Gibbs free E change
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
what is the Gibbs free E change and what does it tell us?
balance between entropy and enthalpy
determines the feasibility of a reaction
give the eqn for the Gibbs free E change:
ΔG = ΔH - TΔS
for a reaction to be feasible, what must the ΔG value be?
0 or-ve
what are the units of ΔG? what does this mean when we are using the ΔG eqn?
kJ mol-1
must /ΔS by 1000 to convert into kJ K-1 mol-1

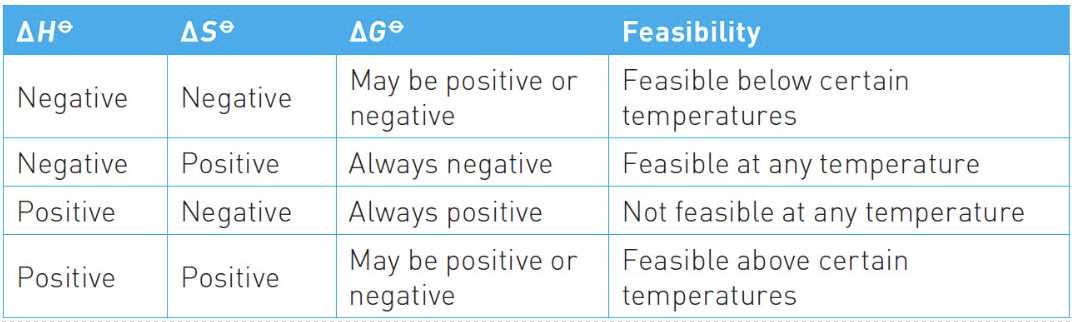
can you complete this table? (1st 3 columns - +ve/-ve)
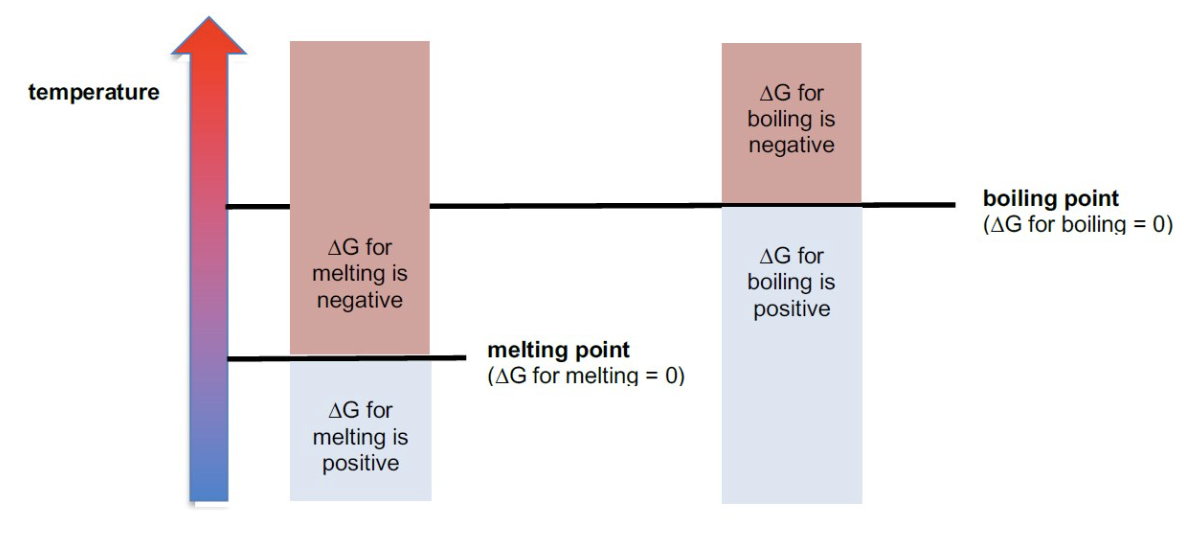
what is the value of ΔG during a change of state?
0 kJ mol-1
for temperatures below the mpt/bpt, is ΔG +ve or -ve?
+ve
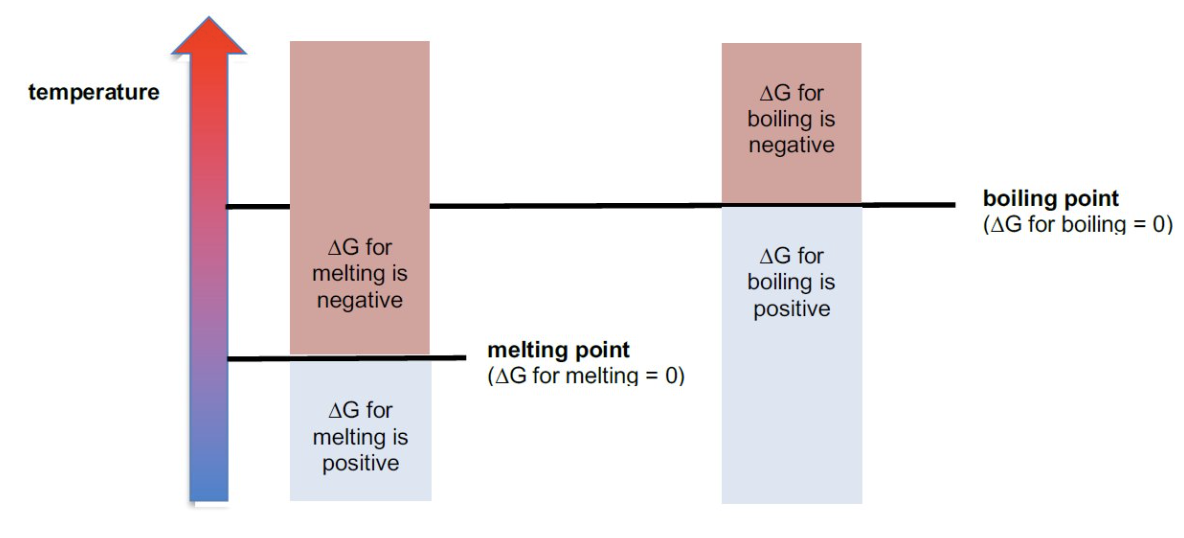
for temperatures above the mpt/bpt, is ΔG +ve or -ve?
-ve
how can we plot a graph of the Gibbs free energy?
eqn ΔG = ΔH - TΔS can be plotted as a straight line graph of ΔS against T (respectively: y = c + m(-x)
∴ gradient = -ΔS and the y intercept is
ΔH
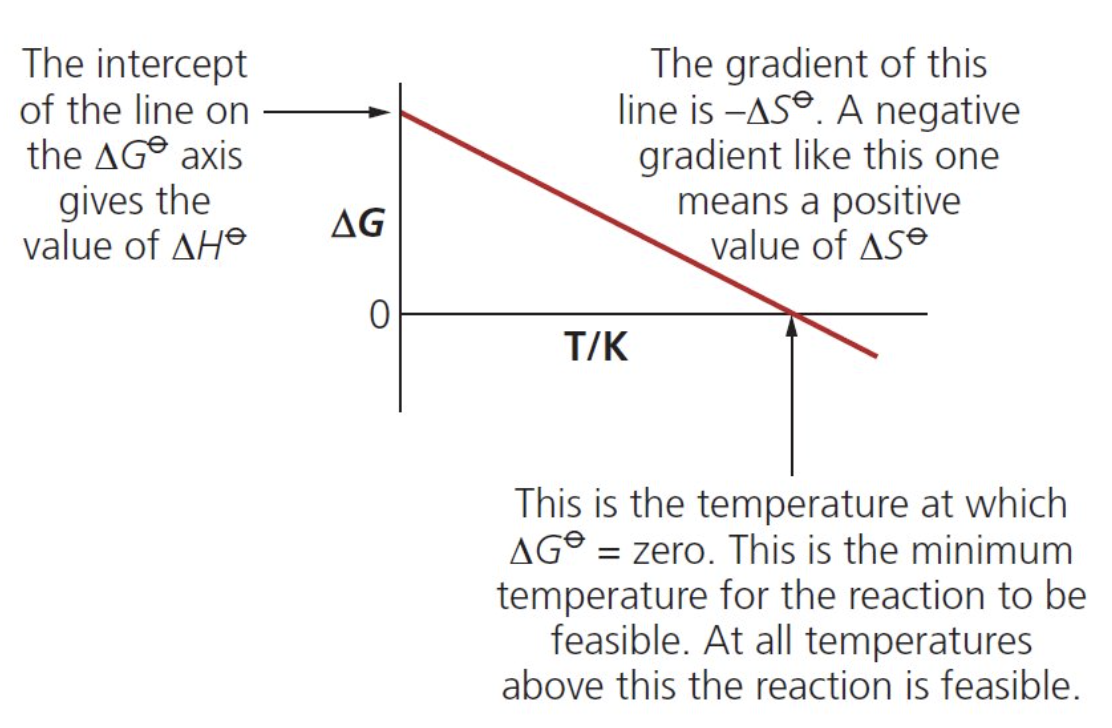
what does the x-intercept represent on a Gibbs-free energy graph?
temperature at which ΔG becomes 0 and so the reaction becomes feasible
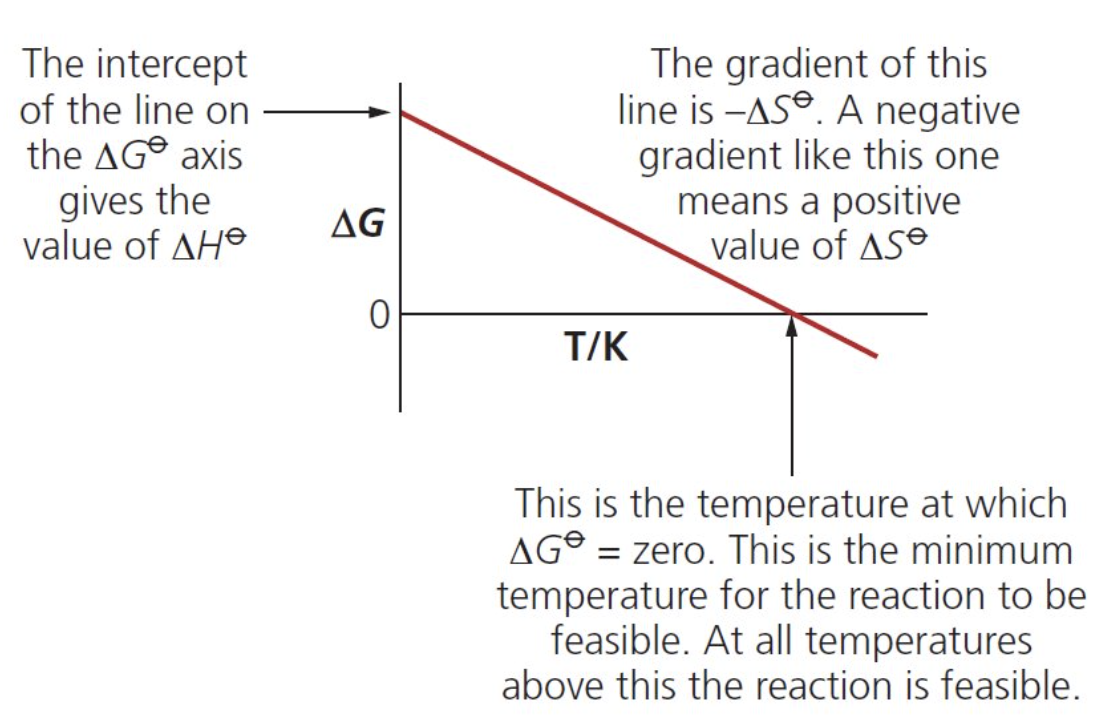
how can we determine whether a reaction is endo or exo based on its Gibbs-free energy graph?
when ΔS is +ve (gradient is -ve) = endo
when ΔS is -ve (gradient is +ve) = exo
give the eqn to calculate T derived from the Gibbs free E change:
T = ΔH / ΔS