Transition metals
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
Transition element
Element forming one or more ions with incomplete d-subshell
D block element
Element where the d sub-shell/ d-orbital has the highest energy electron
Ligand
Donates a lone pair to form a coordinate bond with a metal ion
Complex
A central metal atom or ion surrounded by ligands joined by coordinate bonds
Coordinate number
The number of coordinate bonds to the central metal or ion
Ligand structures
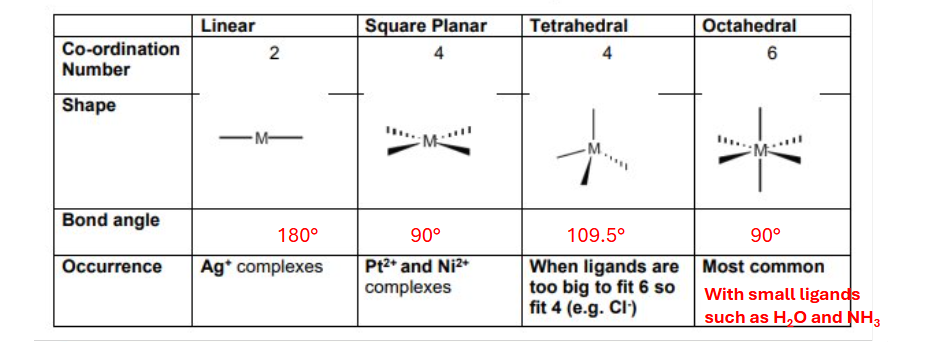
Monodentate ligand
Forms one coordinate bond to the transition metal ion using one lone pair of electrons
H2O: :NH3 :Cl- :CN- :OH- :CO
Cis-Platin: Anti-cancer drug
Advantages - Kills cancer cells
Disadvantages - Hair loss and fertility issues
Bidentate ligands
Donates two lone pairs to form two coordinate bonds
How haemoglobin transports oxygen around the body via ligand substitution
O2 co-ordinately bonds with Fe2+ and then once transported, it is replaced by H2O or CO2
Why carbon monoxide is toxic
It forms a stronger coordinate bond than oxygen. This prevents the transport of oxygen around the body
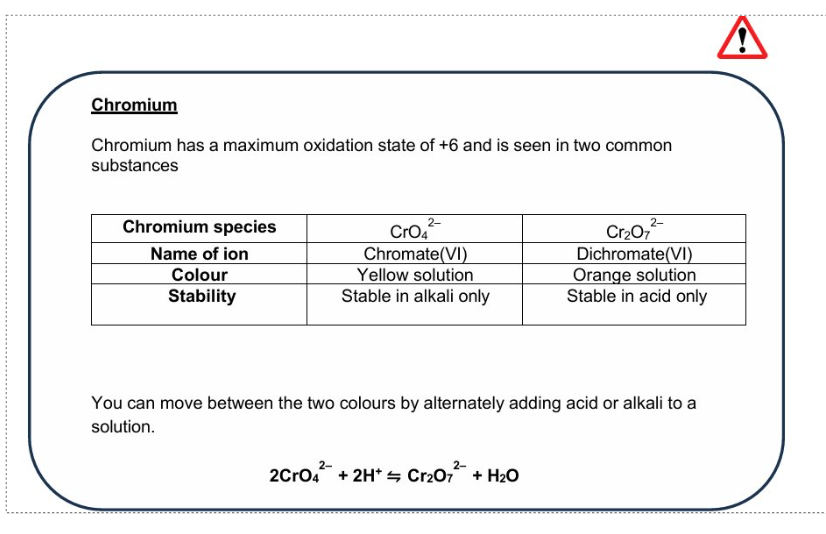
Dichromate ions oxidation states

Chromium oxidation states
Manganese oxidation state

What is a catalyst
A substance that increases the rate of reaction but isnt used up in the reaction
Why are transition metals good catalysts
They have variable oxidation states
Health risks of using transition metals
Long term exposure to copper can cause kidney and liver damage
Exposure to manganese can cause psychiatric problems