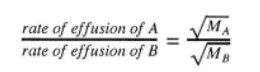Chemistry Gas Laws
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
Observed Properties of Gases
Cats Find Delicious Mice Playfully
May be compressed
Fill containers uniformly
Low density (due to spacing of particles)
Can be mixed (usually a mixture)
Uniform pressure in all directions
Kinetic Molecular Theory of Matter (belief of energy of motion and building block scale)
Lazy Elephants In Soft Valleys
Molecules move in straight lines
Molecules collide with each other without losing energy (elastic collisions)
Molecules are independent (no attraction forces between)
Molecules are spread out
Volume of molecules are small in comparison to space occupied
Relate to properties: Molecules move in straight lines
Fill containers uniformly
Uniform pressure in all directions
Relate to properties: Elastic collisions
Fill containers uniformly
Relate to properties: Molecules are independent
Can be mixed
Relate to properties: Molecules are spaced out
May be compressed
Relate to properties: Volumes of molecules is small compared to space occupied
May be compressed
Low density
Ideal Gas
Follows KMT + Predictions
Theoretical
Real Gas
Real gases deviate from predictions greatly
Everything we use
Low temps or High pressure
Pressure
Pressure is the amount of force applied over an area.
P= F/A
Experiments with gas usually involve controlling or measuring its…
Quantity
Volume
Pressure
Temperature
Quantity is measured in..
n=moles (often held constant)
Volume is measured in…
L, mL, m³,dm³ L=dm³
Pressure is measured in…
kPa, mmHg,torr, atm
Use barometer to measure

STP
Temperature: 0 °C (273.15 K)
Pressure: 1 atmosphere (1 atm) or 101.3 kPa
Standard Molar Volume of Gas
At STP one mole of ANY gas is 22.4L
Ideal Gas Law
PV=nRT
What is temperature a measure of?
Average kinetic energy
Absolute Zero
Theoretically when molecules stop moving
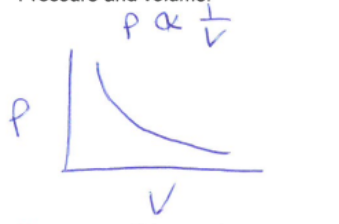
Boyle Law
Constant temp and quantity
Pressure and volume are inversely related
Gas-Lussac Law
Pressure and temperature are proportionally related
Constant volume and quantity

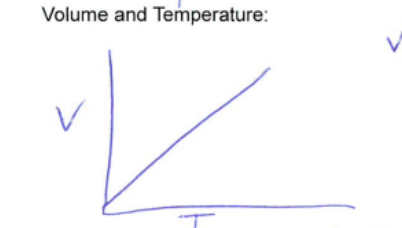
Charles Law
Constant pressure and quantity
Volume and temperature are proportionally related.
How to add molar mass into the combined gas law?
n=mass/molar mass
PV=m/mmRT
How to solve for density
Gas= PM/RT
When is ideal gas law used?
Solve for single variable
Gas density (g/L)
Solve for molar volume (L/mol)
Partial Pressures
The total pressure of a mixture of gasses is made up from the sum of partial pressures of the component gases.
Diffusion
The gradual mixing of two or more gases due to their spontaneous, random motion.
Effusion
The molecules of a gas confined in a container randomly pass through a tiny opening in the container.
Rate of effusion, which effuses faster?
Lighter gases effuse faster
Rate of effusion formula