6.1.1 Aromatic Compounds
1/122
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
123 Terms
What are arenes?
aromatic hydrocarbons containing one or more benzene rings
What is the relationship between aromatic and aliphatic compounds?
they are mutually exclusive
What is the molecular formula of a benzene ring?
C6H6
What is the skeletal formula of a benzene ring?
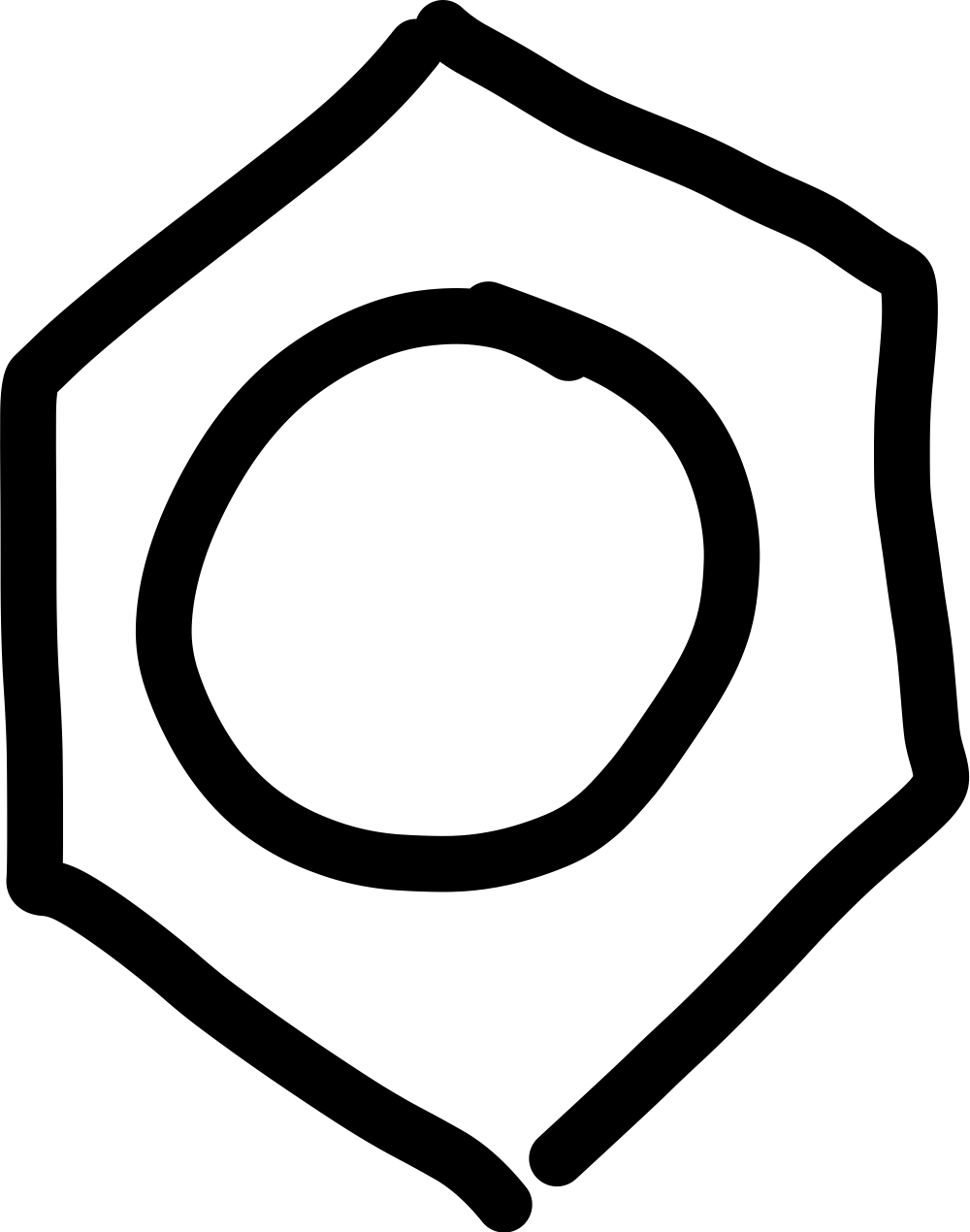
What is the displayed formula of a benzene ring?
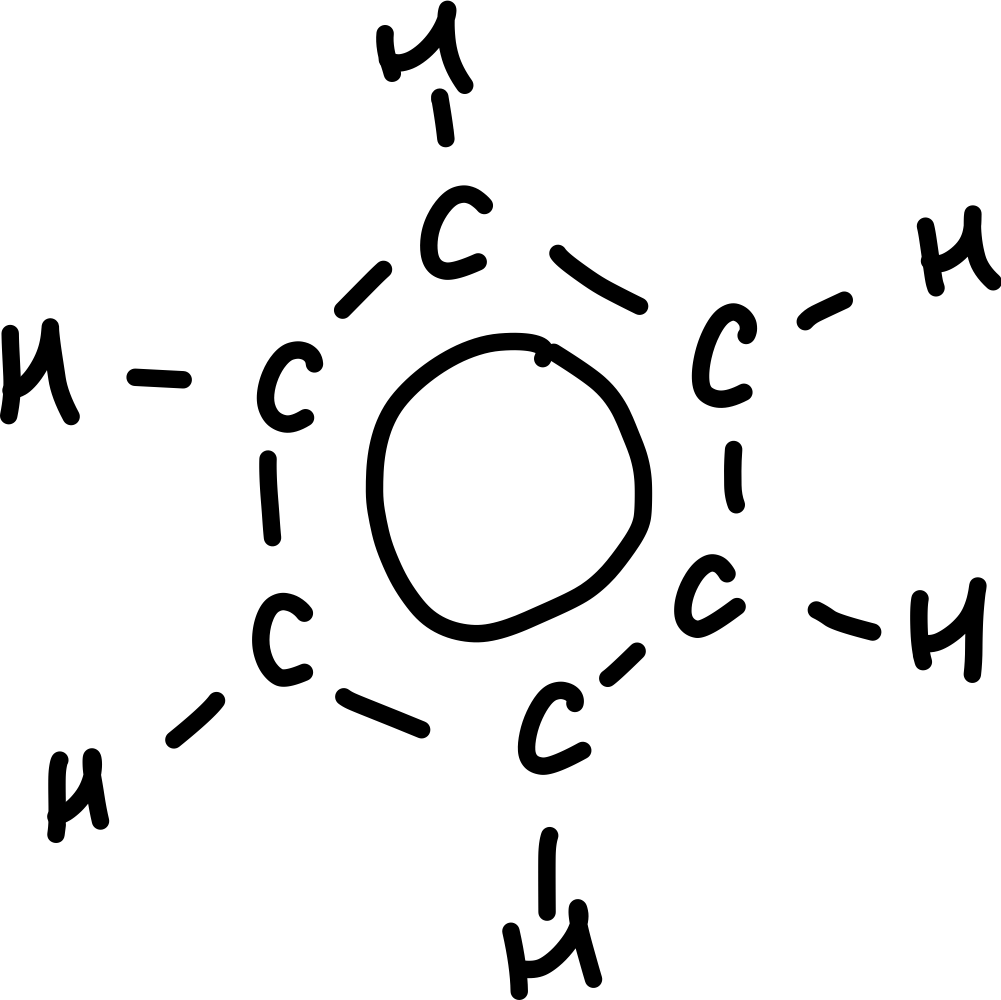
What is the empirical formula of benzene
CH
How are benzene rings with a halogen group named?
halo-benzene
How are benzene rings with an alkyl group named?
alkyl-benzene
How are alcohols with a benzene ring named?
phenyl-alcohol
How are benzene with a ketone group named?
phenyl-ketone
What is a nitro group?
-NO2
How are arenes with a nitro group named?
nitrobenzene
How are arenes with a halogen and alkyl group named?
halo-alkyl-benzene
How is an arene with a carboxylic acid named?
benzoic acid
What is an amine group?
-NH2
How are arenes with an amine group named?
phenylamine
How are are arenes with an OH group named?
phenol
When is the phenyl-prefix used when naming arenes?
when the benzene ring is attached to a alkyl group with 7 or more carbons or that contains a functional group
When is the benzene suffix used when naming arenes?
attached to an alkyl group woth less than 7 carbons or directly attached to a group
What are the exceptions when naming arenes?
phenol
benzoic acid
benzaldehyde
phenylamine
What is the name of C6H4OHNO2?
2-nitro-phenol
Who suggested that the structure of benzene is the same as triene?
Kekule von Stradonitz
What is the Kekule structure of benzene?
triene
Give 3 pieces of evidence that suggest the Kekule structure of benzene is incorrect?
resistance to reacting with bromine
bond lengths
enthalpy change of hydrogenation
Does benzene decolourise bromine at room temperature?
no
Why doesn’t benzene react with bromine?
it doesnt contain C=C bonds
How can the bond lengths of benzene be found?
xray diffraction analysis
Describe the bond lengths in benzene?
all C-C bond lengths are the same
the C-C bond length is between that of C-C and C=C length
What do the bond lengths of benzene suggest?
benzene does not consist of alternating, isolated C-C and C=C bonds
Compare the enthalpy change for the hydrogenation of benzene and the hydrogenation of triene?
the enthalpy change for hydrogenation of benzene is less exothermic than predicted
What does the enthalpy change for the hydrogenation of benzene suggest?
benzene does not contain 3 C=C bonds and is more stable than expected
Compare the stability of benzene and triene
benzene is more stable
What is the theoretical enthalpy change for hydrogenation of triene?
-360
What is the enthalpy change for benzene?
-208
What is the correct structure of benzene?
the delocalised structure
Describe the delocalised structure of benzene
cyclic molecule
shape around each C molecule is trigonal planar (120)
sigma bonds between each carbon atom
a pi system above and below the plane of carbon rings that consists of 6 delocalised electrons
What is the shape and around each C atom of benzene?
trigonal planar, 120
What bonds exist between the C atoms and CH atoms?
sigma bond
How are sigma bonds formed?
single overlap of orbitals directly between the nuclei of C atoms
How is rge pi system of a benzene ring formed?
formed by the sideways double overlap of p orbitals on each c atom on the carbon ring
How many delocalised electrons does the benzene pi system consist of?
6
Describe the delocalised electrons in the benzene system?
they are mobile across all 6 carbon atoms on the benzene ring
What are the similarities between the pi bond in cyclohexene and the pi system in benzene
pi bond formed in the same way
pi bond above and below the plane
What are the differences between the pi bond in cyclohexene and the pi system in benzene
pi system made up of 6 delocalised electrons while pi bond made up of 2
pi system - electrons are delocalised, pi bond- electrons are localised
What are elecrophiles?
electron pair acceptor
Why does benzene attract elecrtophiles?
it has an electron-rich pi system
Does the pi system in benzene make it more or less stable?
more stable
What does benzene maintain when it takes part in chemical reactions?
its delocalised pi system
What type of reaction does benzene undergo?
electrophilic substitution
Why is cyclohexene more reacrive toward electrophiles than benzene?
the pi bond in an alkene is more electron dense than the pi system in benzene
the 2pi electrons in an alkene are localised bwteen 2 C atoms while the 6pi electrons in a benzene are delocalised between 6 C atoms
the pi bond in alkene is sufficiently electron dense to induce a dipole in Br2 and so it attracts electrophiles more densely
Define electrophilic substitution in benzene?
a H atom on the benzene ring is replaced by an electrophile
the electrophile is attracted to the electron-rich pi system where it accepts a pair of electrons
forms a new covalent bond with a C atom if the ring
What does the electrophile do to benzene?
accepts a pair of electrons
What is formed when the electrophile accepts a pair of electrons from the benzene pi system?
a new covalent bond with a C atom of the ring
Draw the general mechanism of electrophilic substitution in benzene
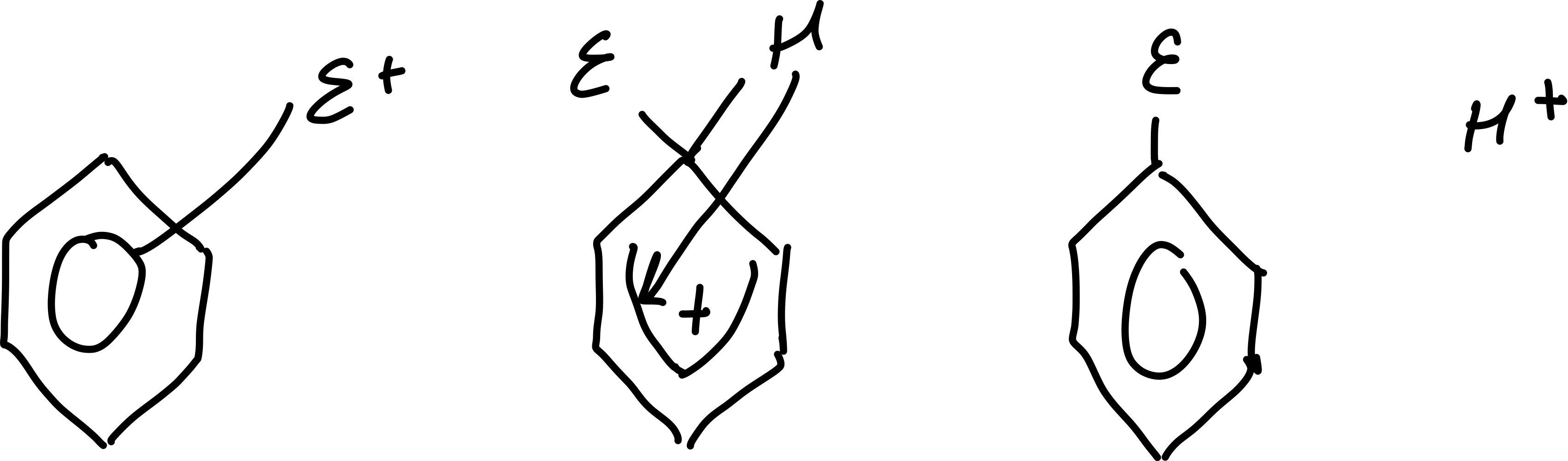
How many electrons are involved in the pi bonding in the intermediate of electrophilic substitution?
4
How many electrons are involved in the pi system of the organic product of electrophilic substitution?
6
Why is the intermediate of electrophilic substitution unstable?
the 4 electrons are delocalised about 5C atoms
What are the reagents for the nitrification of benzene?
concentrated nitric acid
What are the conditions for the nitrification of benzene?
concentrated sulfuric acid, heat under reflux at 50*C
If the temperature for nitrification of benzene was increased to 100*C what would happen?
further substitution
What is the overall equation for the nitrification of benzene?
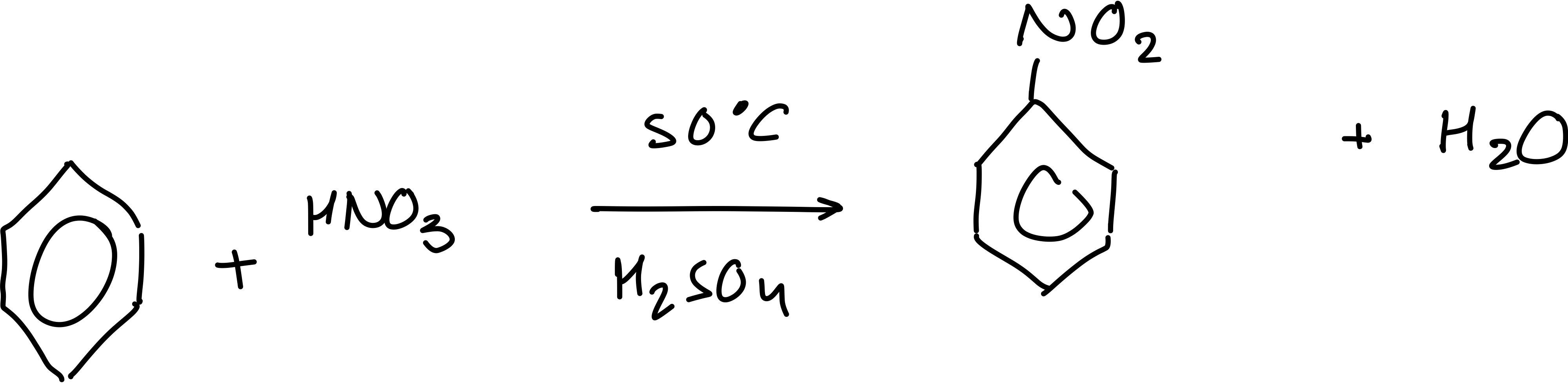
How many steps are involved in the nitrification of benzene?
4
What is the first step of the nitrifcation of benzene?
generation of the electrophile
What is the equation for the generation of NO2+
HNO3 + H2SO4 → NO2+ + H2O + HSO4-
Draw the reaction mechanism between benzene and NO2+
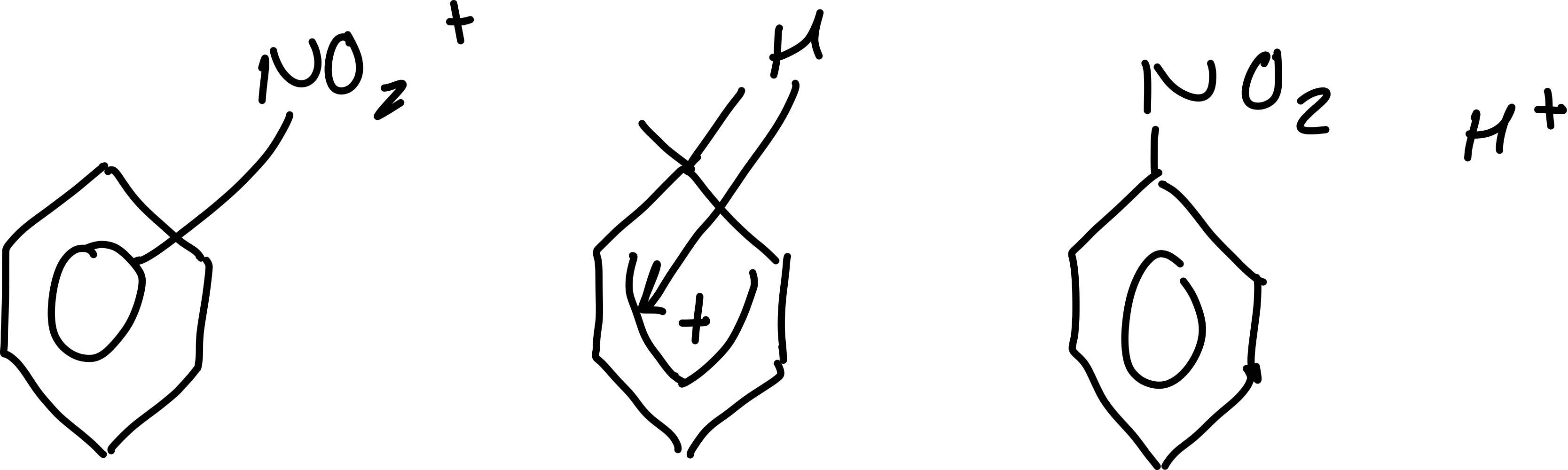
What is the final step in the nitrification of benzene?
regeneration of H2SO4
Write the equation for the regeneration of H2SO4
H+ + HSO4- → H2SO4
What are the reagents for the chlorination of benzene (and bromine)?
chlorine/bromine
What are the conditions for the chlorination of benzene (and bromine)?
FeCl3 / FeBr3
Write the overall equation for the chlorination or bromination of benzene
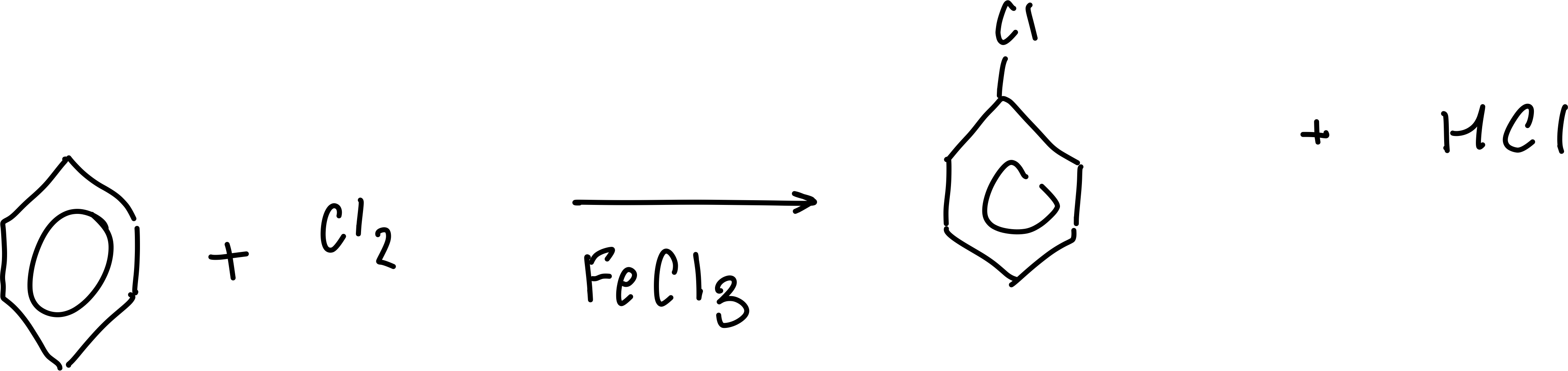
Write the generation of the Cl+/Br+ electrophile
Cl2 + FeCl3 → FeCl4- + Cl+
Draw the mechanism for the electrophilic substitution of Cl+/Br+ and benzene
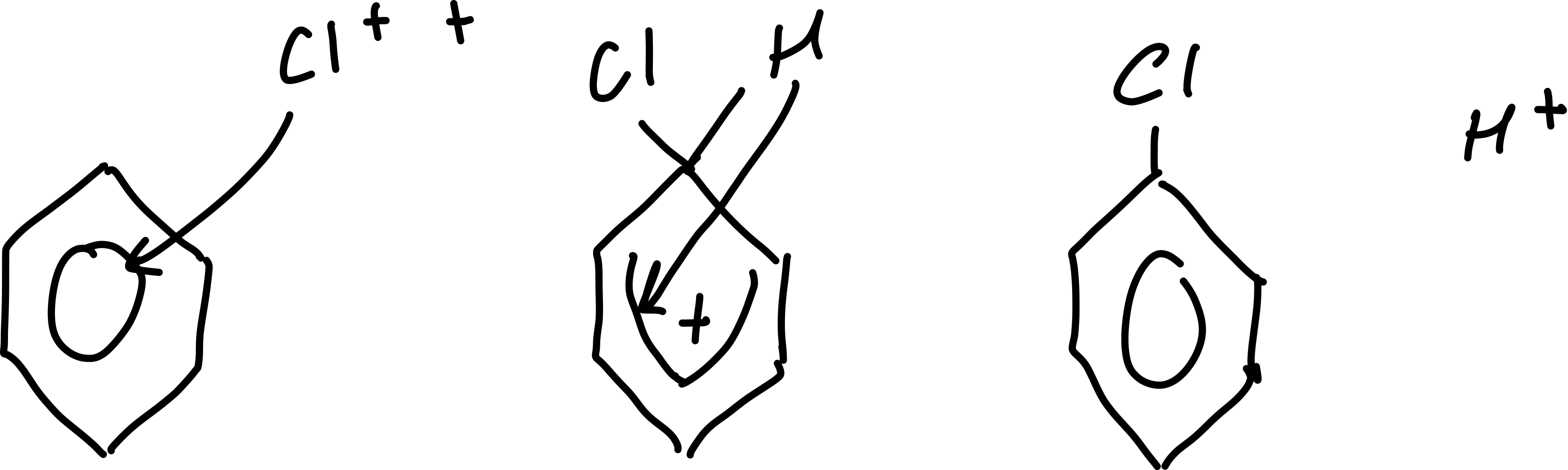
Write the equation for the regeneration of the FeCl3/FeBr3 catalyst
FeCl4- + H+ → FeCl3 + HCl
What are the reagents for the alkylation of benzene?
haloalkane
What are the conditions for the alkylation of benzene?
aluminium chloride
What is the equation for the alkylatio of benzne
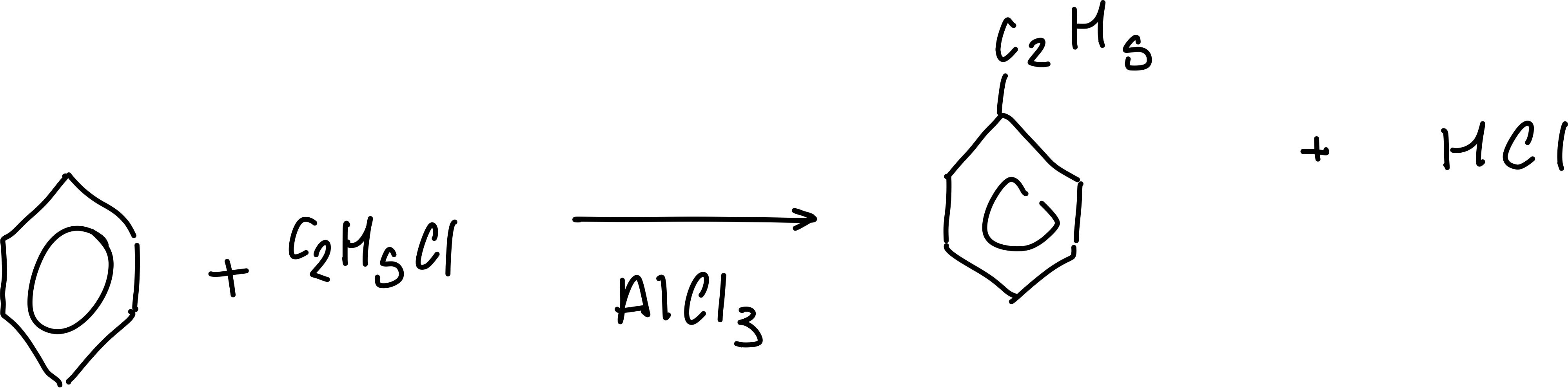
What is the equation for the acylation of benzene?
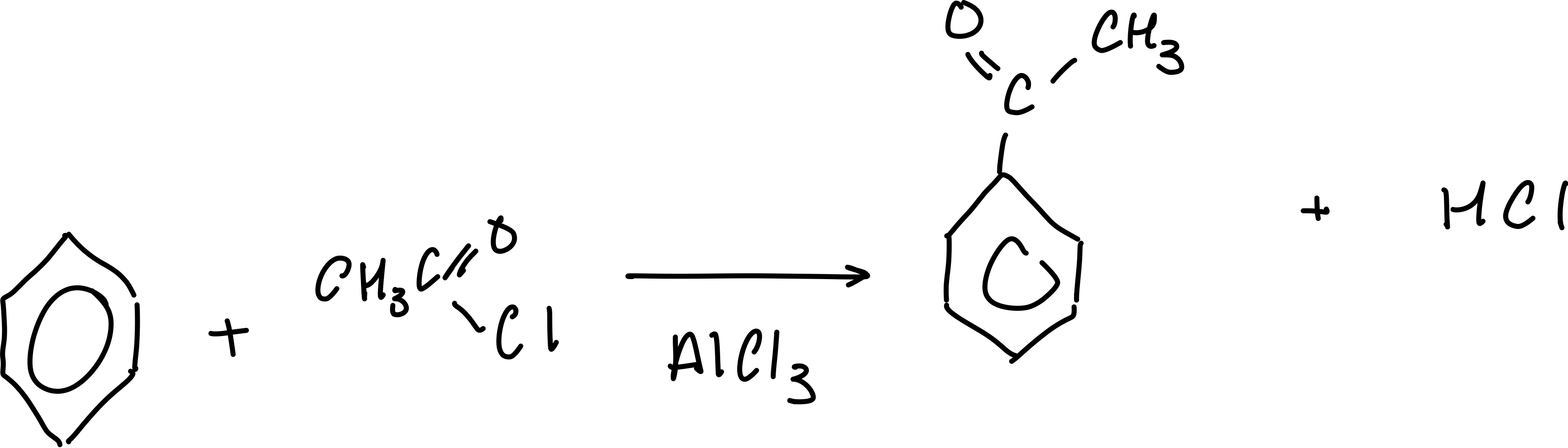
What are the reagents for the acylation of benzene?
acyl chloride
What are the conditions for the acylation of benzene?
aluminium chloride
What is the directing effect of an -OH group on benzene?
activates- 2,4 directing
What is the directing effect of a -NH2 group?
activates, 2,4 directing
What is the directing effect of an -NHR group?
activating 2,4 directing
What is the effect of an -NO2 group?
deactivating, 3 directing
What is the effect of an electron donating group?
they make the arene more reactive
Why do electron donating groups make arenes more reactive?
they make the pi system more electron dense, making the ring more susceptible to attack by electrophiles
What is the effect of electron withdrawing groups?
less reactive
Why do electron withdrawing groups make arenes less reactive?
they make the pi system less electron dense making the ring less susceptible to attack by electrophiles
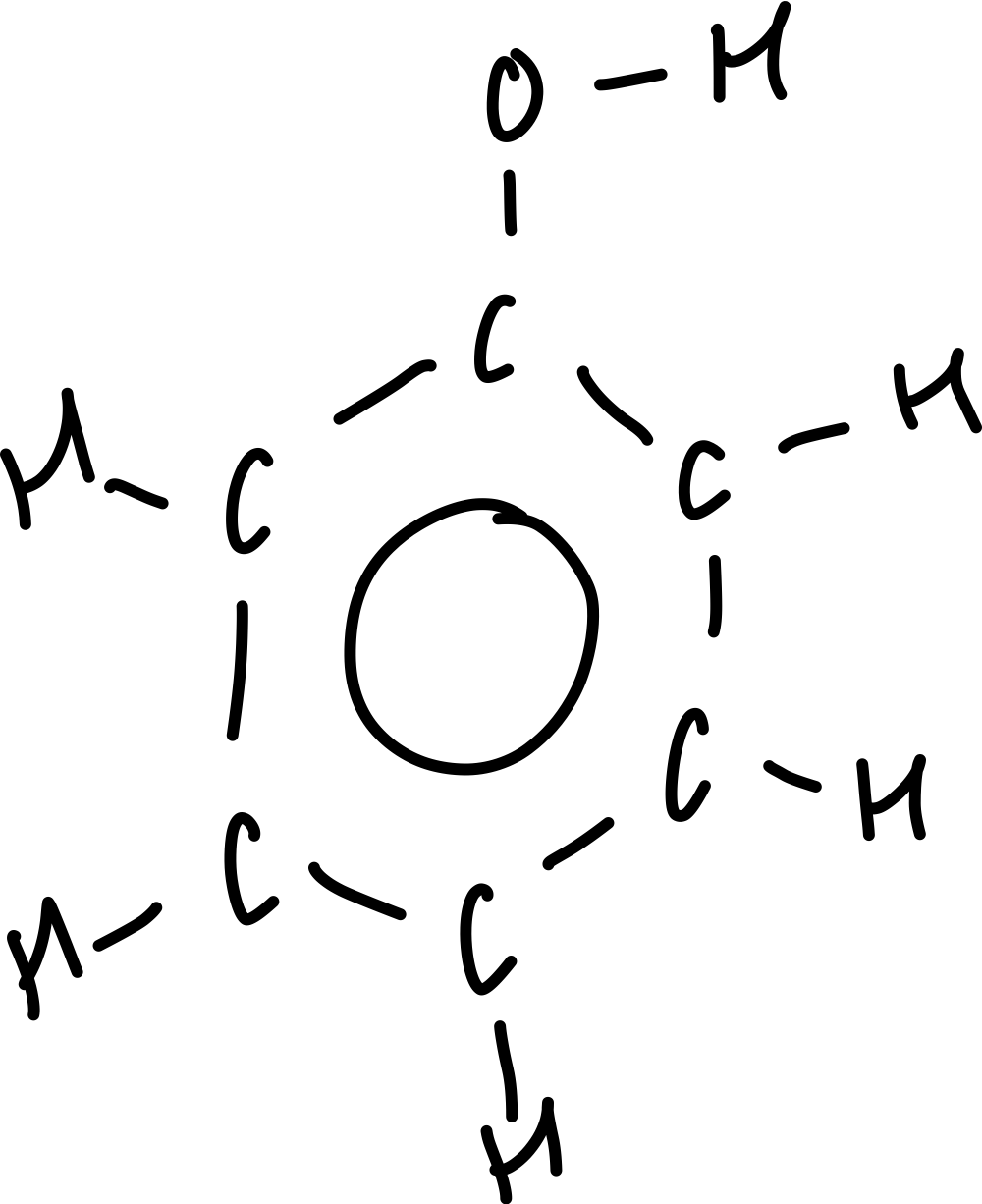
What is the molecular formula of phenol?
C6H6O
What is the displayed formula of phenol?
What is the skeletal formula of phenol?
What are common uses of phenol?
antiseptic
disinfectants
plastics
resins in paints
What is the use of trichlorophenol?
antiseptic
What is the use of thymol?
antiseptic (thyme)
what is the use of bisphenol A?
plastics and resins
What state is phenol at room temperature?
solid
Why is phenol solid at room temperature?
it forms hydrogen bonds with itself which require lots of heat energy to overcome
Why is phenol soluble in water?
it forms hydrogen bonds with water
What type of reaction does phenol undergo?
electrophilic substitution
Compare the reactivity of benzene and phenol?
phenol is more reactive
What are the reagents for the nitrification of phenol?
dilute nitric acid