Signal transduction 3
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
40 Terms
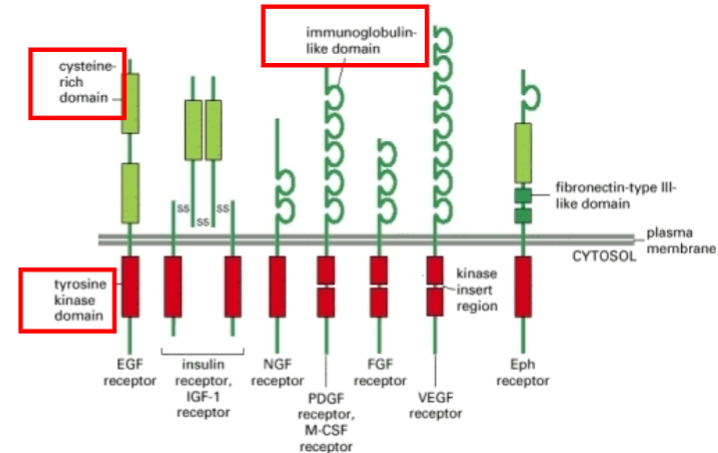
What are the four major types of membrane-bound receptors?
Ion channel enclosing R
7 transmembrane R
1 Hydrophobic domain R
R with enzyme activity
R with no enzyme activity
Which class of R does the insulin receptor belong to?
Signalling through 1 Hydrophobic domain R carrying enzymatic activity
Signalling through 1 Hydrophobic domain Receptors are aka what
Receptor tyrosine kinases (RTKs).
What is the signal that activates receptor tyrosine kinases (RTKs)?
Growth factors and trophic factors such as cytokines, typically acting in a paracrine manner.
How many RTKs exist in humans and how many subfamilies are they split into?
58 RTKs grouped into 20 sub-families
What is the structure of an RTK?
One transmembrane region, variable extracellular ligand-binding domain, and intracellular tyrosine kinase domain.
What is the immediate effect of ligand binding to RTKs
Induction of RTK dimerisation, which leads to activation.
Explain generally what happens between a RTK being inactive to finally that signal being relayed to the cell
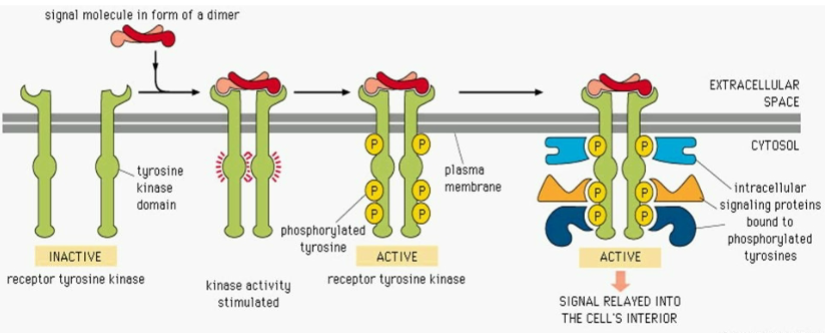
What process happens to RTKs after ligand binding?
The receptor undergoes trans-autophosphorylation on multiple tyrosine residues in a precise sequence
What is the significance of phosphorylated tyrosines on RTKs?
They serve as docking sites for enzymes and adaptor proteins.
What are adaptor (scaffold) proteins
Proteins with platforms to bind multiple proteins
Ability to bring protein complexes together
What type of proteins can dock on the phosphorylated tyrosines of RTKs
Proteins with SH2 (Src homology domain 2) and PTB (phosphotyrosine binding) domains.
What determines the downstream signalling and biological response that comes from an RTK
The type of adaptor protein
What are the three main pathways activated by RTKs?
Ras pathway → cell proliferation
PLC pathway → cell proliferation/differentiation
PI3K pathway → cell survival and metabolism
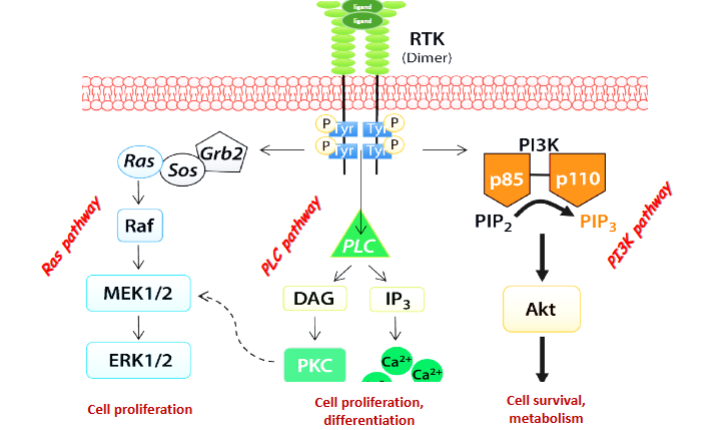
What type of protein is Ras?
A small GTPase, similar to G proteins.
What does Ras activate
Enzymes such as Raf (a serine/threonine kinase) and MAPK (mitogen activated protein kinases) signalling cascades.
Why is Ras clinically important?
The Ras family (HRAS, NRAS, KRAS) is one of the most frequently mutated oncogenes in human cancers.
What two regulatory proteins control Ras activity?
GEF (guanine nucleotide exchange factor)(Sos) → activates Ras by promoting GDP-GTP exchange
GAP (GTPase-activating protein) → inactivates Ras by stimulating GTP hydrolysis
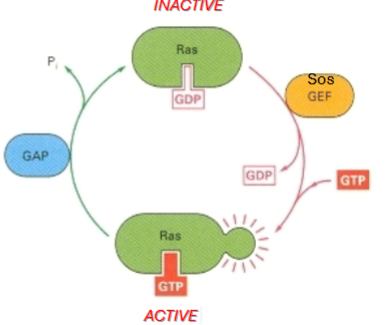
Why does Ras have to rely on GAPs for inactivation
Ras has slow intrinsic GTPase activity. It would eventually inactivate from GTP to GDP but it would take a long time.
If this regulation is lost, Ras becomes an oncogene.
Explain the Ras activation cascade
Grb2 scaffold protein binds P-Tyr on RTKs via its SH2 domain.
The 2 SH3 domains on Grb2 recruit Sos (a GEF).
Sos activates Ras by exchanging GDP for GTP.
Active Ras activates Raf (MAPKKK).
Raf activates MEK1 & MEK2 (MAPKKs).
MEK activates Erk1 & Erk2 (MAPKs).
What is the role of Erk1/2 in the Ras pathway?
They mediate cytosolic responses (e.g., regulation of proteins involved in apoptosis, & kinase activation)
Nuclear responses (e.g., transcription factor activation - Fos & Jun → Cyclin D)
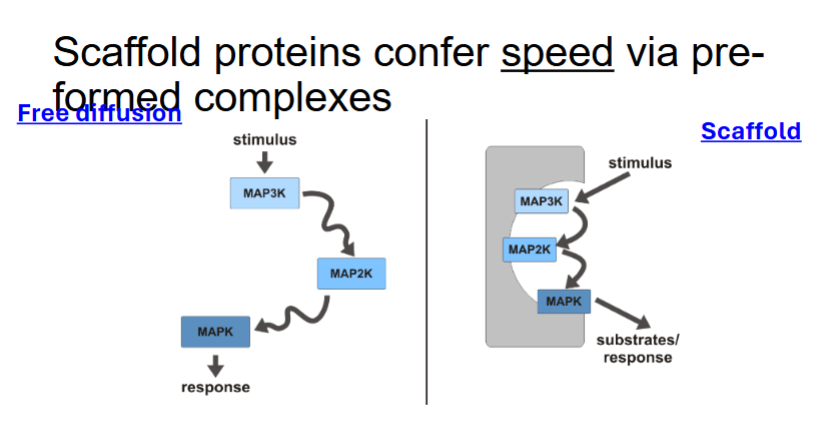
How do scaffold proteins influence Ras signalling?
Provide docking sites for multiple proteins.
Link proteins in a signalling cascade.
Increase speed and specificity.
Control location of signalling.
Can inhibit signalling at high concentrations.
Example of a scaffold protein in cascade of Ras signalling?
KSR (Kinase Suppressor of Ras).
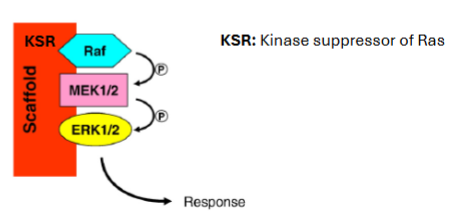
What happens if scaffold proteins are expressed at very high levels?
They can act as inhibitors - inhibit signalling by sequestering pathway components away from each other.
What type of mutation makes Ras oncogenic?
Which isoform most commonly mutates?
Gain-of-function mutations that affect GTP binding/hydrolysis, locking Ras in its active state.
Of the 3 Ras isoforms (HRAS, NRAS, KRAS), KRAS mutations are the most common oncogenic Ras mutations, especially in Pancreas, colon & lung cancers
What cellular outcomes are mediated by the PI3K (phosphatidyl inositol 3 kinase) pathway
Cell survival and metabolism.
What does PI3K phosphorylate? & what does that generate
Membrane lipid phosphatidylinositol 2-phosphate, generating PIP3
Why is PI3K clinically significant?
It is one of the most frequently mutated pathways in human cancers.
What is the structure of PI3K?
A heterodimer with a regulatory subunit and a catalytic subunit.
What motifs do PI3K subunits contain?
Multiple protein-binding motifs, including SH2 and SH3 domains.
What is the second messenger generated by PI3K activity?
PIP3.
Why is PIP3 a good second messenger?
It is membrane-bound, transient, and recruits proteins with PH domains.
How is Akt (Protein kinase B) activated in the PI3K pathway?
Inactive Akt resides in cytosol.
PH domains in Akt and PDK1 bind to PIP3 at the membrane.
Akt is phosphorylated at Thr308 by PDK1 (Phosphoinositide-
dependent kinase 1).
Akt is phosphorylated at Ser473 by mTORC2.
Phosphorylation stabilizes Akt, allowing it to dissociate into the cytosol.
How does activated Akt regulate acceptor proteins
By phosphorylating serine and/or threonine residues on them
Name four major targets of Akt and their effects.
Akt activates mTOR → promotes cell growth and metabolism
Akt activates GLUT4 → increases glucose uptake
Akt inhibits GSK-3 → promotes glycogen metabolism and cell cycle progression
Akt inhibits Pro-apoptotic proteins (Bad, Bim, Bax) → promotes cell survival
How does Akt affect transcription factors?
Inhibits Forkhead (FoxO1/3a) transcription factors, reducing expression of pro-apoptotic genes like Bim
What enzyme inactivates the PI3K pathway?
PTEN (phosphatase and tensin homologue deleted on chromosome 10).
What does PTEN do?
It is a dual protein and lipid phosphatase. It dephosphorylates PIP3 back to PIP2, switching off the response
Why is PTEN important clinically
It is one of the most frequently inactivated tumour suppressor genes in cancer.
Are PI3K and Akt oncogenes or tumour suppressor
Proto-oncogenes.
(Proto-oncogenes are the “normal accelerators” of cell growth. When mutated, they become oncogenes that jam the accelerator pedal down → cancer)