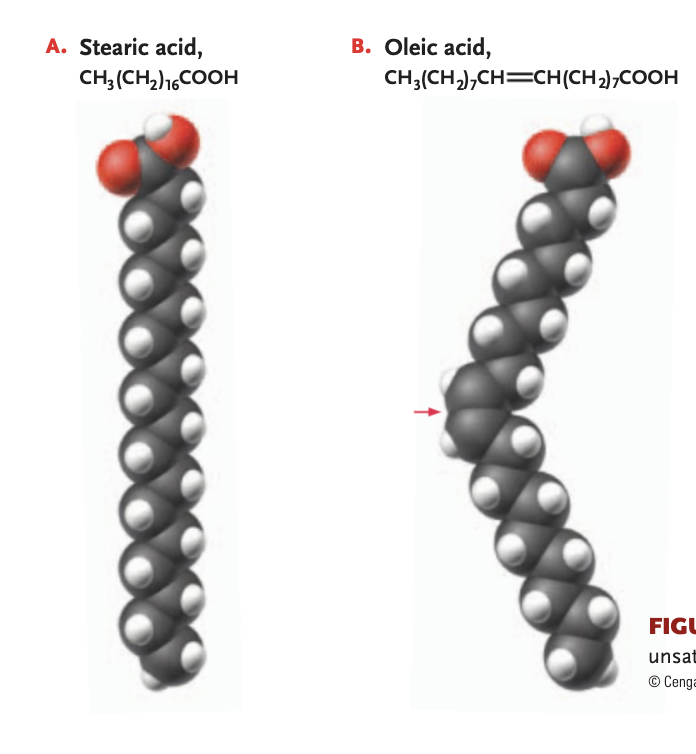
unsaturated fat
1+ double bonds link the carbons (reducing # of hydrogen atoms bonded) —> will have a kink in the chain, cis double bond, will be more liquidy at room temperature
ethylene
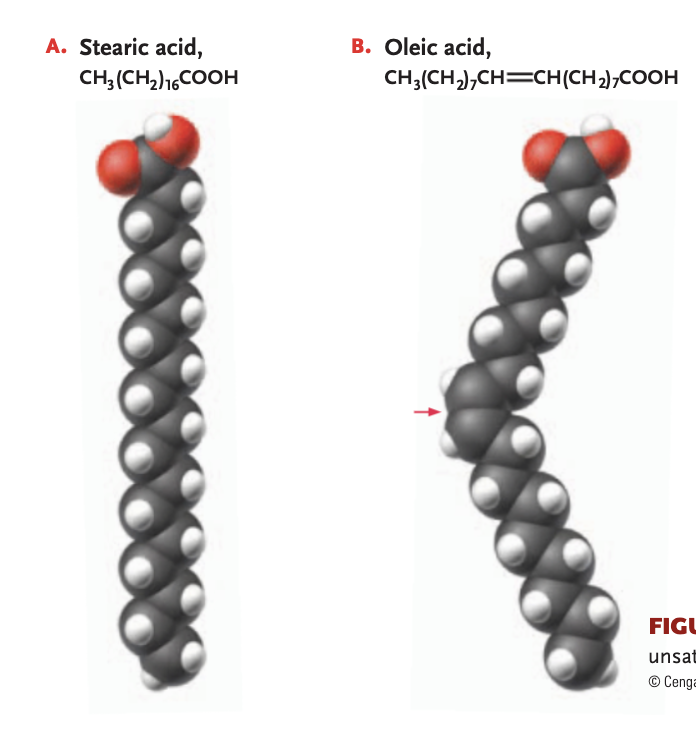
monounsaturated fat
1 double bond linking the carbons
polyunsaturated fat
1+ double bond linking the carbons (can’t pack as tightly at room temp)
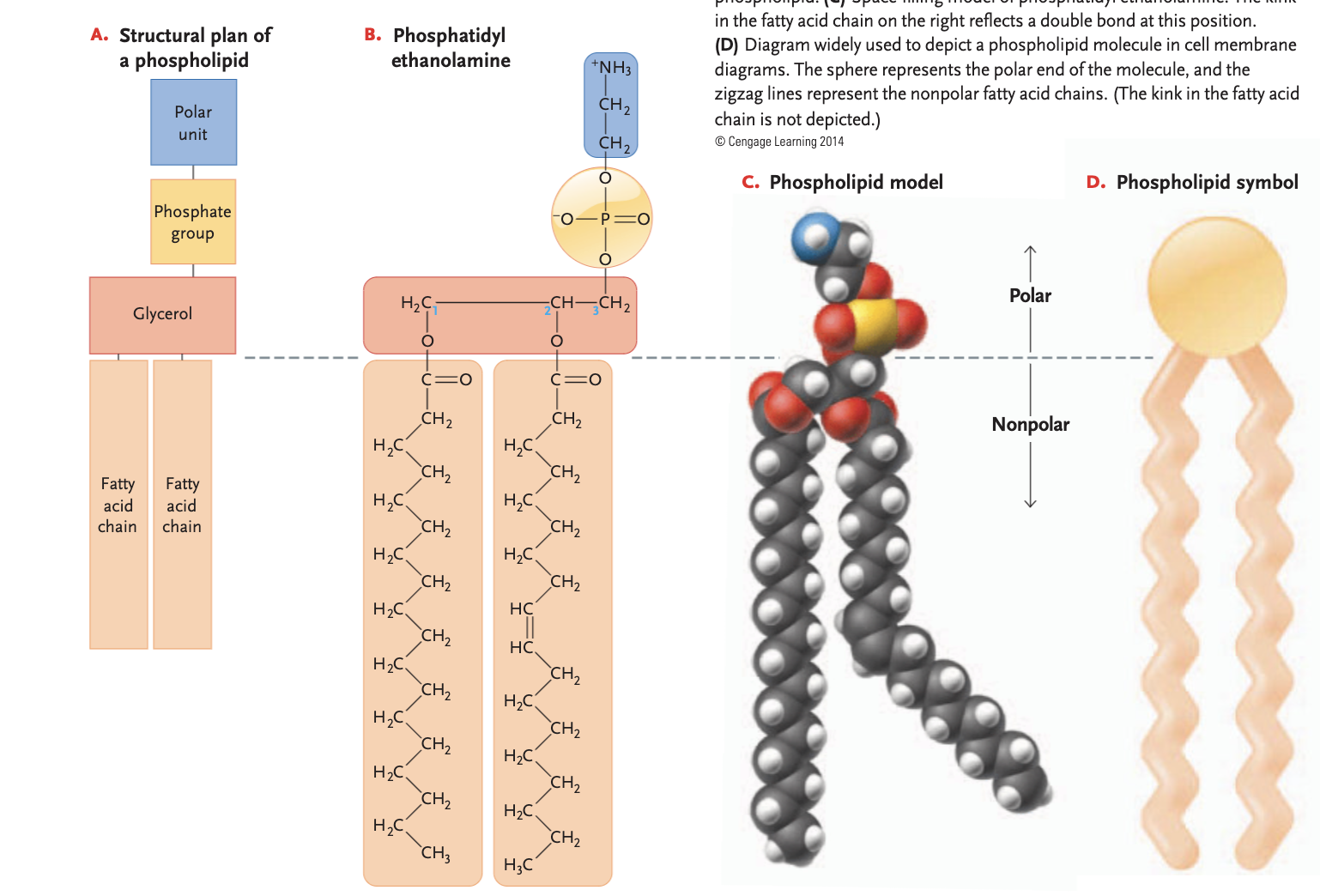
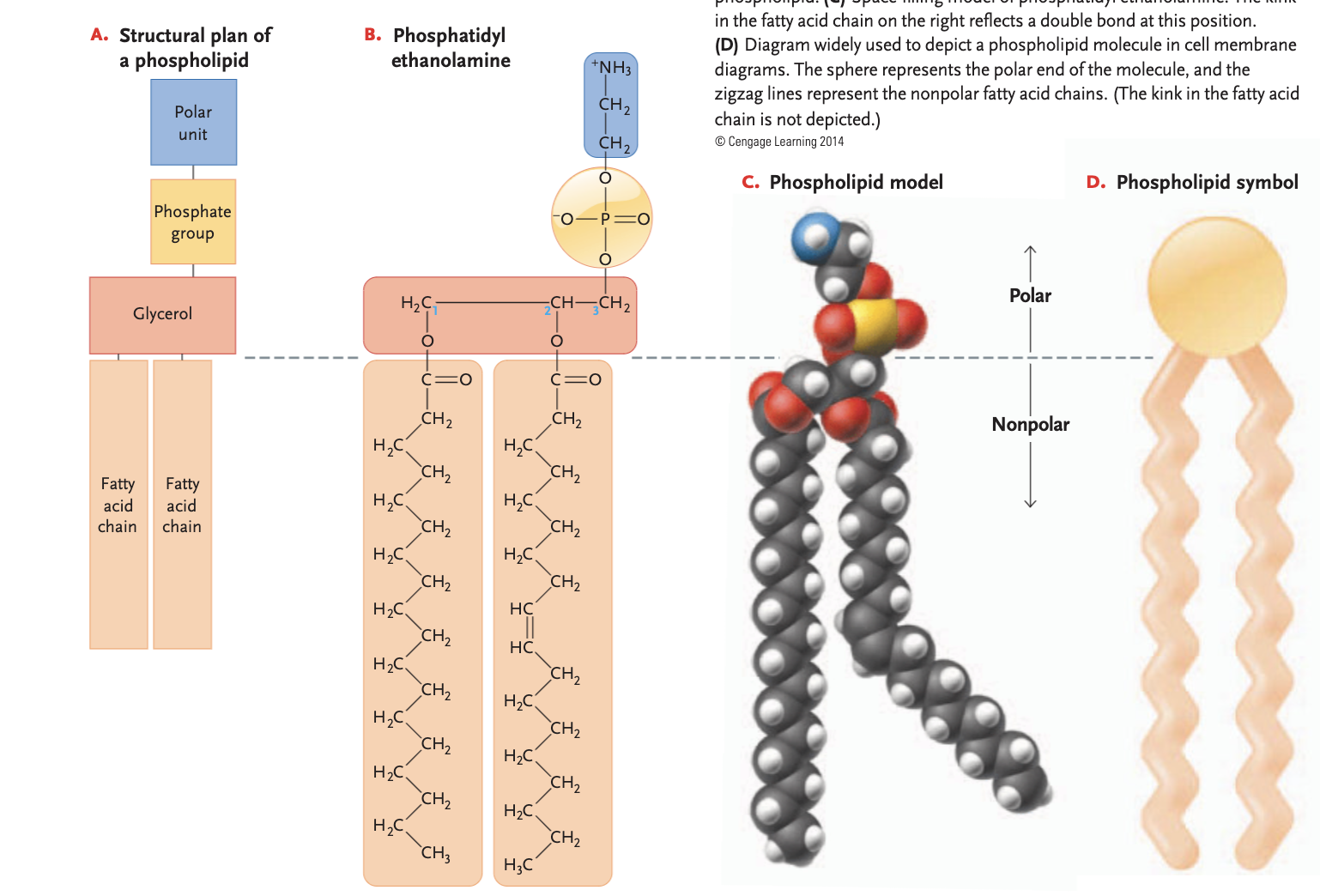
phospholipids
glycerol + 2 fatty acids, 3rd binding site to a polar phosphate group instead of a 3rd fatty acid (bonded to another polar unit)
fatty acid side is non-polar (hydrophobic)
phosphate end is polar (hydrophillic)
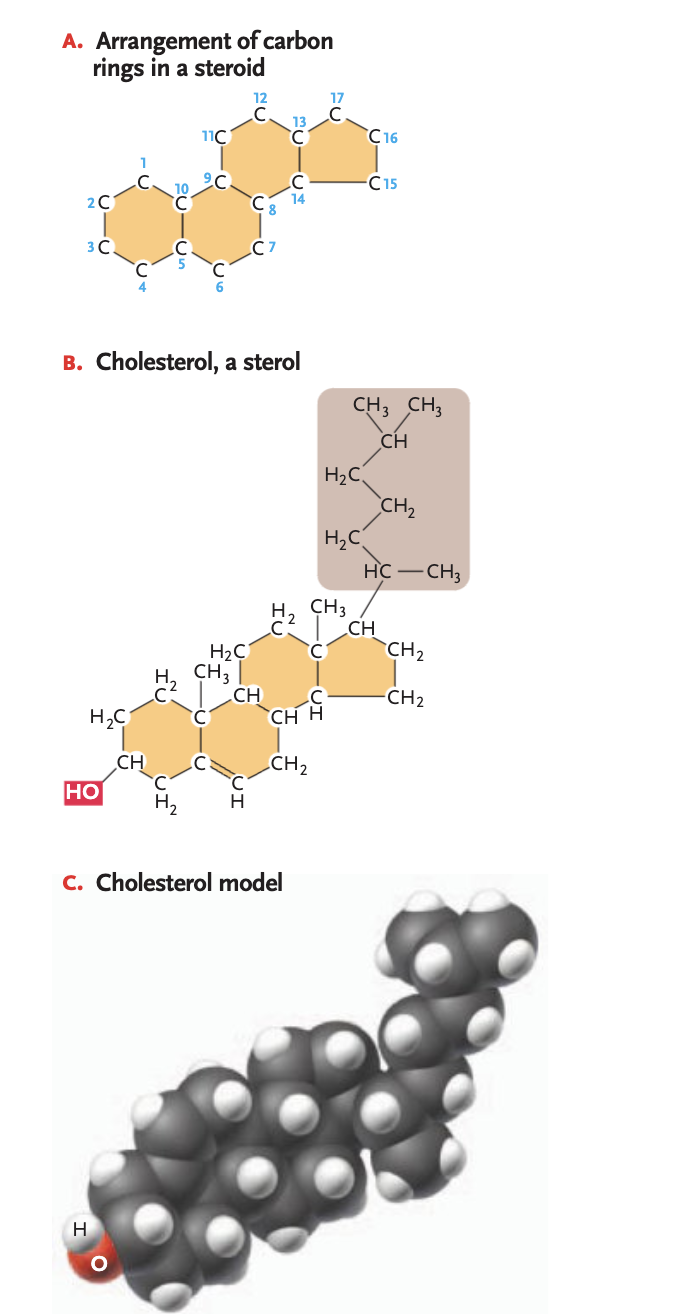
steroids
group of lipids w/ structures based on 4 carbon rings
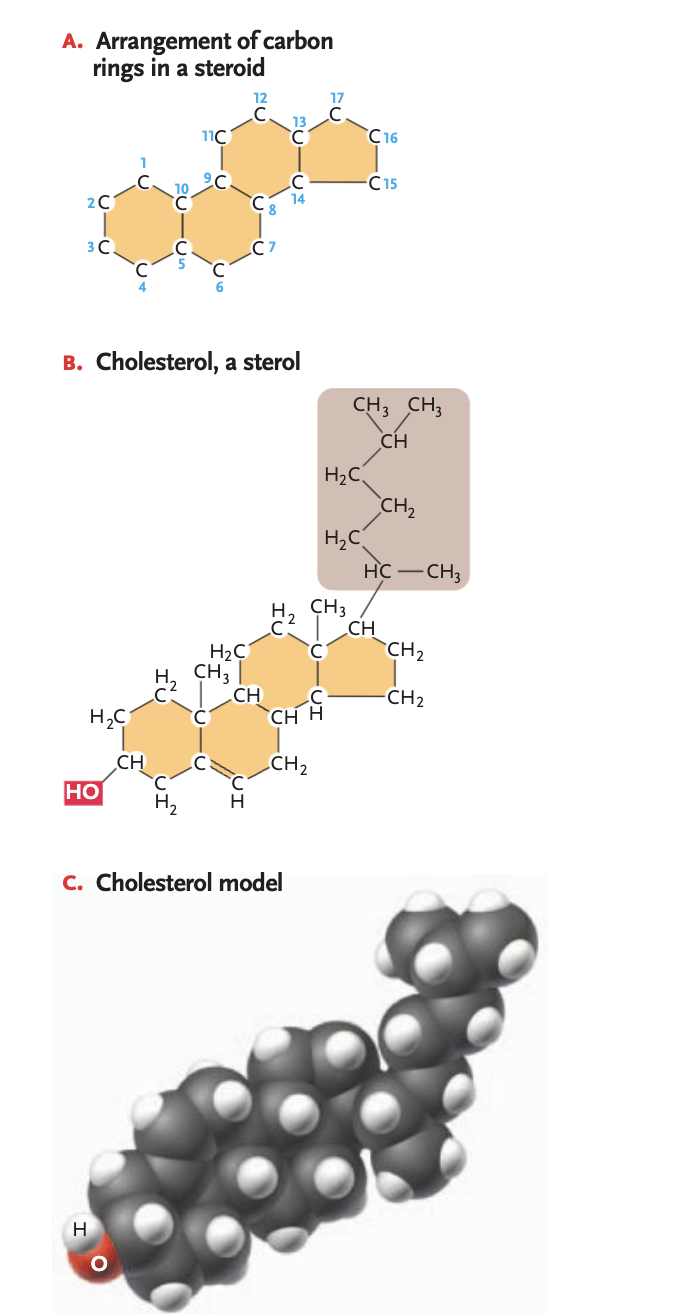
sterols
most abundant steroids, single-OH (polar at one end of a framework, non-polar hydrocarbon cain on the other end (ex. cholesterol)
in the phospholipid bilayer will fit in between phospholipids (another example of why it is so important that the tails are unsaturated —> tails are fluid and won’t pack together
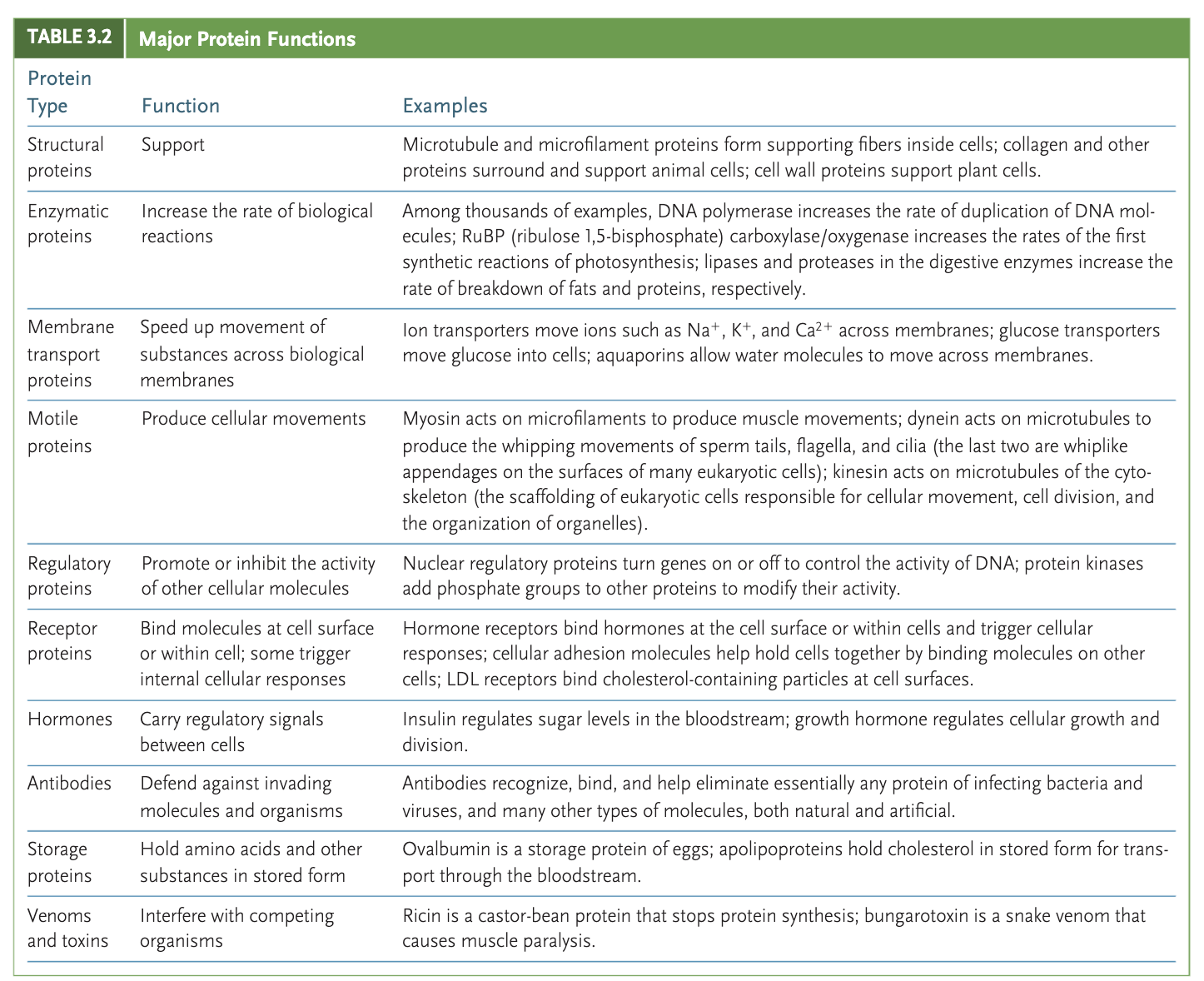
Protein
macromolecule made out of amino acids
ex: enzymes, antibodies (protection against diseases), transport of substances, hormonal proteins, movement, cellular communication through signaling proteins and receptors, regulation of cell processes
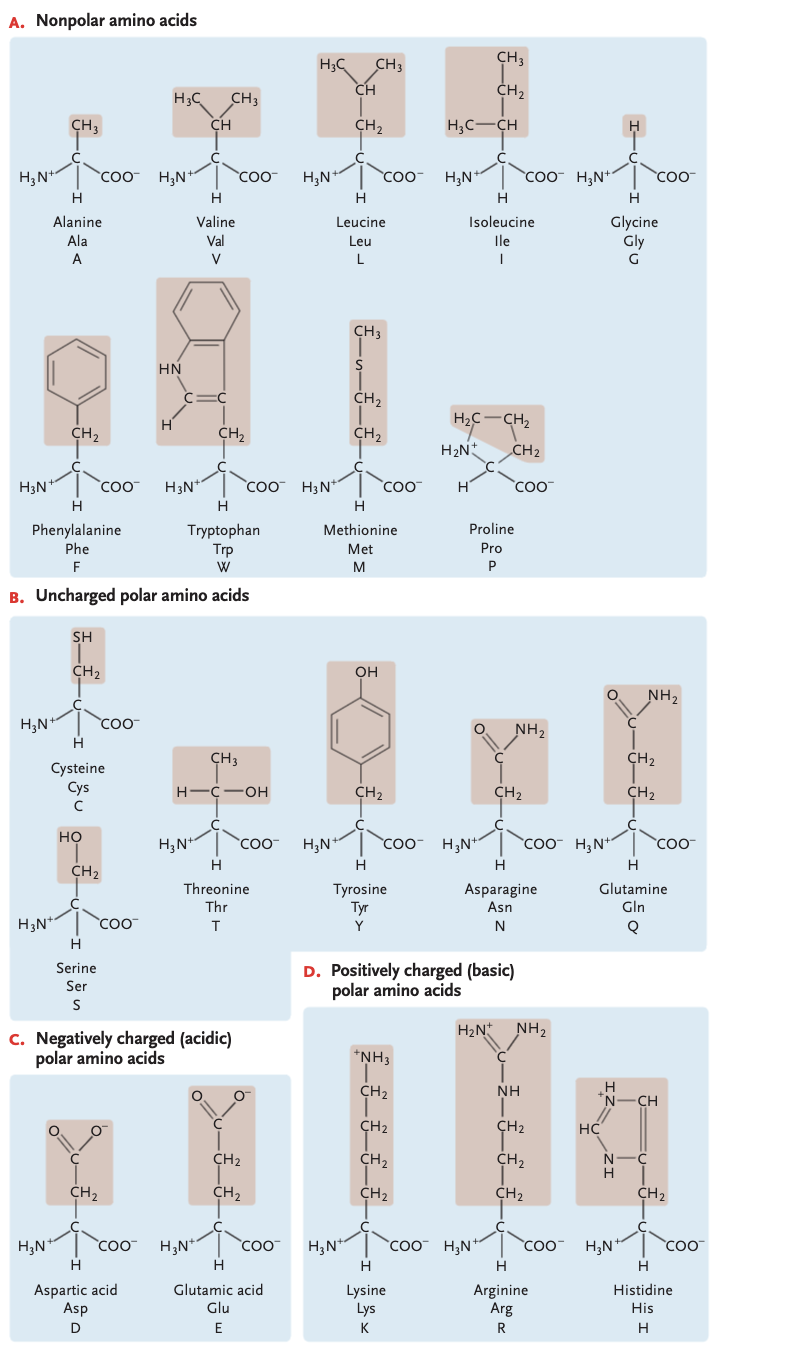
protein monomer
amino acids, charged amino acids can form ionic bonds
peptide bond
formed by dehydration synthesis reaction between amino group of one amino acids + carboxyl group of a second one (protein covalent bonds)
n-terminal end of amino acid
ends of acid w/ NH3+
c-terminal end of amino acid
end of acid with COO-
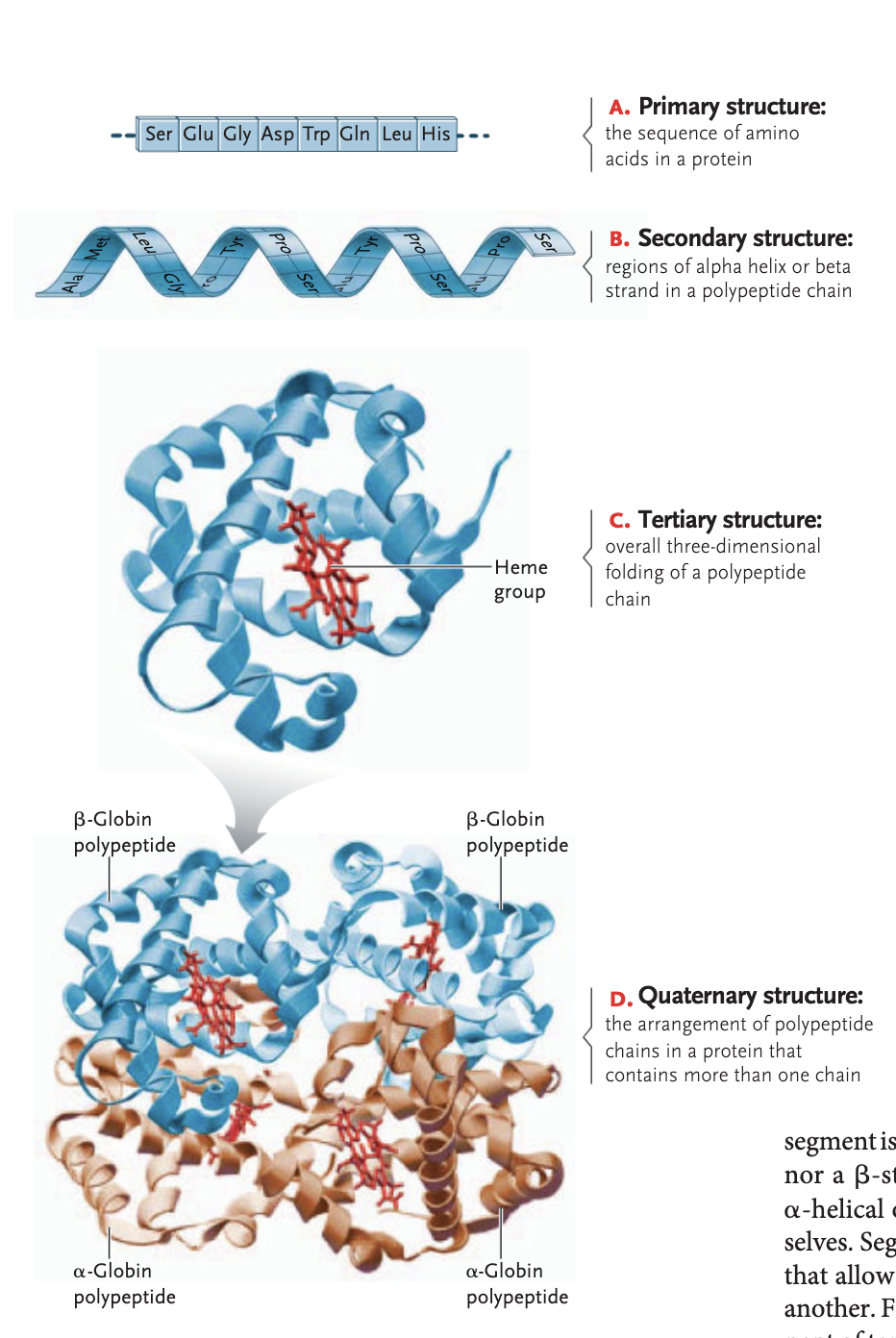
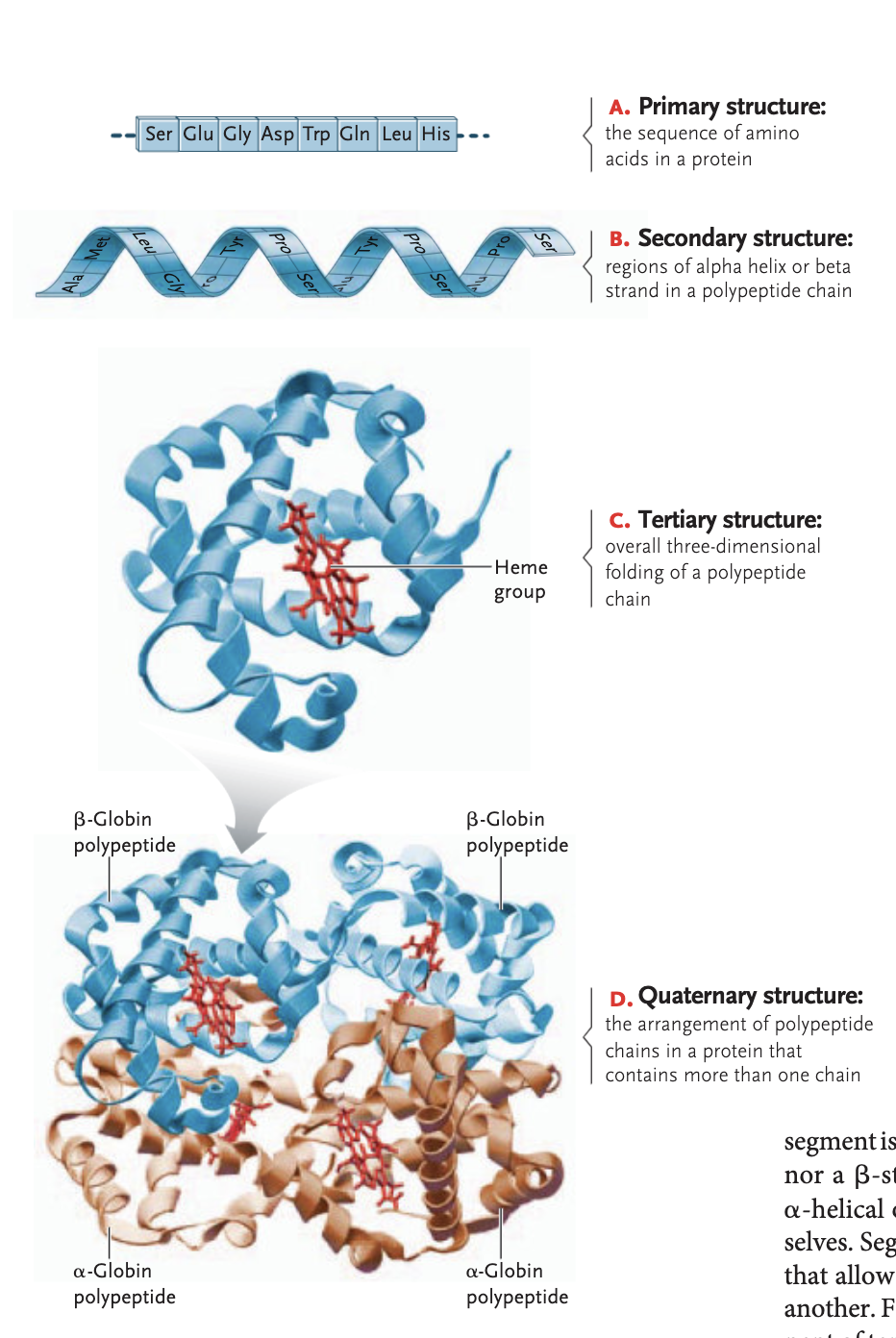
primary structure protein
sequence in which amino acids are linked
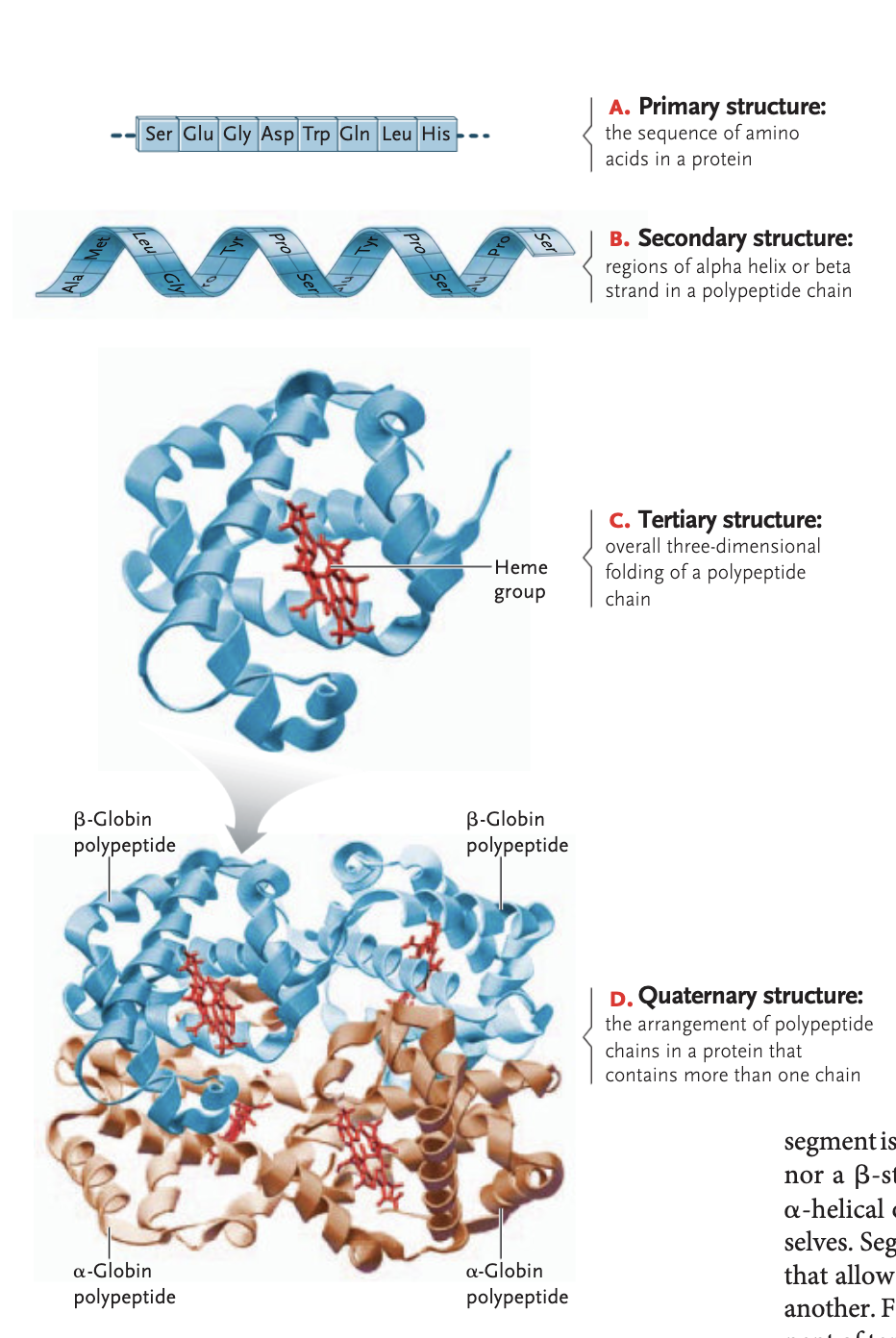
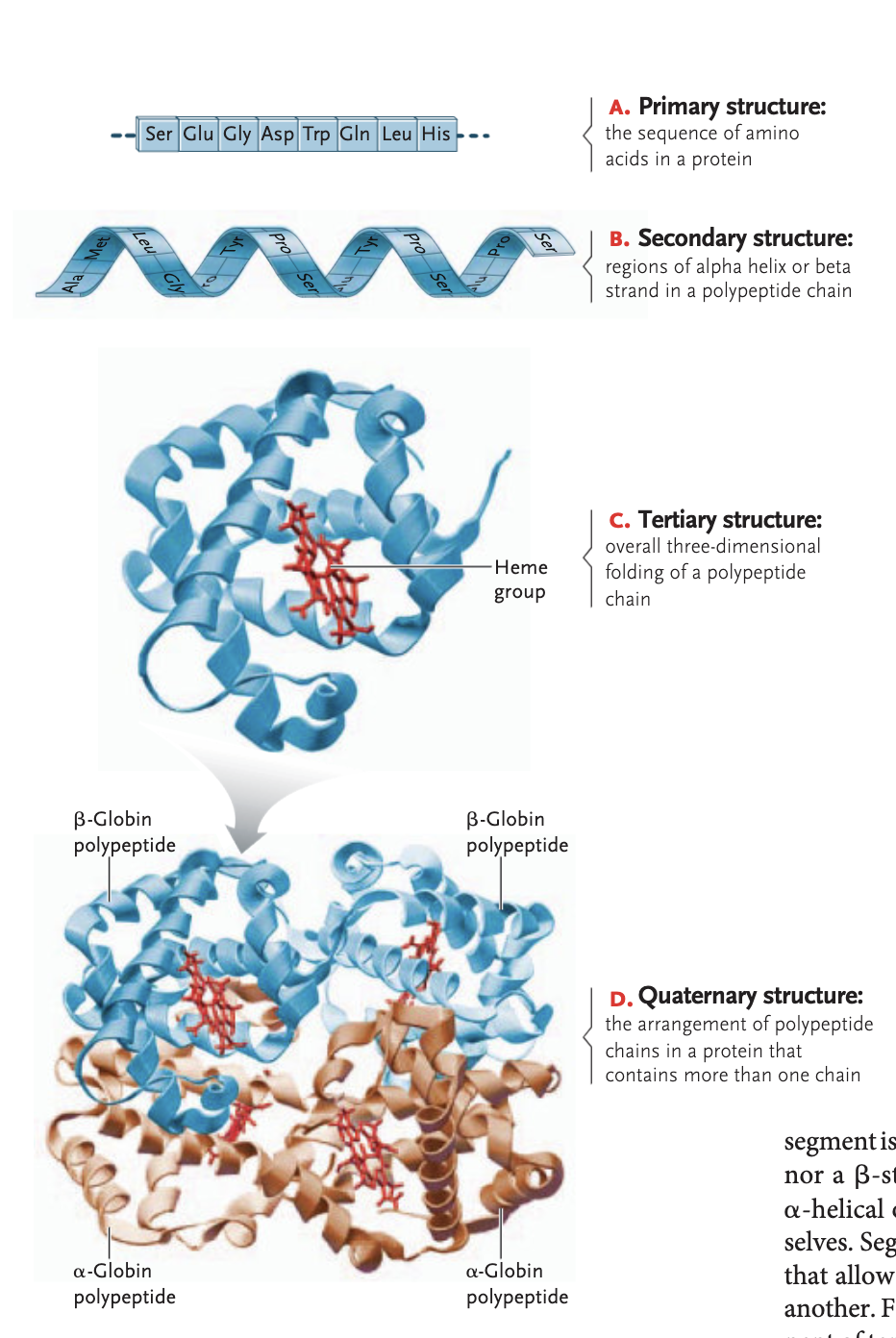
secondary structure of a protein
regions of an alpha helix and beta sheets in a polypeptide sheets formed between hydrogen bonding interactions (alpha helix are rigid coils, beta sheet zigzags on a flat plane)
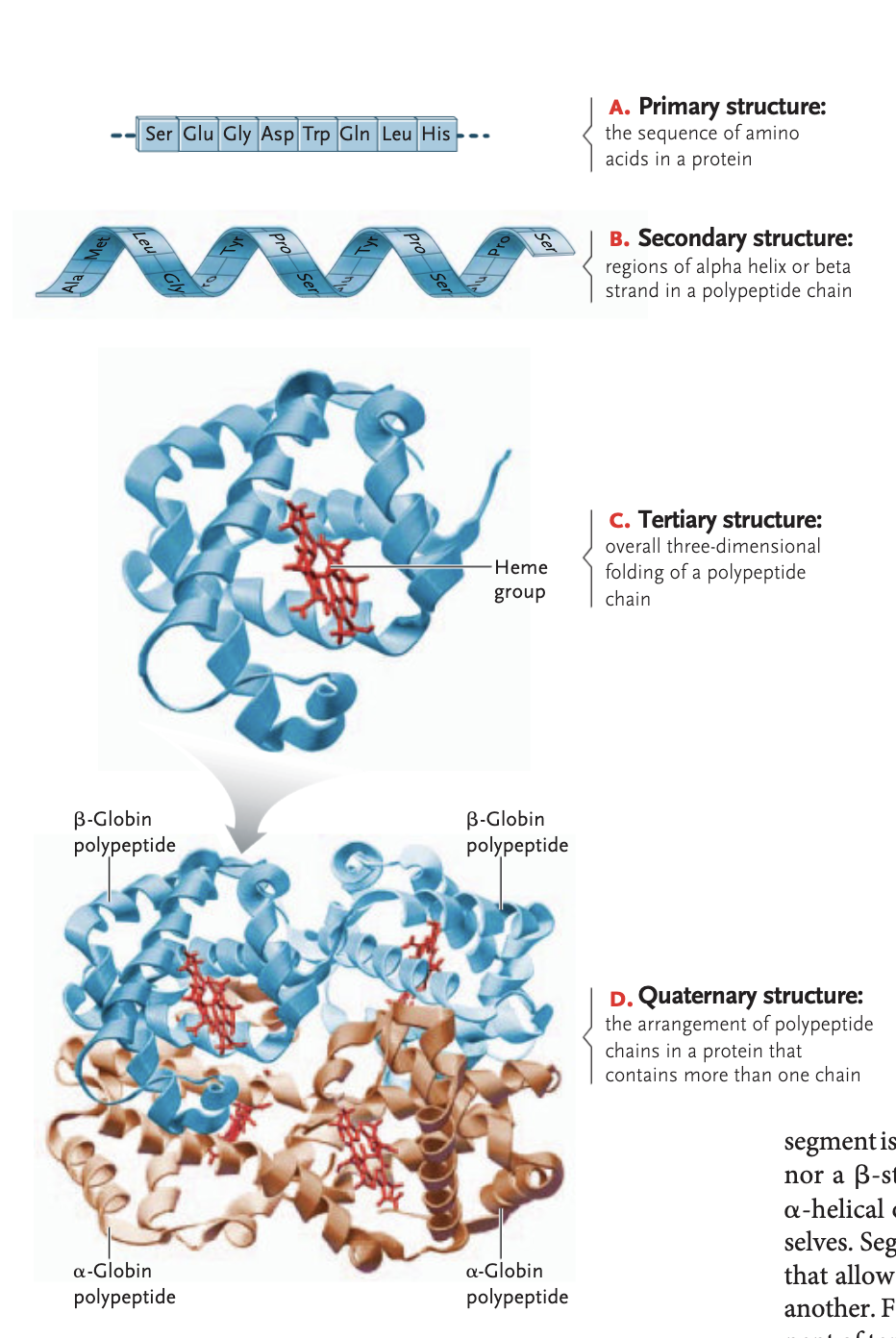
tertiary structure of a protein
overall 3D folding of a polypeptide chain —> attraction between ± and polar/non-polar side groups
stablized through hydrophobi interactions, van der waals interactions, ionic bonds, hydrogen bonds, and disulfide bridges
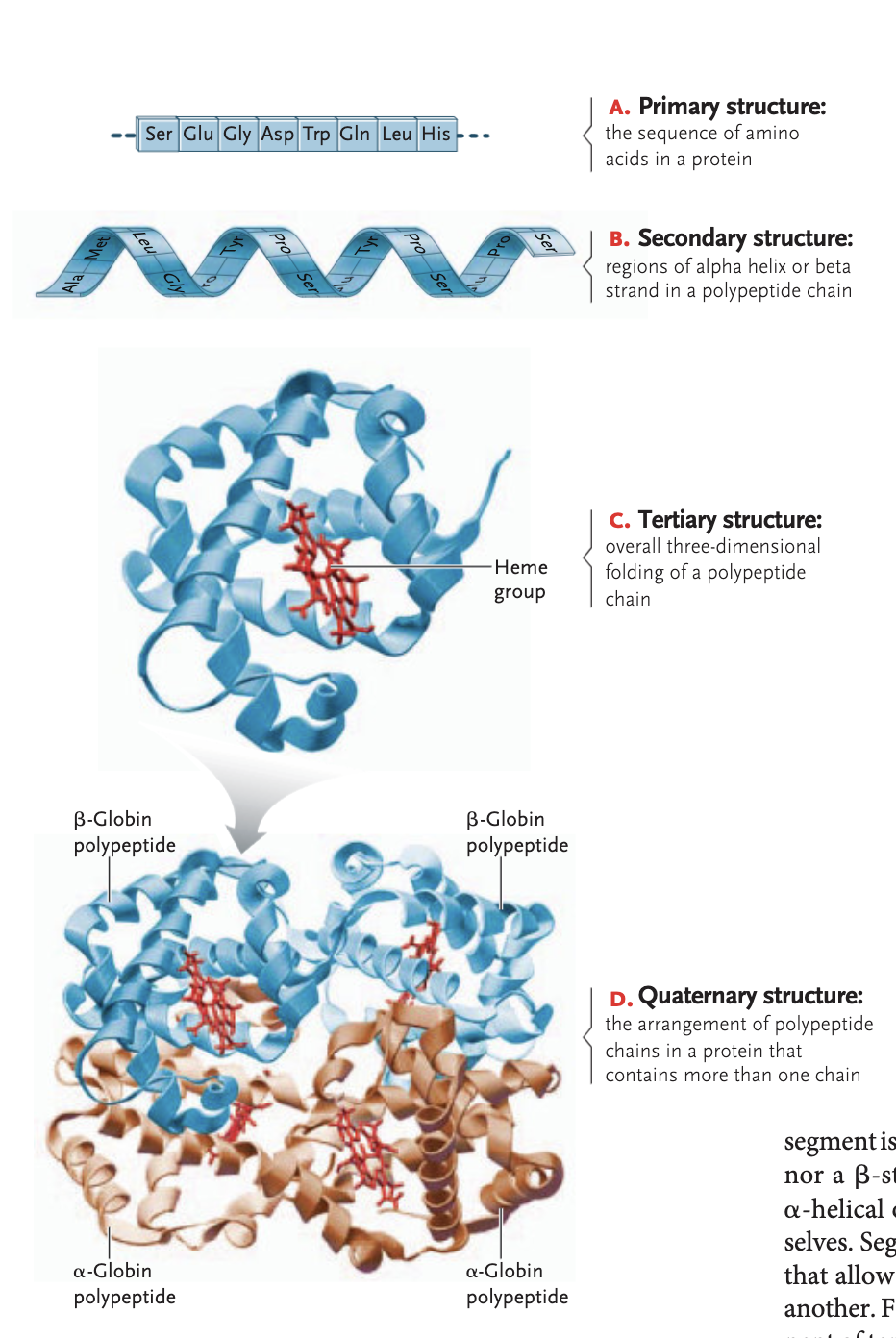
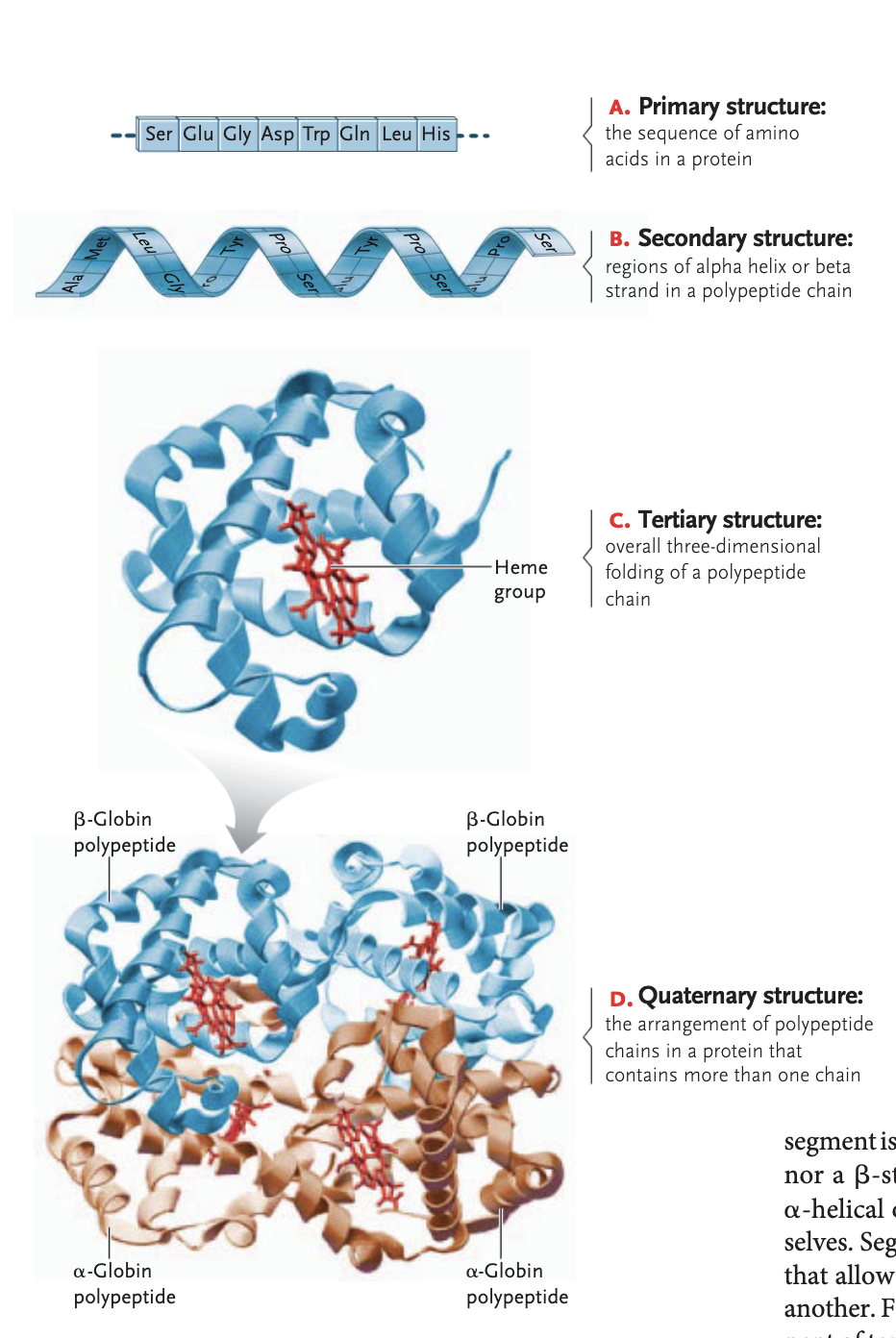
quaternary structure of a protein (optional)
arrangement of a polypetide chains in a protein that contains more than one chain in order to be functional
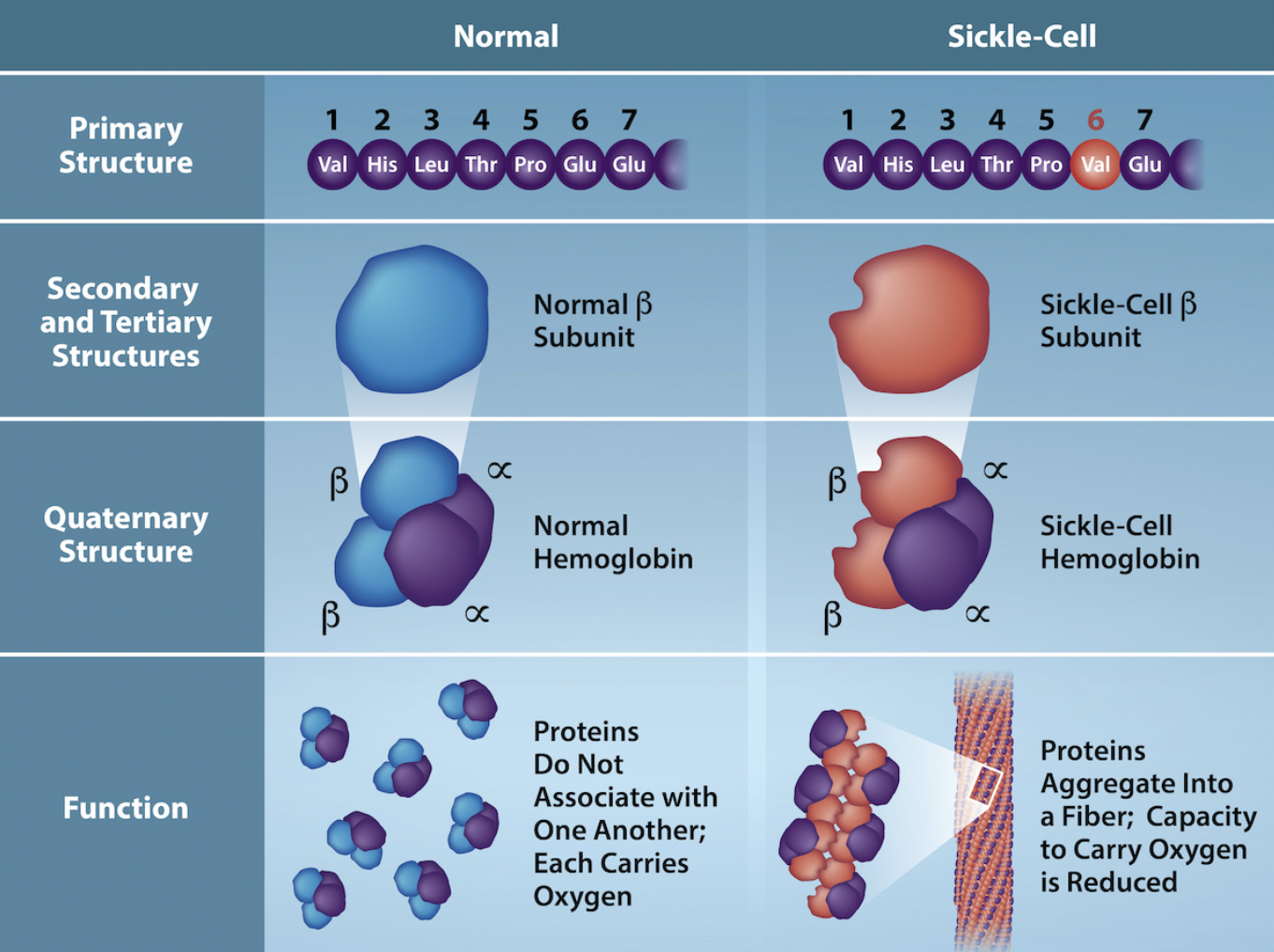
ex hemoglobin, sickle-cell disease: beta subunit of quaternary structure is messed up
conformational change
the alteration of a protein’s or macromolecule's three-dimensional shape, often involving shifts in its tertiary or quaternary structure
protein denaturation
the structural modification, or unfolding, of a ____ 3D shape (2nd, 3rd, 4th) while the primary amino acid chain remains intact (temperature, pH changes0
protein renaturation
the process by which a denatured ___ refolds into its functional, native 3D structure, regaining biological activity
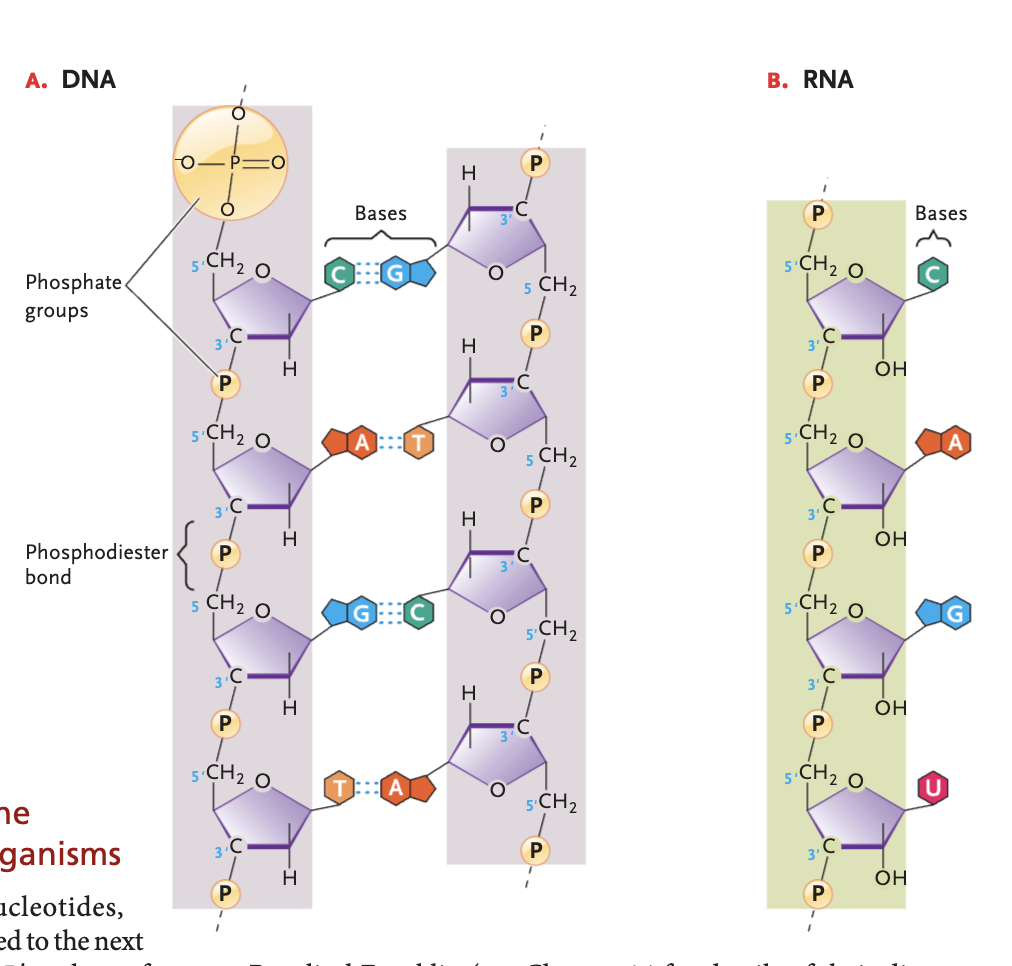
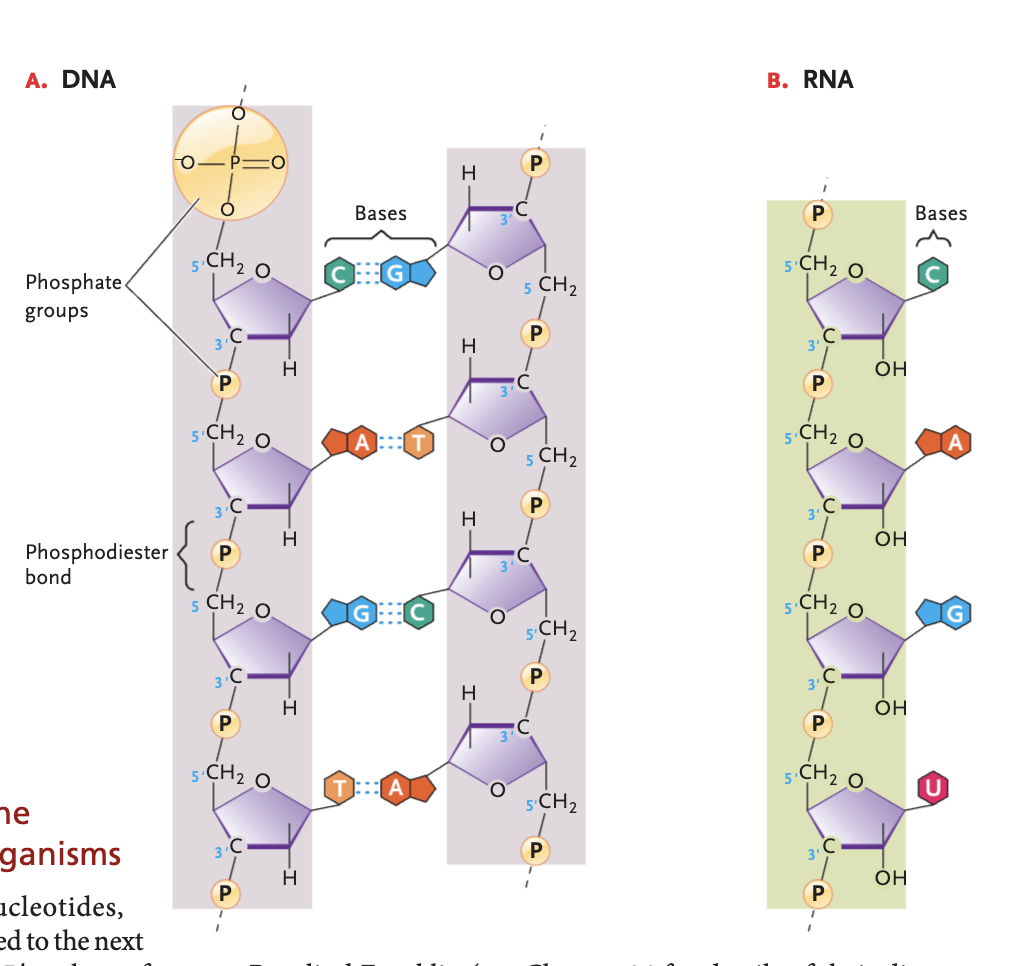
Nucleotide
(monomer of nucleic acids) consist of a nitrogenous base, a 5-carbon sugar, and 1+ phosphate groups (typically 1-3)
nitrogenous base
nitrogen-containing molecule that accepts protons
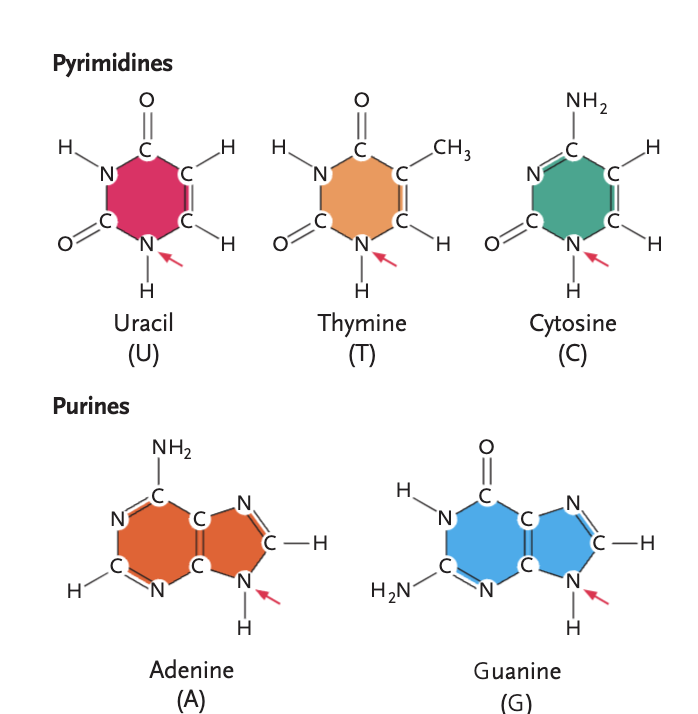
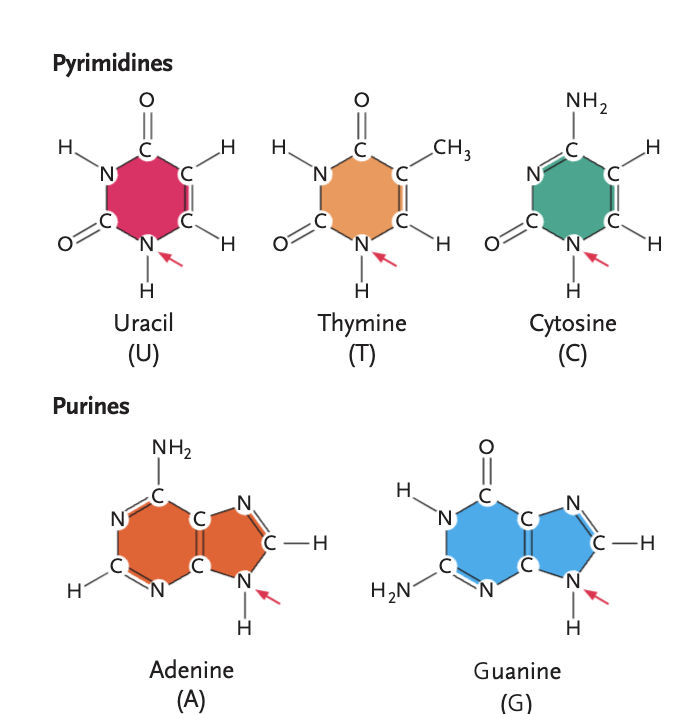
pyrimidines
uracil (U), thymine (T), cytosine (C)
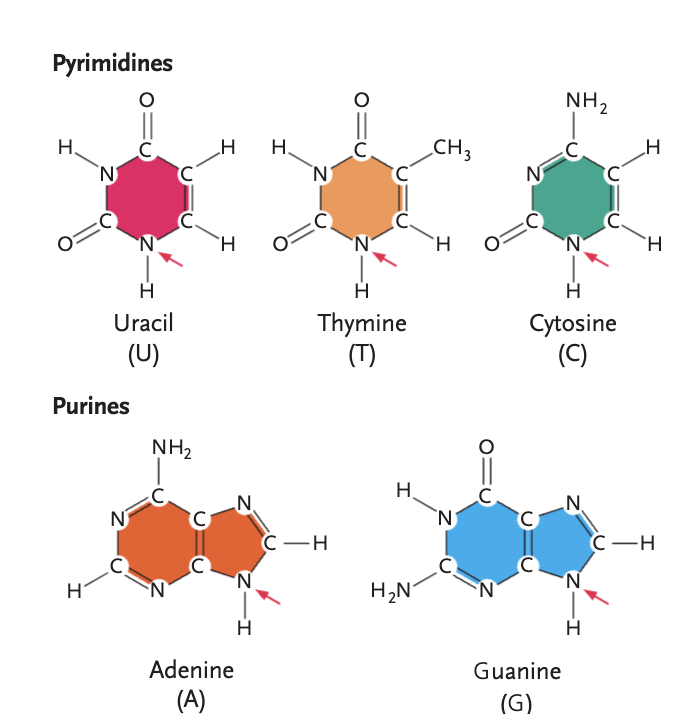
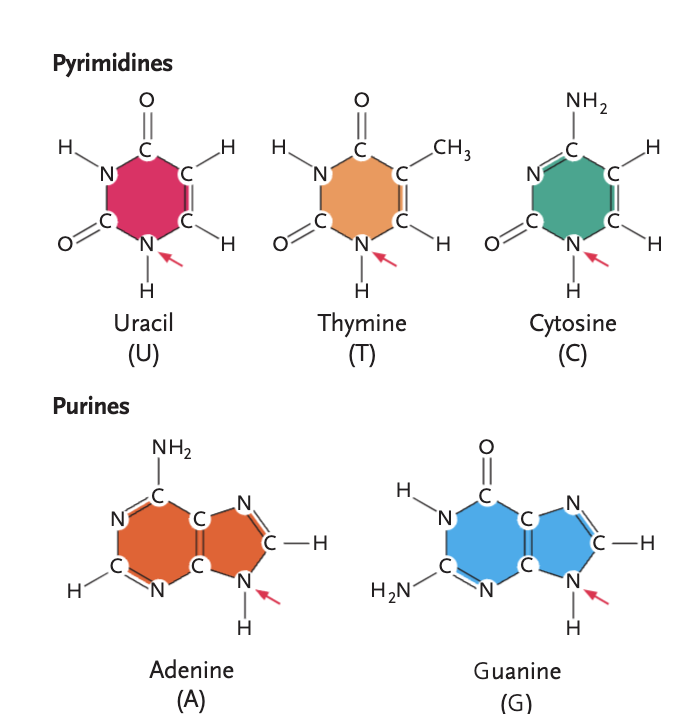
purines
adenine (A), guanine (G)

nucleoside
contains only a nitrogenous based and sugar

deoxyribose sugar
a 5-carbon sugar with a -H at the 2’ carbon
ribose sugar
a 5-carbon sugar with a -OH at the 2’ carbon (what nitrogenous bases will covalently link to)
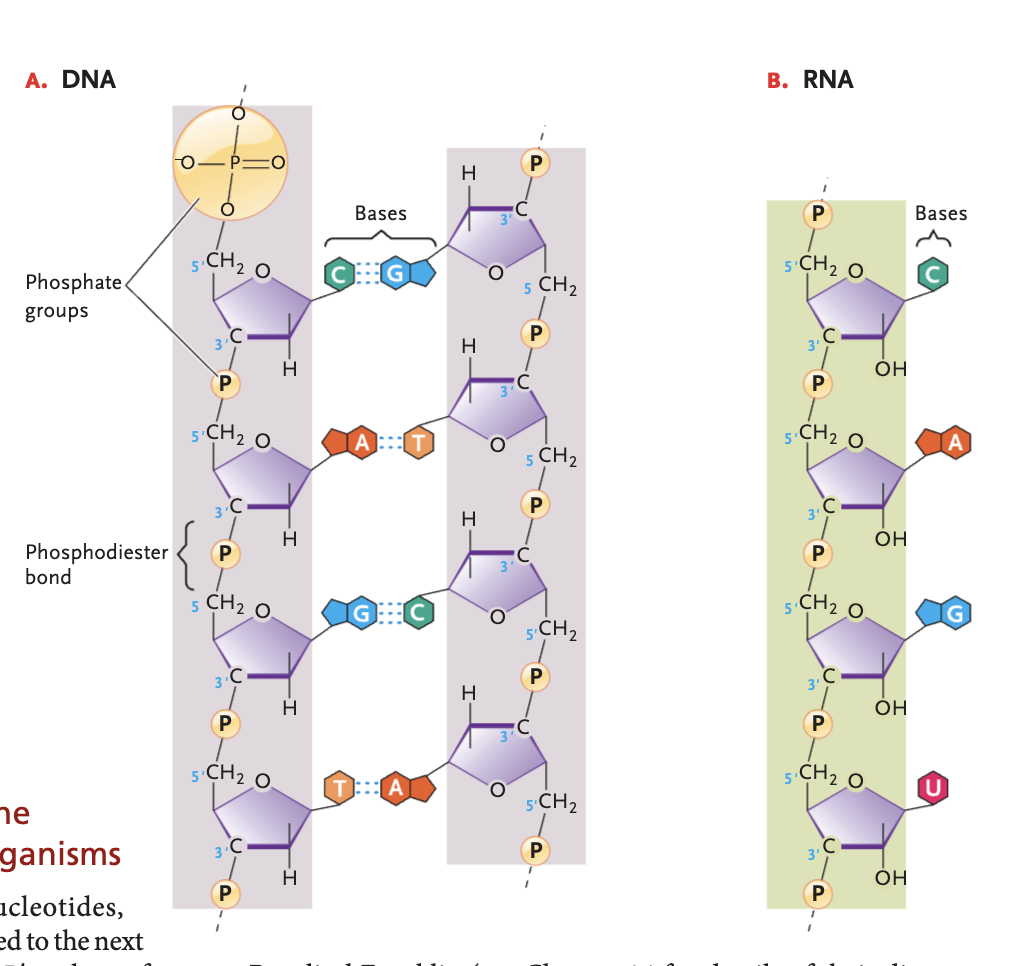
phosphodiester bond
strong covalent linkages between the 3' carbon atom of one sugar molecule and the 5' carbon atom of another in nucleic acids forming the structural backbone of them, connected by a phosphate group
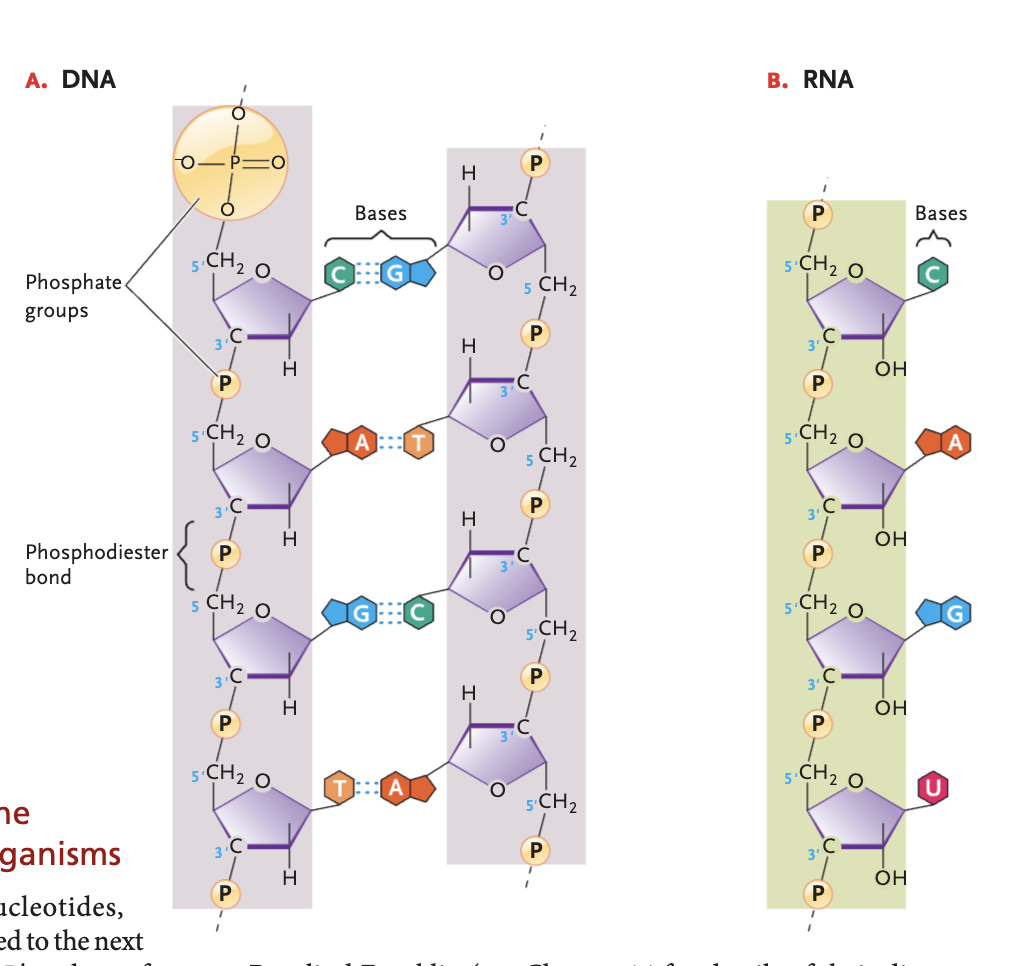
DNA bases (and pairs)
A-T, G-C
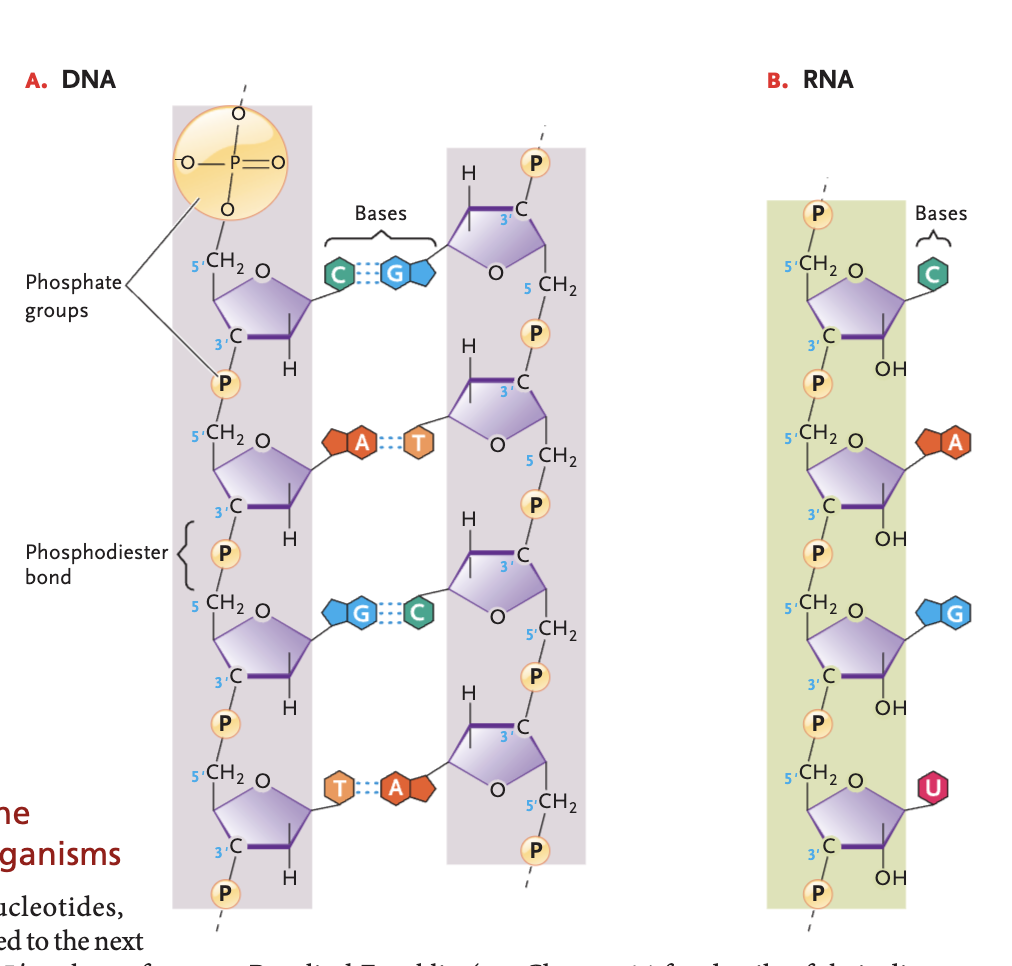
RNA bases (and pairs)
A-U, G-C
Thymine vs. Uracil
different only by functional group, thymine uses methyl, and uracil uses -H
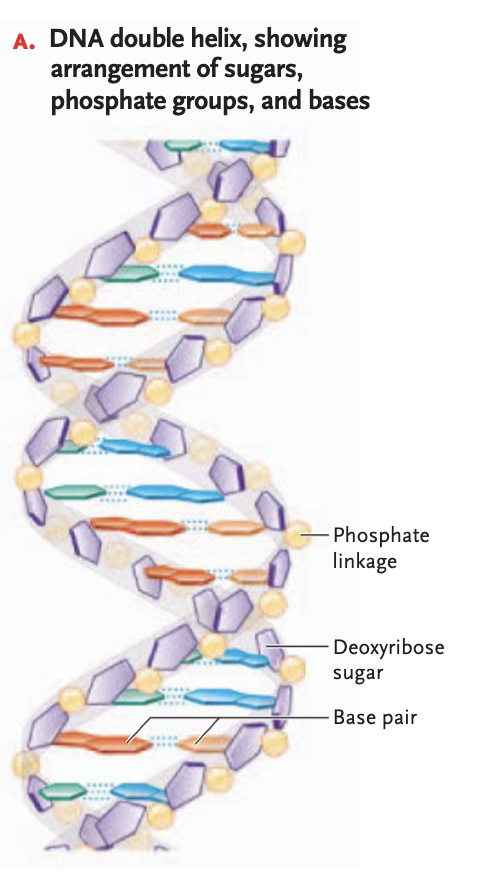
DNA double helix structure
a twisted-ladder structure composed of two antiparallel polynucleotide strands
2nd Law of Themodynamics
from intiial —> final state, the disorder of a system (cell) and its surroundings increases
entropy
a fundamental scientific concept measuring the unavailability of a system's energy to do useful work
measure of molecular disorder (releasing energy into surroundings even when moving towards organization —> takes a lot of energy to overcome disorder)
spontaneous reactions
a chemical/physical reaction that occurs without the input of energy, delta G will be negative, reactants have higher energy that products do
free energy
portion of a system’s energy that is available to do work
equation for free energy
delta G - delta H -TdeltaS
delta g: free energy
delta h: change in enthalpy
delta s: change in entropy
exergonic reactions
releases free energy (delta g is negative)
endergonic reactions
products contain more energy than reactants (delta g is positive)
metabolic pathway types
catabolic and anabolic pathways
catabolic pathway
energy is released by breakdown of complex molecules (overall delta G is negative- —> individual reactions can have negative or positive delta G)
breaking down polymers
anabolic pathway
energy used to build complex molecules from smaller ones (overall delta G is positive —> individual reactions can have negative or positive delta G)
can also be called a biosynthetic reaction, building up polymers
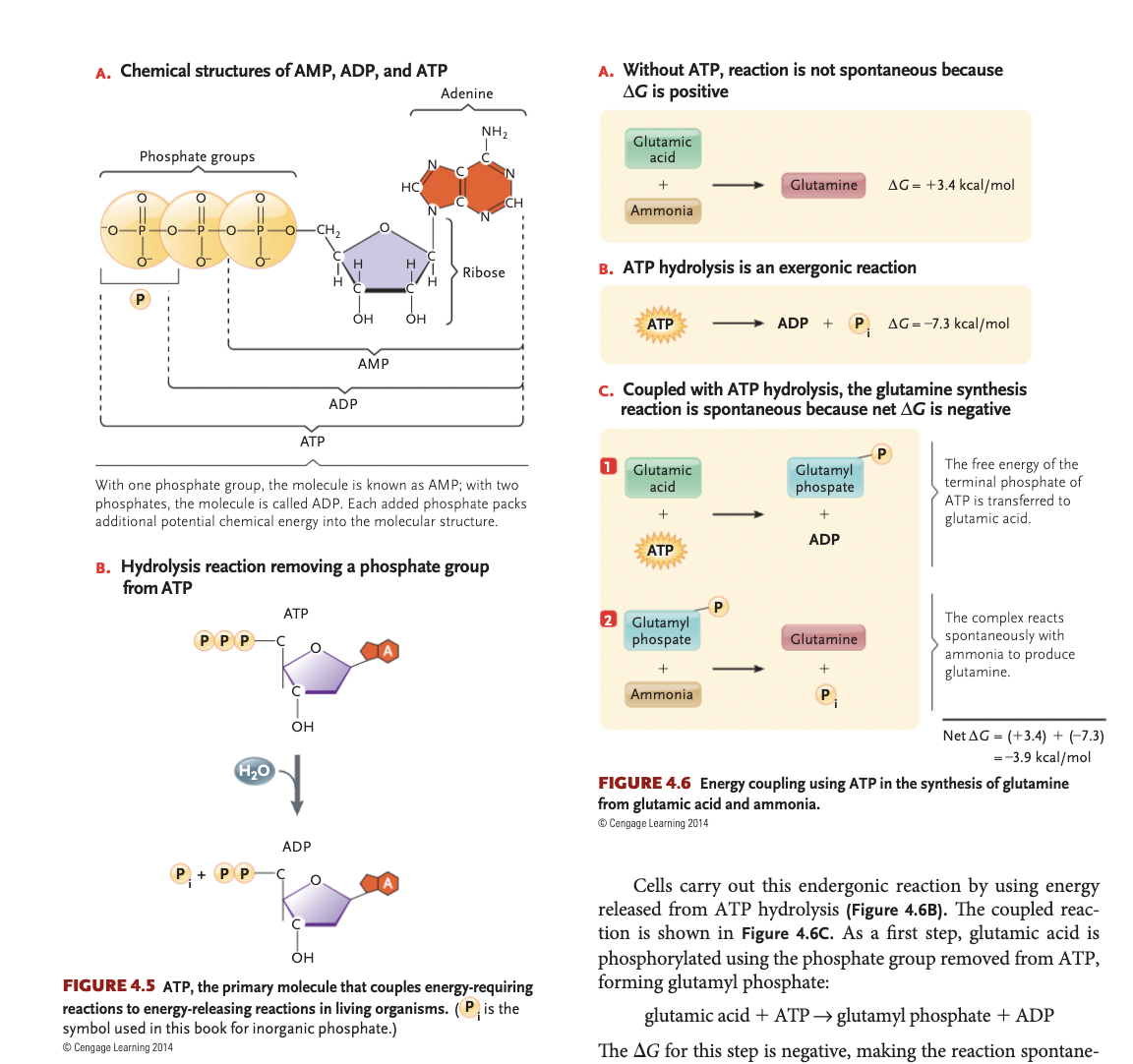
ATP hydrolysis
ATP+H2O→ADP+Pi+free energy, delta G = -7.3 kcal/mol, this Pi can couple reactions
energy coupling
(cell usually breaks things down into smaller processes) using an exergonic reaction (ex. ATP hydrolysis) to drive an endergonic one
ex. ATP is hydrolysed and phosphate group is transferred to the reactant molecule of an endergonic reaction —> typically the only way that an endergonic/energetically unfavorable reaction can occur (exergonic reaction has to be of a high enough magnitude so the total delta G is negative)
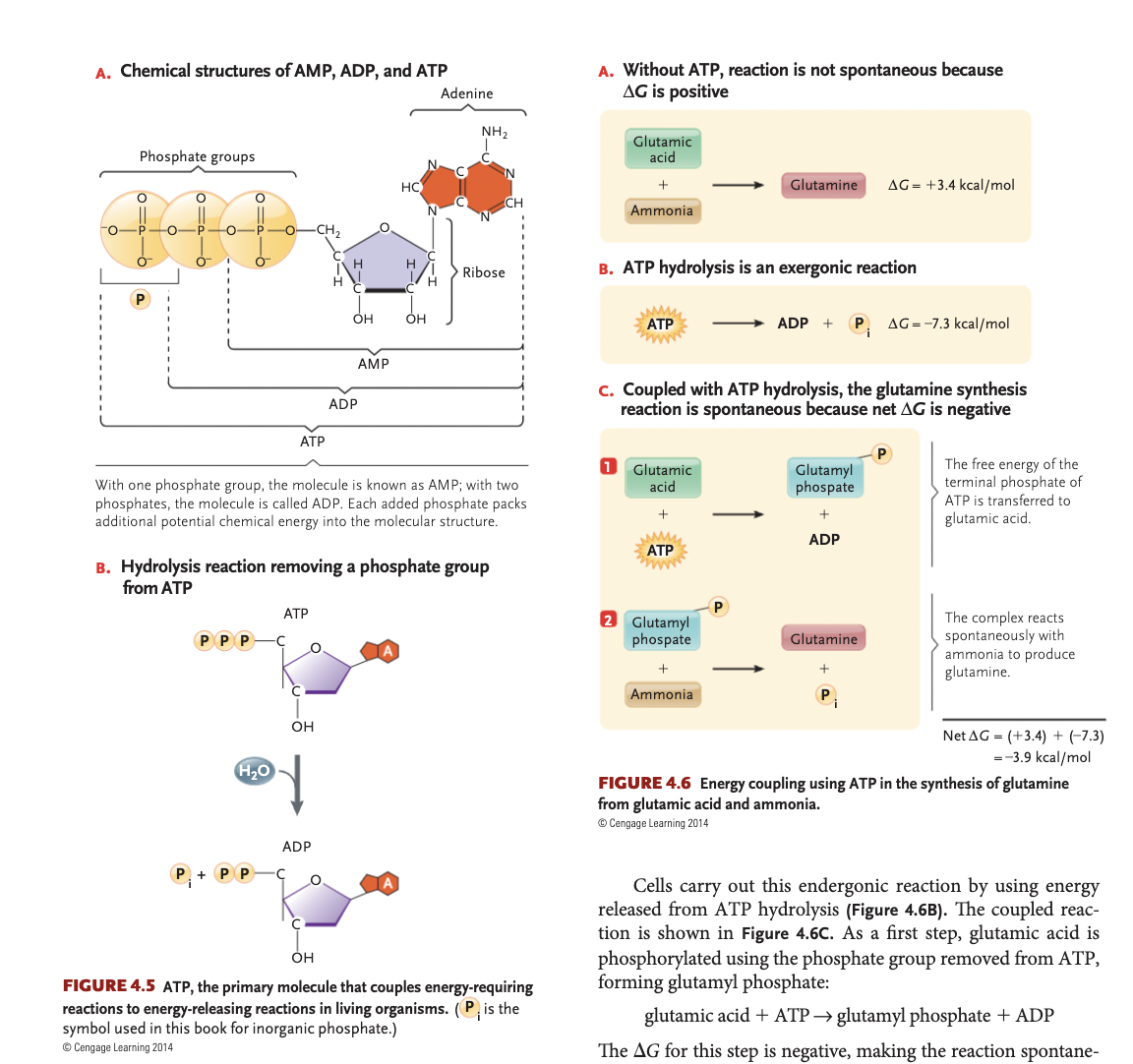
phosphorylation
addition of a phosphate group to a molecule, makes molecules less stable
ATP synthesis
an endergonic reaction, that requires energy from glutamine synthesis
glutamic acid gets phosphorylated (is unstable), ammonia replaces the Pi to produce glutamine
can happen in the opposite direction as well, formation of glutamic acid catalyzed by ATP hydrolysis
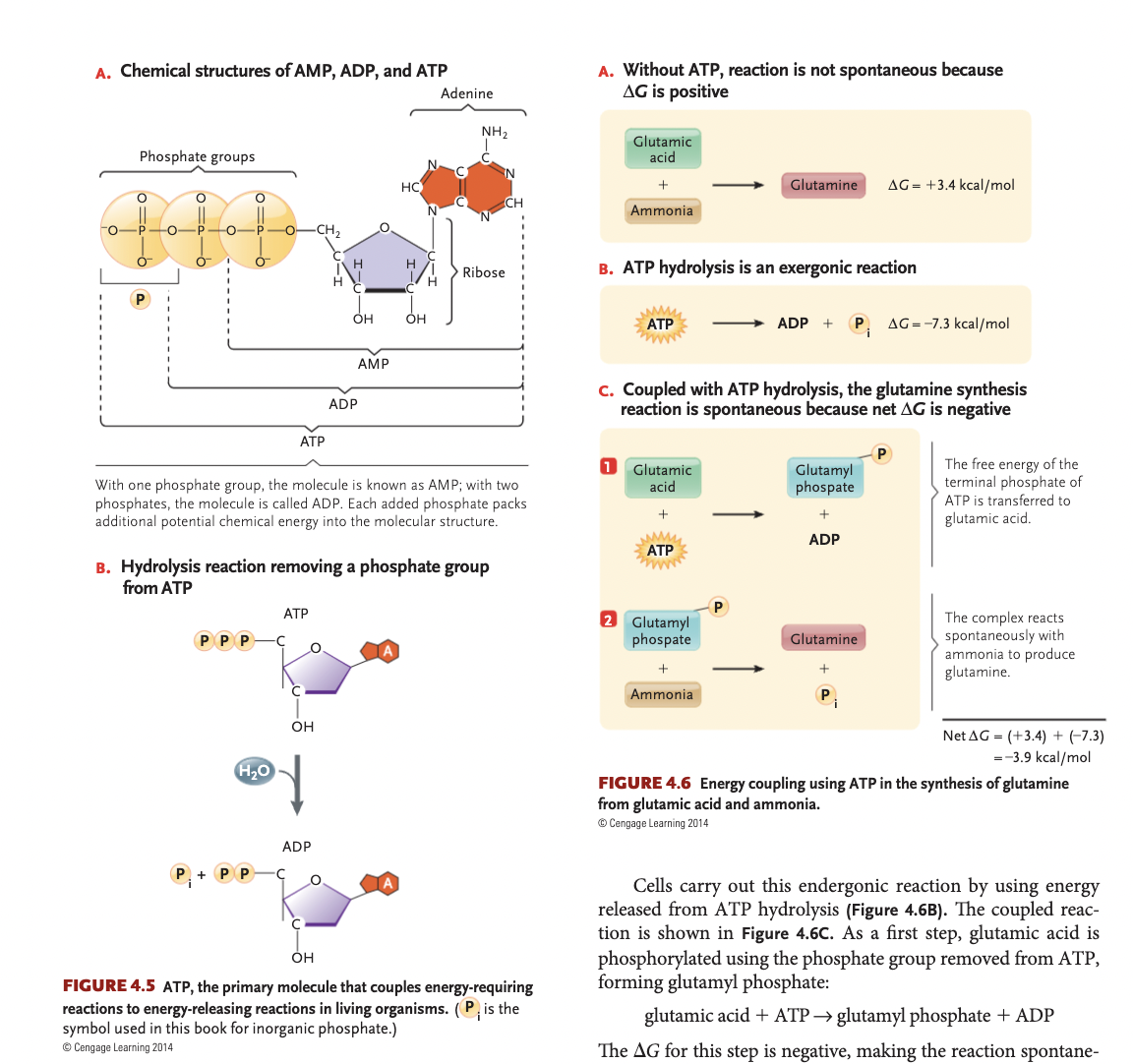
ATP Hydrolysis + Sodium-Potassium pump
(endergonic reactions) The sodium-potassium pump is an essential transmembrane enzyme that uses energy from ATP hydrolysis to actively move 3 Na+ ions out of the cell and 2 K+ ions in
catalysis
the acceleration of a chemical reaction rate by adding a substance, known as a catalyst, which is not consumed in the process
at high concentrations of substrate (excess) the rate of catalysis is proportional to the amount of enzyme, rate of reaction is limited by enzyme
saturation
when enzymes are already cycling as quickly as possible —> additional substrate won’t affect reaction rate
purpose of enzyme
meant to lower activation energy,
catalyzing a reaction,
does not get used up over the course of reaction
activation energy
barrier in the movement of reaction progression
plasma membrane
what all cells are bounded by, phospholipid bilayer w/embedded protein molecules
some water-soluble substances can penetrate the membrane through transport proteins
Role: enclose the organelles, provide cellular structure (being defined by extracellular matrix), receive and transfer signals from the extracellular environment to intracellular pathways, barrier between outside and inside of the cell, regulates which molecules enter and exit the cell
Genes
DNA sections that code for individual proteins
cytoplasm
all parts of the cell between the plasma membrane and central DNA-containing region
organelles
found in cytoplasm - specialized, membrane-bound structures within eukaryotic cells that perform essential, organ-like functions
cytosol
aqueous solution containing ions and various organic molecules
cytoskeleton
protein-based framework of filamentous structures that maintain cell shape + mitosis
interacts with motor proteins to produce cell motility, other organelles use motor protein “feet” to move around, top can attach to a vesicle and move w/ ATP
prokaryotes
(ex. bacteria + archaea) unicellular organisms
some bacteria have 2 cell membranes
nucleoid
DNA-containing region in prokaryotes (has no boundary separating it from the cytoplasm) —> no internal memberane
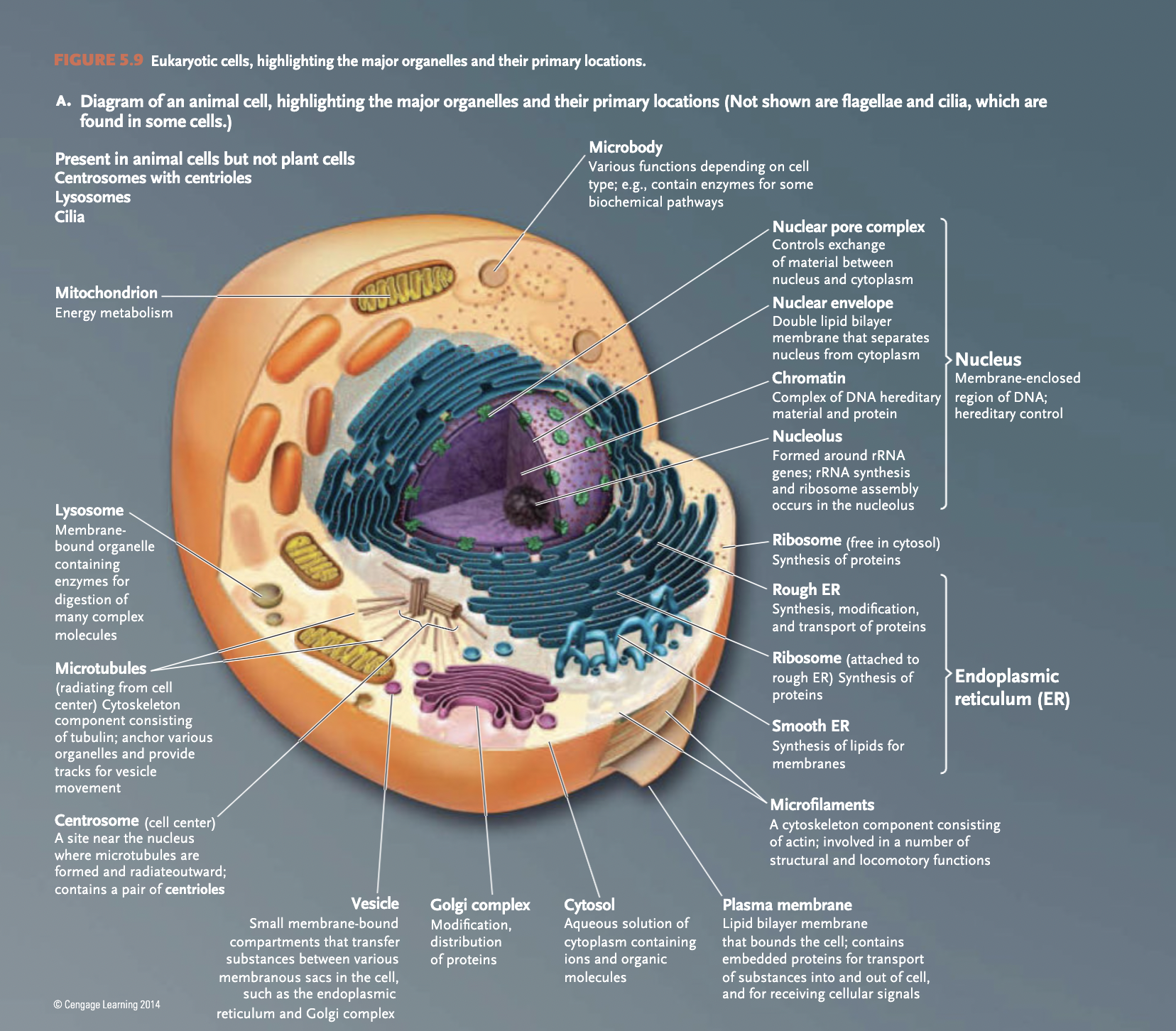
eukaryotes
(eukarya) multicellular organisms
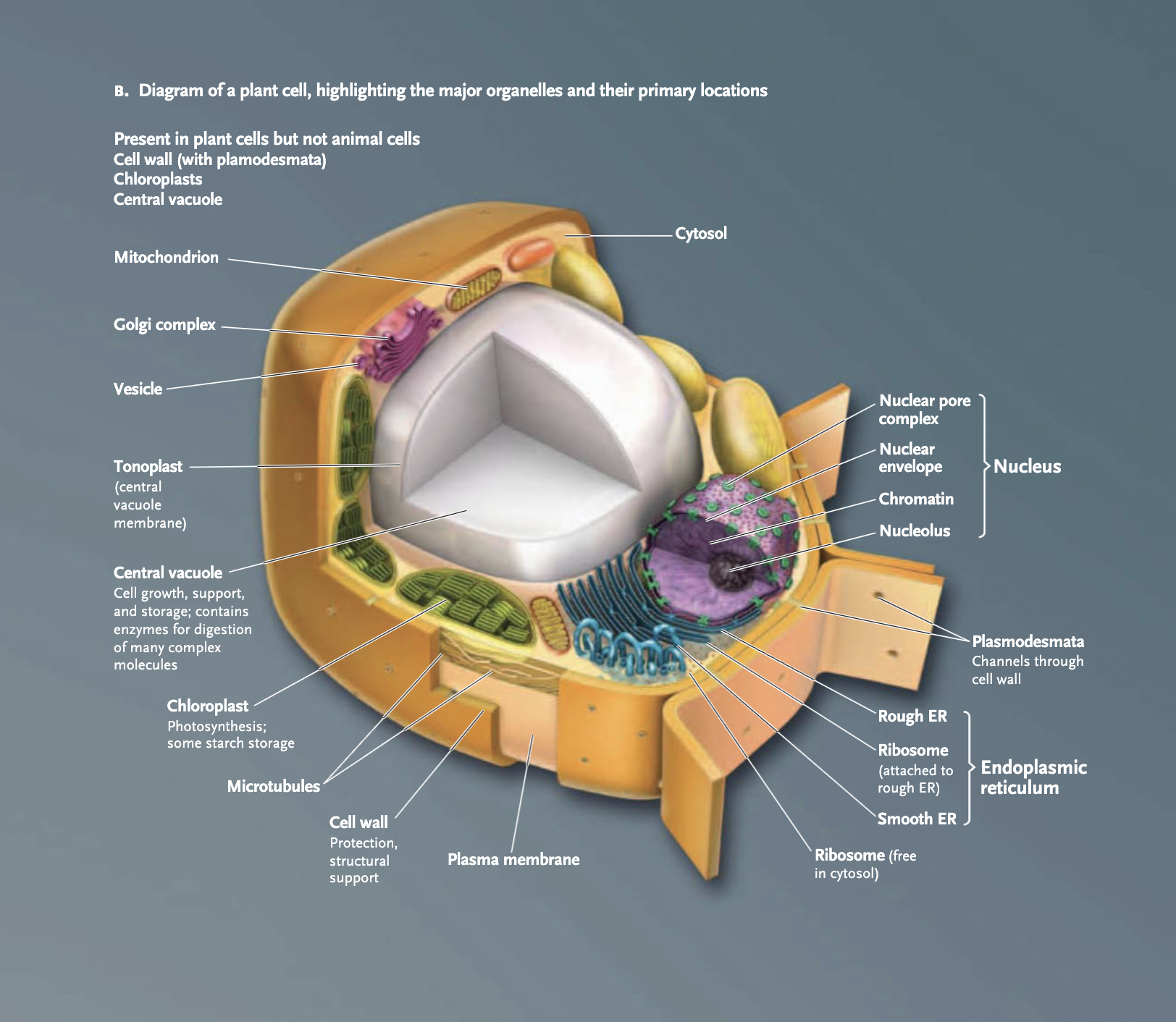
Cell wall
(plant cells, prokaryotes): surrounds plasma membrane, rigid external layer of material
helps protect cell from physical damage
in many prokaryotic cells, cell wall is coated with an external layer of polysaccharides called glycocalyx —> slime layer forms when glycocalyx is loosely associated with the cells, forms a capsule when the glycocalyx is gelatinous and attached more firmly to cells
flagella/bacterial flagellum
threadlike fibers which extend from cell surface,
helically shaped rotates in a socket to push the cell through a liquid medium
pili
hairlike shafts of protein attaching the cell to surfaces of other cells
nuclear envelope
consists of 2 lipid bilayer membranes, one just outside the other and separated by a narrow space
nuclear pore complex: large octagonally shaped symmetric complex in the cell, formed of many types of proteins called nucleoporins
nuclear pore: a channel through nuclear pore complex is the path for assisted exchange moleucles (proteins, RNA)
exocytosis
a form of active, bulk transport where cells export materials—such as neurotransmitters, hormones, and waste—by fusing intracellular vesicles with the plasma membrane
endocytosis
an active transport mechanism where cells internalize external materials—fluids, nutrients, or pathogens—by engulfing them within a plasma membrane-derived vesicle
peroxisomes
small, single-membrane-bound, metabolic organelles found in almost all eukaryotic cells, crucial for breaking down fatty acids and neutralizing toxic materials.