ap chem unit 2: chemical bonding
1/48
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
49 Terms
what are inter and intra molecular forces based upon?
coulombic attractions
what is the difference between inter and intra molecular forces?
inter: interactions between covalent molecules (typically weak) - hydrogen bonding, dipole-dipole, LDFs
intra: within compounds (both strong) - ionic and covalent
the ability of an atom within a covalent bond to attract electrons to itself
electronegativity
what happens if two covalently bonded atoms have the exact same electronegativity?
the electrons will be equally shared and distributed, making it a non-polar covalent bond
example: diatomic elements, elements that have similar electronegativities

what is the difference in electronegativities chart?
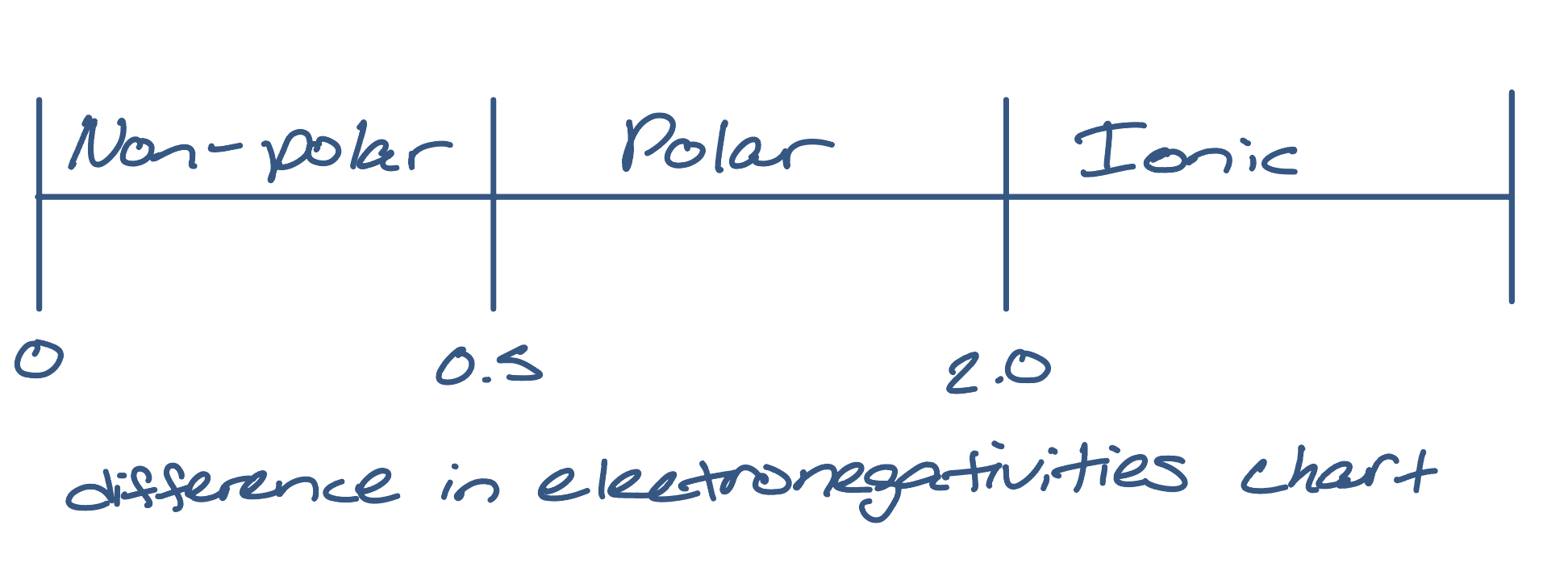
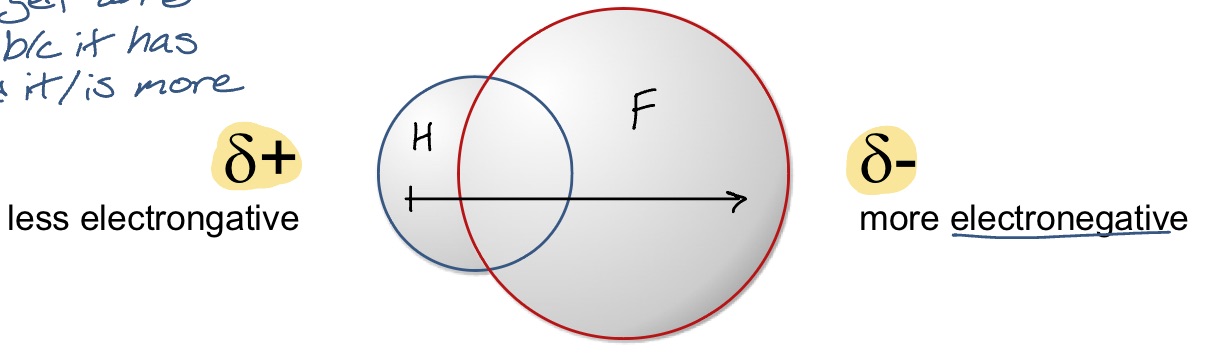
what happens in situations where one atom has a much higher electronegativity than the other (covalent bonding)?
electrons are attracted towards the more electronegative atom, leading to a re-distribution of the electron cloud. the molecule is then said to have a dipole and is classified as polar-covalent
example: HF
what do the chemical and physical properties of a compound rely upon?
the presence or absence of dipoles and the intermolecular coulombic attraction determine the type of intermolecular forces present. in turn, the type of intermolecular forces present can greatly influence the properties of the molecule
what is the difference between molecules and particles?
molecules: covalently bonded compounds
particles: ions and ionicly bonded compounds
explain covalent bonding
valence electrons shared to achieve full s and p valence sub shells (octet), forming molecules, typically between two nonmetalse
one shared pair of electrons represent a single bond (longest and weakest)
two shared pairs represent a double bond (shorter and stronger)
three shared pairs of electrons represent a triple bond (shortest and strongest)
explain the leonard jones internuclear distance versus potential energy plot
x axis is bond length
y axis is bond strength and how much energy it requires to break it apart
the lower on the y-axis, the stronger the bond
explain ionic bonding
the differences in electronegativities are so great that one atom can completely take an electron away from the other (transfer of electrons)
all atoms are neutral. once they gain/lose electrons, they are charged particles (ions)
strong bond: higher melting and boiling points
explain how coulomb’s law applies to ionic bonding and give an example
the strongest ionic bonds are formed between ions that are small (because they can get closer to one another) and that have the highest magnitude charges
smaller+higher charge=stronger bond
example: BeO is stronger than FrI
when comparing sodium chloride and sodium bromide, which compound would be expected to have the strongest ionic bonds, i.e., the greatest Coulombic attraction? explain your answer
Sodium chloride (NaCl) has stronger ionic bonds (greater Coulombic attraction) than sodium bromide (NaBr) because the chloride ion is smaller than the bromide ion, resulting in a shorter distance between ions and thus a stronger coulombic attraction.
how are ions in ionic solids positioned?
in an ionic/crystal lattice held together by strong electrostatic forces, giving ionic solids high melting and boiling points

can ionic substances conduct electricity?
no, unless they are molten (liquid) or in solution (aqueous).e
explain what solvation does to an ionic solid
solvation: act of dissolving
polaw water molecules are attracted to oppositely charged ions, penetrating the lattice and attaching themselves to ions
ion to dipole
process is called hydration
ions conduct electricity in this state because ions are free to move
what are the solubility rules?
any ionic compound with Na+, K+, NH4+, or NO3- will always dissolve
can non polar solvents dissolve polar bonds?
no
explain ionic substance with some covalent character
if the cation is small and highly charged, it will have the ability to distort the charge cloud around the anion. distortion will be at a maximum when the cation is small and highly charged and when the anion is large and highly charged (electrons more loosely held)
further away electronegativities
example: BeTe
Be is small and highly charged; higher coulombic attraction to electrons, distorting/pulling the electron cloud of Te
Te is large with a high charge

explain covalent substance with some ionic character (polar covalent)
if one atom in a covalent bond has a higher electronegativity than the other, then the electrons are attracted towards the more electronegative atom, leading to an electron could distortion and re-distribution of electron charge density
closer electronegativities

explain nonpolar/polar/and ionic based on periodic table
the closer the elements are on the periodic table, the more likely they are to be non polar covalent. the further away they are, they gain greater ionic character and polarity because of greater electronegativity differences
how do you make lewis structures?
count the number of valence electrons on each atom
put the least electronegative element in the center
bond until all atoms have a full octet
exceptions:
H has a duet and can ONLY be an outside atom
elements in period 3 have expanded octets (10, 12)
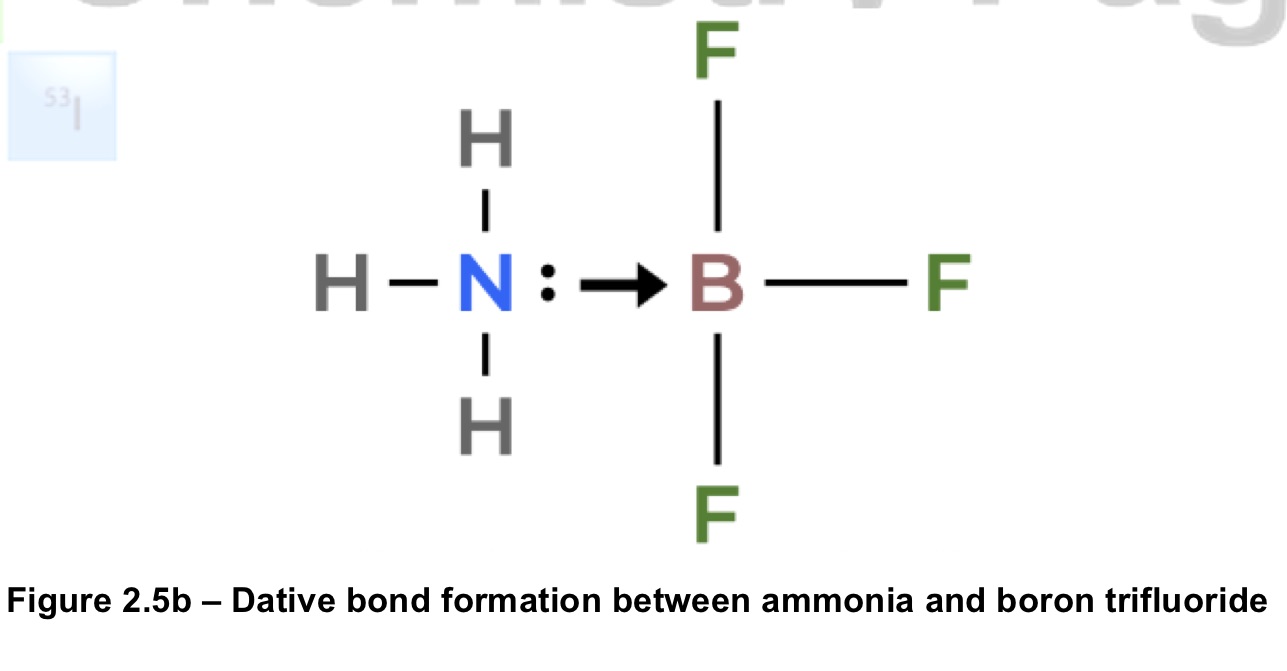
what are dative/co-ordinate bonds? give an example
a covalent bond where one atom donates both of the shared electrons in a bond, unlike a normal covalent bond where each atom contributes one electron. an example is boron, which often does not have a complete octet (only has 6 electrons) but can make up the octet by forming bonds with other compounds that have a non-bonding pair such as NH3
explain the chart for hybridization

explain single, double, and triple bonds in the terms of sigma and pi bonds
single: 1 sigma bond
double: 1 sigma, 1 pi
triple: 1 sigma, 2 pi
pi bond is evidence of unhybridized orbital
what are the names of hydrocarbons based on single, double, and triple bonds
single bonds: alkane
double bonds: alkenes
triple bonds: alkynes
what are isomers?
same formula, different arrangement of atoms. to be isomers, it has to have another object to compare with

what are resonance structures?
a lewis structure that involves multiple bonds and therefore can be drawn in different ways; shows differing arrangements of electrons
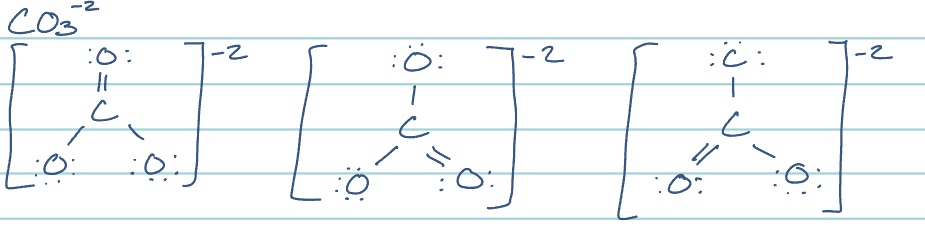
how do you determine the best lewis structure if it is not equally likely?
use formal charge of individual atoms
formal charges of 0 is best
smallest total formal charge is second best (smallest: absolute value closest to 0)
negative numbered formal charge goes on the most electronegative element
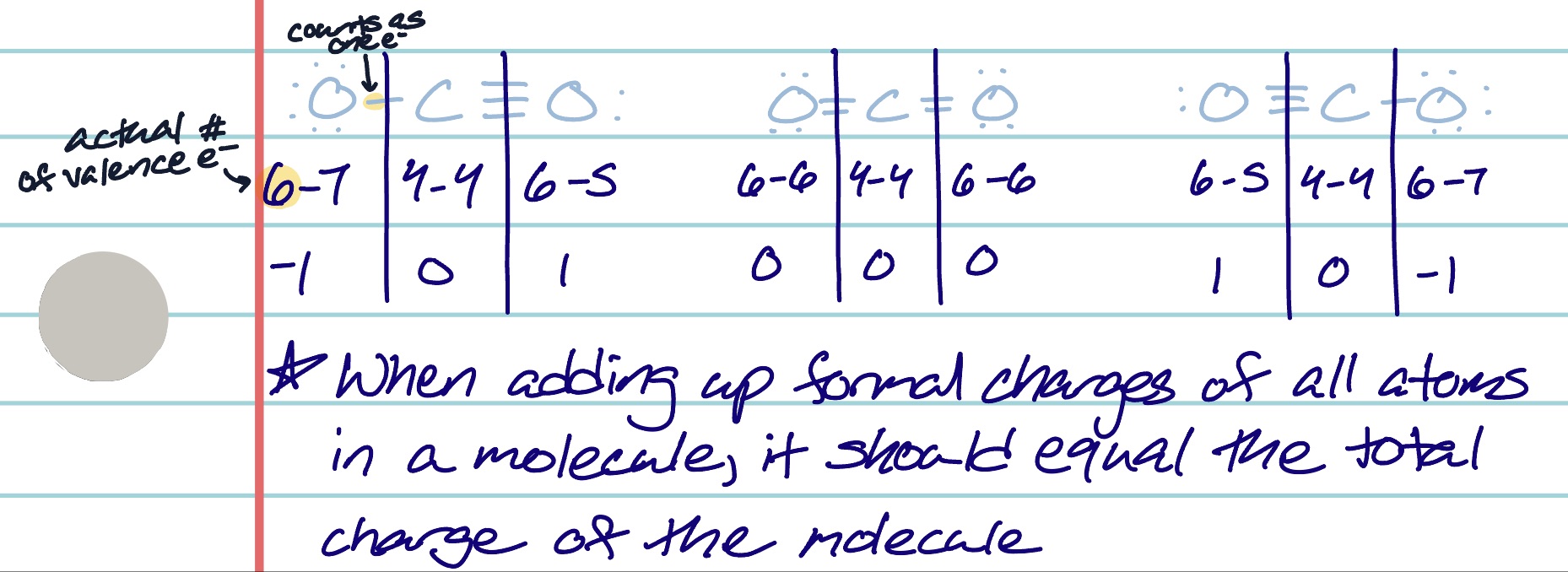
what happens if all resonance structures are likely to occur on the same molecule?
electrons and bonds move around
actual measurements of bonds are an average of the lengths of all bonds because the bond is split between all atoms
what are the rules of polarity?
for a molecule to be polar, 2 things must be true:
molecules must contain at least one bond dipole
bond dipoles must not cancel out due to symmetry (make sure to check ALL angles of symmetry because if you can find evidence for asymmetry, then it is polar)
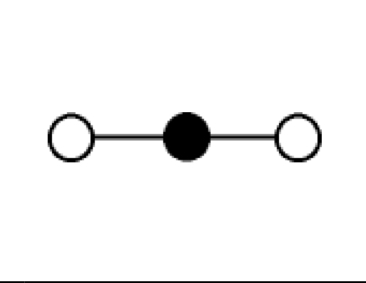
what are the bond angles, electron geometry, and molecular geometry of a molecule that has 2 bonds and no lone pairs around the central atom? what does it look like?
2 bonds, no lone pairs
bond angles: 180 degrees
electron geometry: linear
molecular geometry: linear
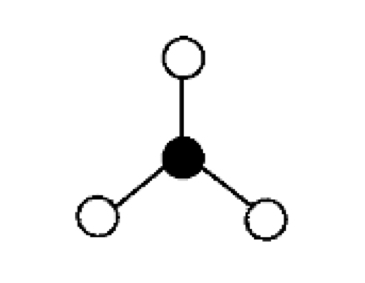
what are the bond angles, electron geometry, and molecular geometry of a molecule that has 3 bonds and no lone pairs around the central atom? what does it look like?
3 bonds, no lone pairs
bond angles: 120 degrees
electron geometry: trigonal planar
molecular geometry: trigonal planar
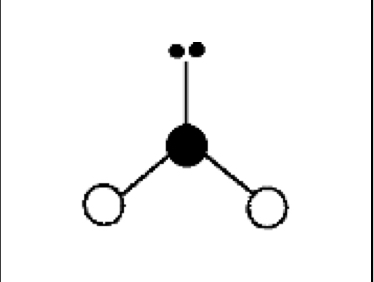
what are the bond angles, electron geometry, and molecular geometry of a molecule that has 2 bonds and 1 lone pairs around the central atom? what does it look like?
2 bonds, 1 lone pair
bond angles: slightly less than 120 degrees
electron geometry: trigonal planar
molecular geometry: bent or v shaped
what are the bond angles, electron geometry, and molecular geometry of a molecule that has 4 bonds and no lone pairs around the central atom? what does it look like?
4 bonds, no lone pairs
bond angles: 109.5 degrees
electron geometry: tetrahedral
molecular geometry: tetrahedral
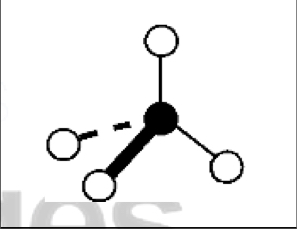
what are the bond angles, electron geometry, and molecular geometry of a molecule that has 3 bonds and 1 lone pairs around the central atom? what does it look like?
3 bonds, 1 lone pair
bond angles: 107.5 degrees
electron geometry: tetrahedral
molecular geometry: trigonal pyramidal
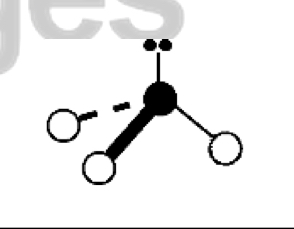
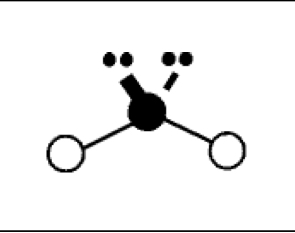
what are the bond angles, electron geometry, and molecular geometry of a molecule that has 2 bonds and 2 lone pairs around the central atom? what does it look like?
2 bonds, 2 lone pairs
bond angles: 104.5 degrees
electron geometry: tetrahedral
molecular geometry: bent or v shaped
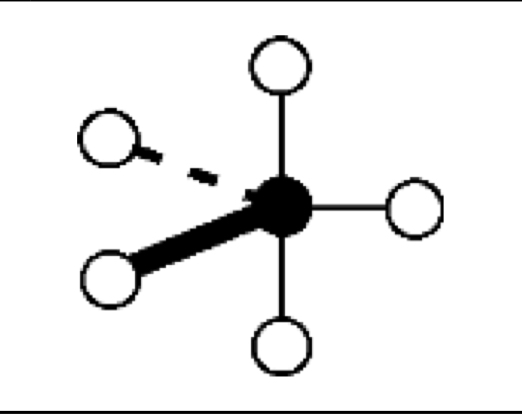
what are the bond angles, electron geometry, and molecular geometry of a molecule that has 5 bonds and no lone pairs around the central atom? what does it look like?
5 bonds, no lone pairs
bond angles: 120 degrees in plane, 90 degrees perpendicular to plane
electron geometry: trigonal bipyramidal
molecular geometry: trigonal bipyramidal
what are the bond angles, electron geometry, and molecular geometry of a molecule that has 4 bonds and 1 lone pairs around the central atom? what does it look like?
4 bonds, 1 lone pair
bond angles: complex
electron geometry: trigonal bipyramidal
molecular geometry: seesaw
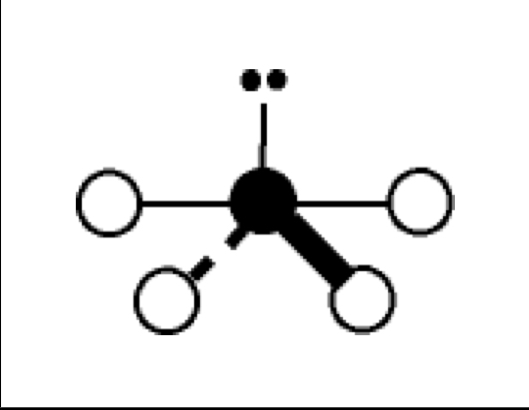
what are the bond angles, electron geometry, and molecular geometry of a molecule that has 3 bonds and 2 lone pairs around the central atom? what does it look like?
3 bonds, 2 lone pairs
bond angles: about 90 degrees
electron geometry: trigonal bipyramidal
molecular geometry: t shaped
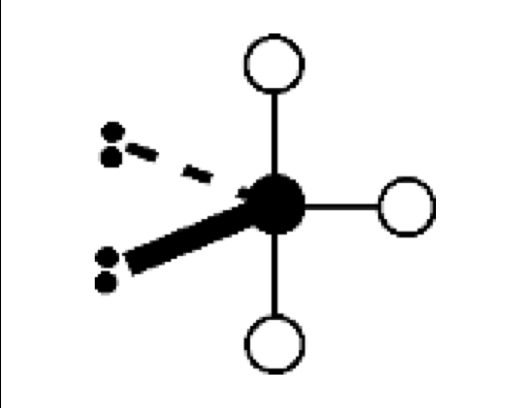
what are the bond angles, electron geometry, and molecular geometry of a molecule that has 2 bonds and 3 lone pairs around the central atom? what does it look like?
2 bonds, 3 lone pairs
bond angles: 180 degrees
electron geometry: trigonal bipyramidal
molecular geometry: linear
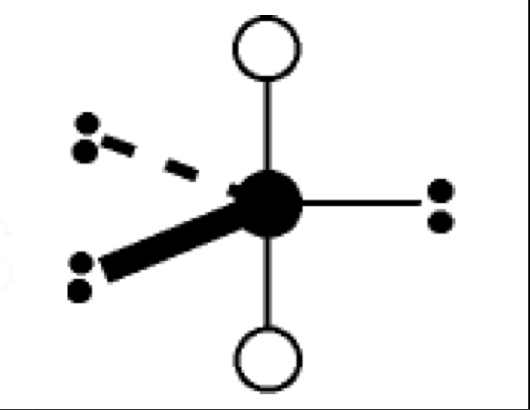
what are the bond angles, electron geometry, and molecular geometry of a molecule that has 6 bonds and no lone pairs around the central atom? what does it look like?
6 bonds, no lone pairs
bond angles: 90 degrees
electron geometry: octahedral
molecular geometry: octahedral
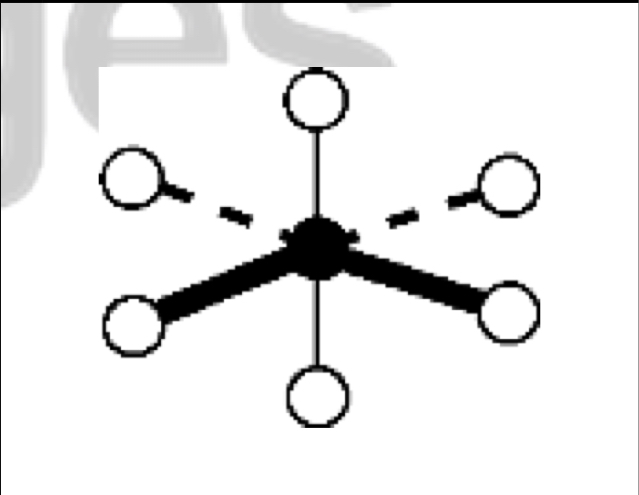
what are the bond angles, electron geometry, and molecular geometry of a molecule that has 5 bonds and 1 lone pairs around the central atom? what does it look like?
5 bonds, 1 lone pair
bond angles: about 90 degrees
electron geometry: octahedral
molecular geometry: square pyramidal
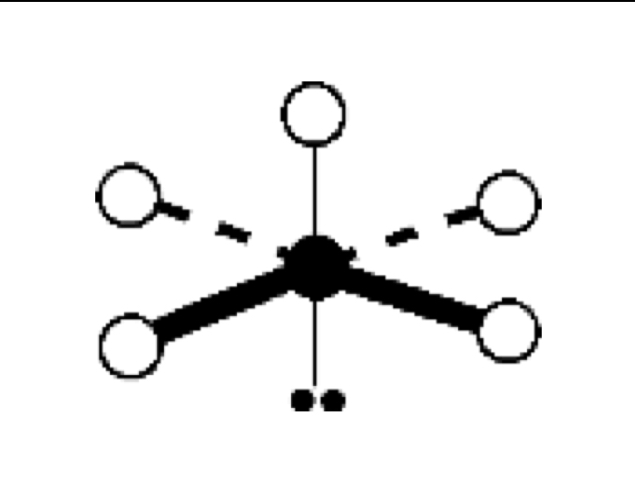
what are the bond angles, electron geometry, and molecular geometry of a molecule that has 4 bonds and 2 lone pairs around the central atom? what does it look like?
4 bonds, 2 lone pairs
bond angles: 90 degrees
electron geometry: octahedral
molecular geometry: square planar
what is a special property of metals and what other properties does that cause?
metals have a sea of delocalized electrons that surrounds the positively charged cations
makes metals good conductors of heat and electricity
causes malleability/ductilitywh
what is an alloy?
a mixture between multiple metals or a metal and nonmetal
NO chemical bonding occurring because it is a mixture
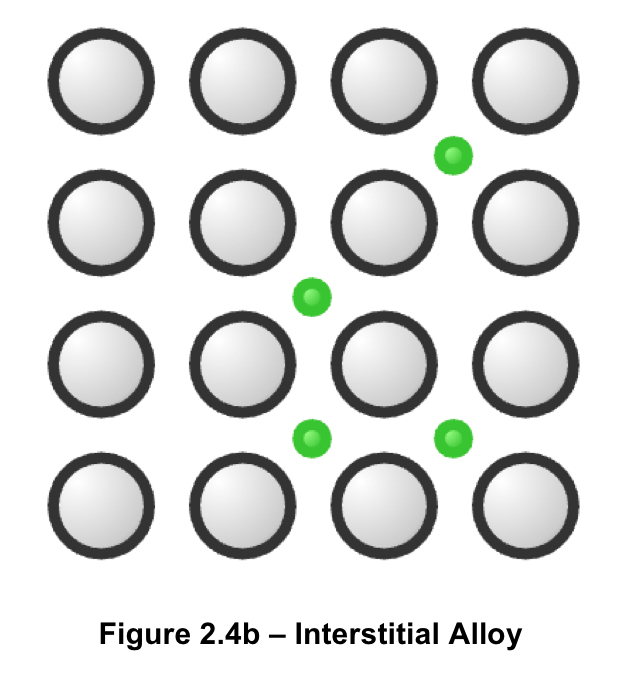
what is an interstitial alloy? give an example
smaller atoms fill the spaces between larger atoms
happens when there is a large difference in atomic radii
typically formed between elements of different periods
typically stronger than metallic counterpart
example: steel (iron and carbon)
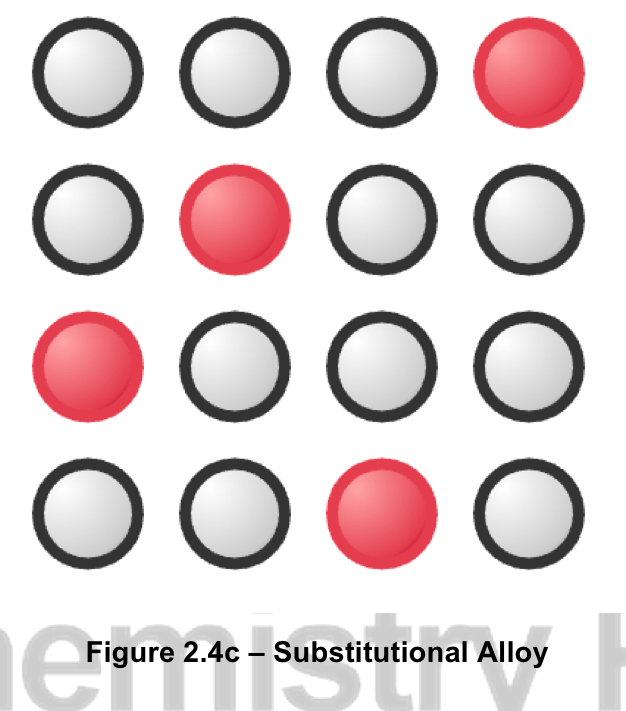
what is a substitutional alloy? give an example
atoms of one metal are replaced by the atoms of another metal, like they’re mixed together basically
happens when atomic radii are similar
typically formed between elements of the same period
example: brass (copper and zinc)
what are the similarities between alloys and metals?
generally good conductors of electricity
sometimes the surface will have different properties from the core
outside can react with oxygen and form an oxide, which has different properties from the inside portion which hasn’t reacted