Lewis Structures
0.0(0)
Card Sorting
1/6
Earn XP
Description and Tags
Study Analytics
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
7 Terms
1
New cards
CBr4 - Carbon tetrabromide

2
New cards
Carbon dioxide (CO2)
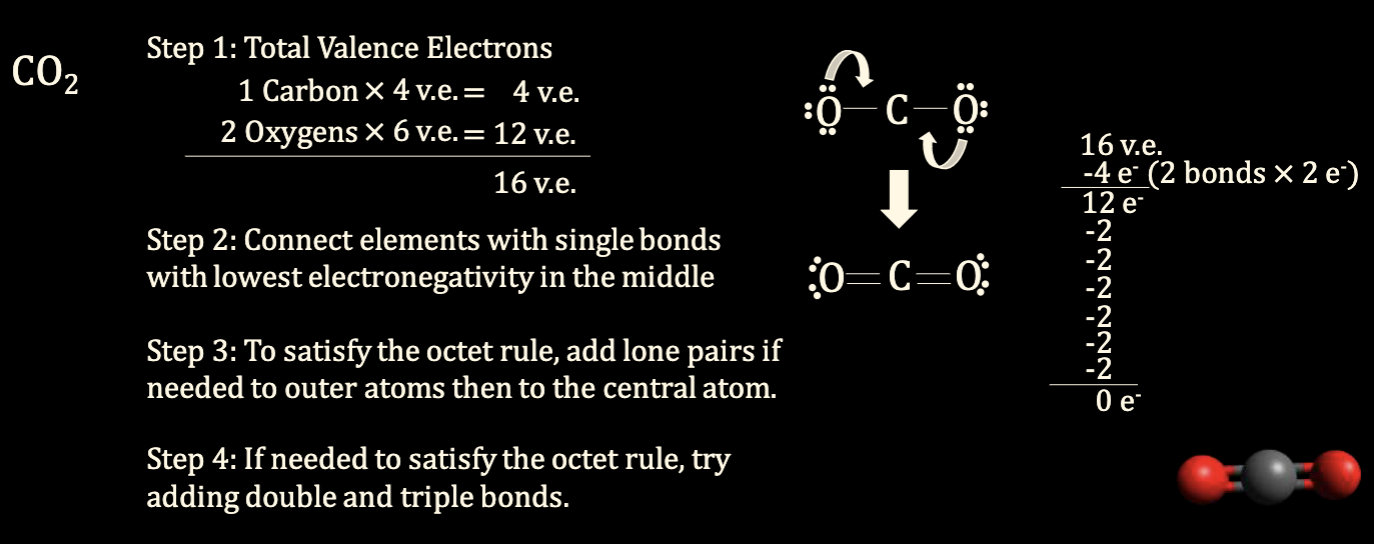
3
New cards
C2H5Br2 Ethylene Dibromide

4
New cards
C2H3Br - Bromoethyne
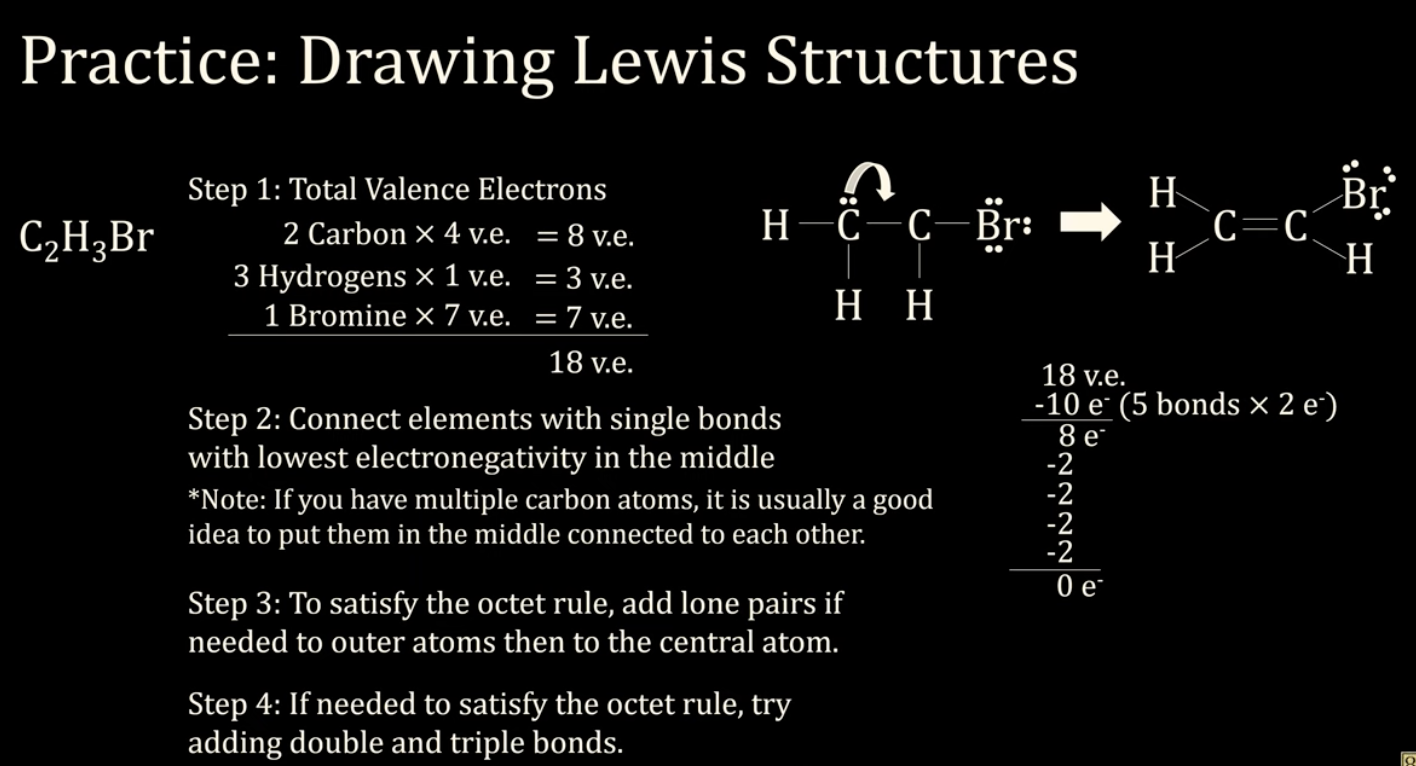
*If we have 2. carbons, we typically place them in the center. Also we need to distribute H around them.
5
New cards
HO —

Lewis structure for Hydroxide with negative charge - to communicate a charge, put the structure into brackets
6
New cards
Electron Deficiency - BF3 and NO examples

7
New cards
Expanded Valence Configuration
