Exam 1 Hong
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
What are the steps of the Drug Development Cycle
1) Discovery, 2-4 years
2) Preclinical, 1-2 years
3) IND
4) Clinical Trials, 4-6 years
5) New drug approval
6) Post-marketing surveillance, 1-4 years
Understand the role of the Food and Drug Administration (FDA) in the drug development and review process in the US
•Regulates pharmaceutical market
•Ensuring human safety & efficacy
•Federal food, drug and cosmetic act
Discovery
•Biology experts share knowledge of target and develop tools to further study
•Chem engages to identify chemical matter to interact with desired target
•ADME experts understand what body does to drug
•Toxicology experts for safety
•Pharmacuetical sciences- expert in formulation and delivery
Target identification then validation
→Clinical proof of concept (POC)= right target + right drug
Creation of New Drug Moecule
Canditiate selection stage→ basic material properties, in vitro cell testing, animal
Lead optimization
→Improve safety and efficiacy, SAR
Preclinical
Preclinical stage→Detailed material properties, in vitro safety testing, two animal species testing
PreClinical testing→ Therapeutic index Toxic Concen/Efficacous Concen
Drug substance scale up→ Make enough drug to use up through early clinical studies
IND Application (Investigational New Drug)
•Must be approved prior to First in Human clinical trial
•Contains: Acute toxicity in 2 species of animals, short term toxicity studies, pharmacological profile of drug substance
Phase 1 Clinical Trials
•FIH dosing to healthy volunteers
•Admin of single escalating doses of drug to small number of subjects
•Short term multiple doses
•10-15 volunteers
•Assess safety & PK
Phase 2 Clinical Trials
•First dosing to patients
•Fewer dosages studied than in Phase I
•First evaluation of efficacy
•Safety profile & PK monitored
•20-100 subjects studied
•Assess proof of concept
Phase 3 Clinical Trials
•Test effectiveness of drug for particular indication in patients
•Common effects documented
•Over 1000 subjects
•Finalize prescribing label
Phase 4 Clinical Trials
•Post marketing approval studies
•More info of effectiveness and safety
•300-30000 volunteers
New Drug Application
• Proposed labeling details for the products
• Results on safety & efficacy of the drug
• Results from clinical trials
• Details on long term studies and post marketing surveillance
• Method of manufacture of drug and quality control analysis
NDA requirements (New Drug Application)
Chemistry
Manufacturing
Controls
Labeling
Testing
Animal studies
Clinical studies
Bioavailability
ANDA requirements (Abbreviated New Drug Application)
Chemistry
Manufacturing
Controls
Labeling
Testing
Bioequivalence
Cmax
Effectiveness, Safety
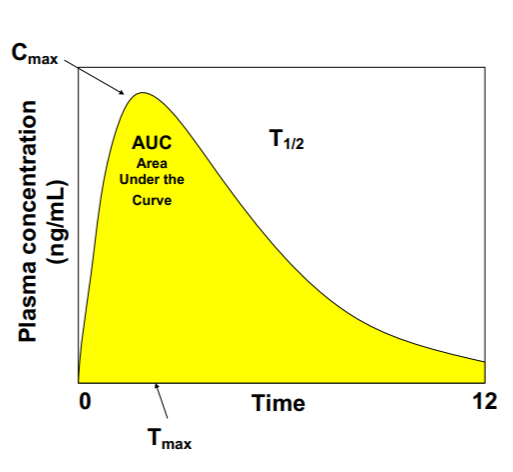
AUC
Exposure, effectiveness, bioavailability
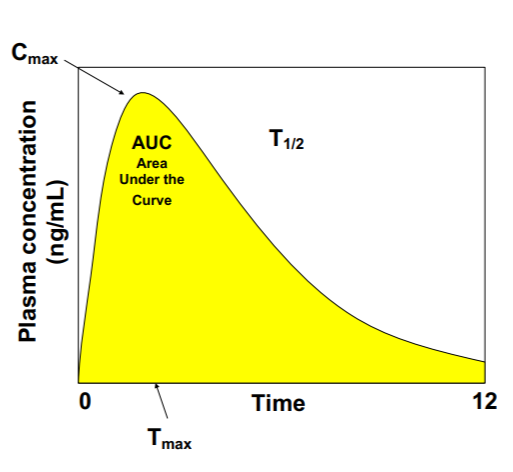
Tmax
Absorption rate, input rate
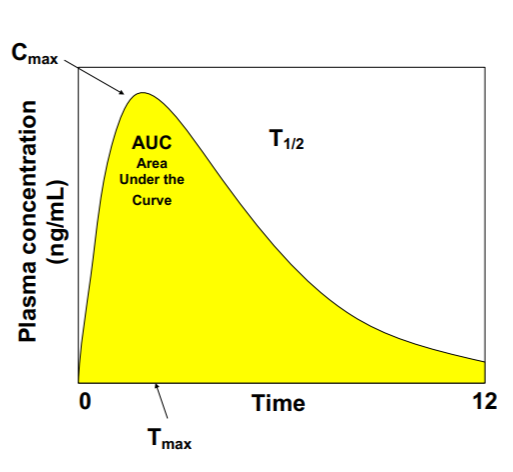
T ½
Elimination Rate
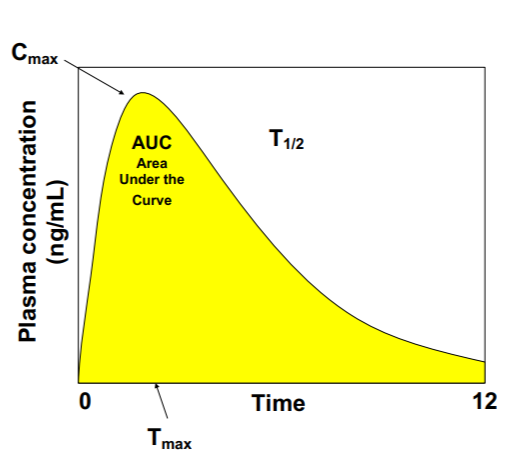
Bioavailability
•Rate and extent to which an active agent is absorbed and available at the site of action & yields a therapeutic response
•Often accepted that equilibrium exists between the concentration of drug at the site of action and concentration of drug in blood
How would a drug be 100% orally bioavailable
must be completely released from the dosage form into solution, completely stable in the GI fluids, pass through GI epithelium and undergo no first-pass metabolism in the gut wall or liver, e.g. caffeine
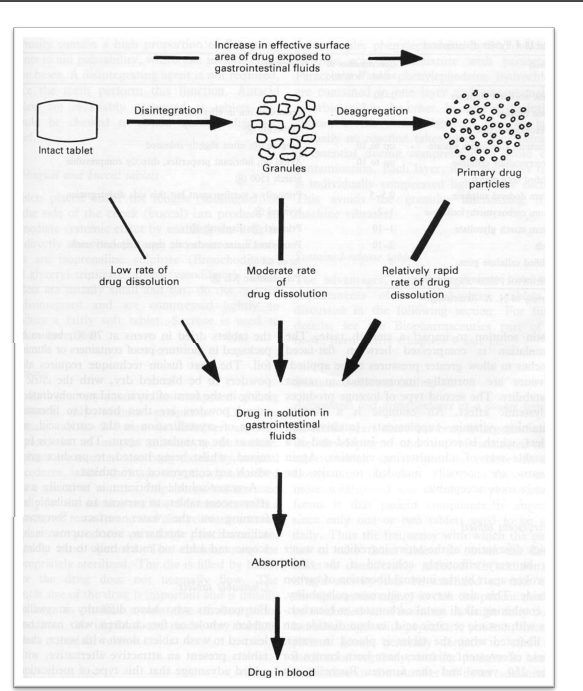
Describe the effect of surface area on dissolution
Increased surface area leads to more rapid rates of dissolution
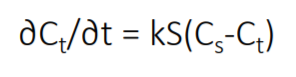
Noyes Whitney Equation
Calculating dissolution rate
Cs= solubility of drug
Ct= drug concentration in bulk fluid
k= dissolution rate constant
S= surface area
Lipinski’s Rule of 5
•Molecular weight <500 g/mole
•logK <5
• <5 H bond donors
• <10 H bond acceptors (N &O)
Roles of Excipients
• Promote manufacturability
• Promote Stability
• Promote bioavailability
•Promote accurate dosing
• Promote patient acceptance and convenience
•Mostly inert
Categories of drug delivery
•Immediate release
•Delayed release of one dose then full release (ex; enteric coated)
• Prolonged release to increase intervals between dosing
•Controlled release: spatial and temporal control
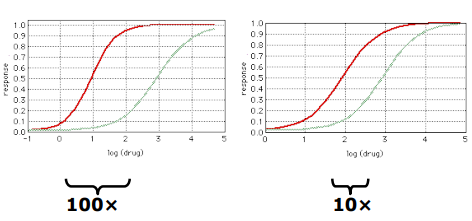
Therapeutic Index
Degree of separation between toxic and therapeutic doses
Types of Systemic controlled drug delivery
• Oral route
• Intravenous route
• IM or SC
• Transdermal
• Sublingual or buccal
• Intranasal
• Pulmonary
What is the goal of Controlled local drug delivery
The goal is minimum systemic absorption and optimal drug concen reaches local site