BIOC 4331 Lecture 33-35 (OxPhos)
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
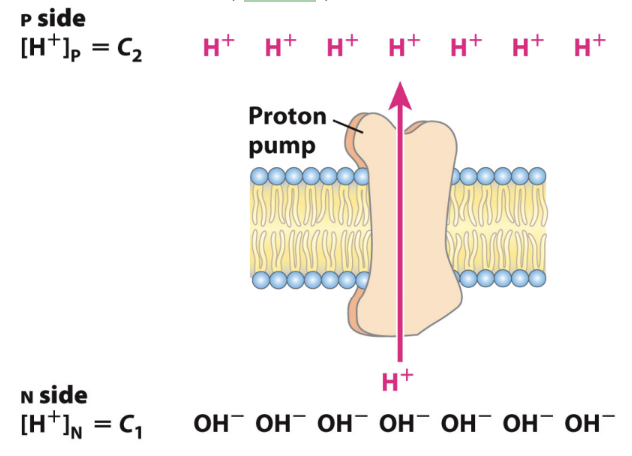
What is the proton-motive force (PMF)?
The electrochemical gradient of H+ across the inner mitochondrial membrane.
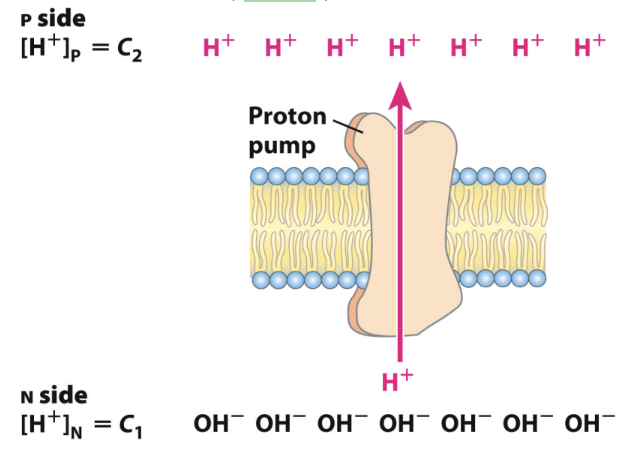
What are the two components of the proton-motive force?
A chemical gradient (ΔpH): Concentration difference of H+ (more acidic on P side)
An electrical potential (Δψ): Separation of charge across the membrane without moving a counterion (more + on P side)
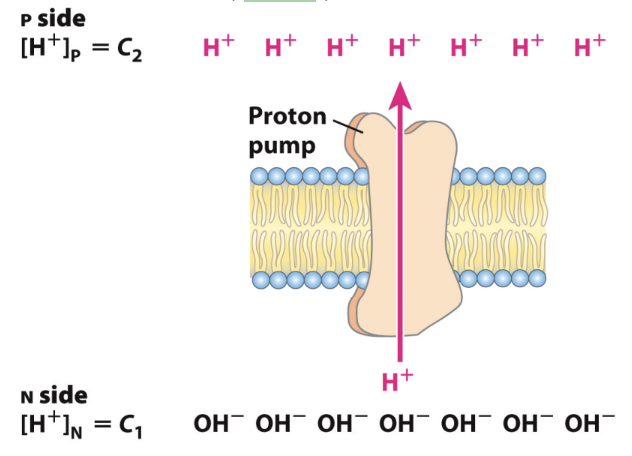
What is the chemical component of PMF?
Higher [H+] on the P side than the N side.
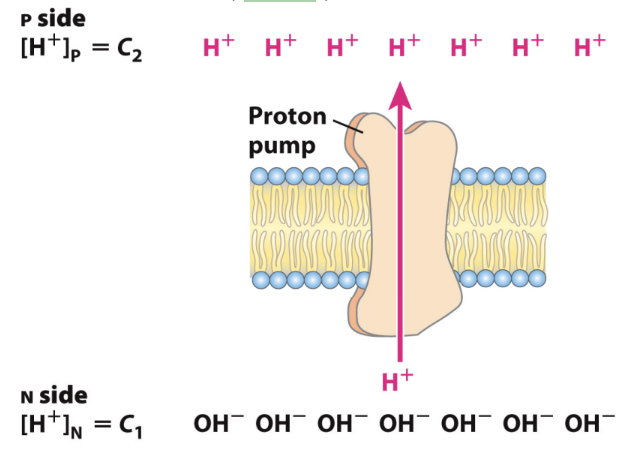
What is the electrical component of PMF?
The P side is more positive and the N side is more negative.
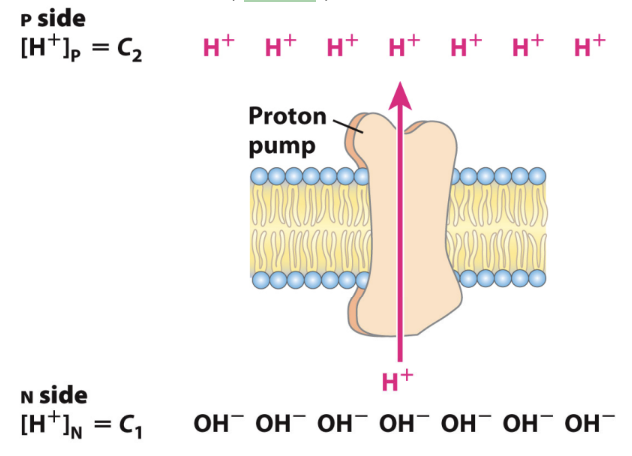
Why is the N side relatively negative?
Because protons are pumped out without a counterion.
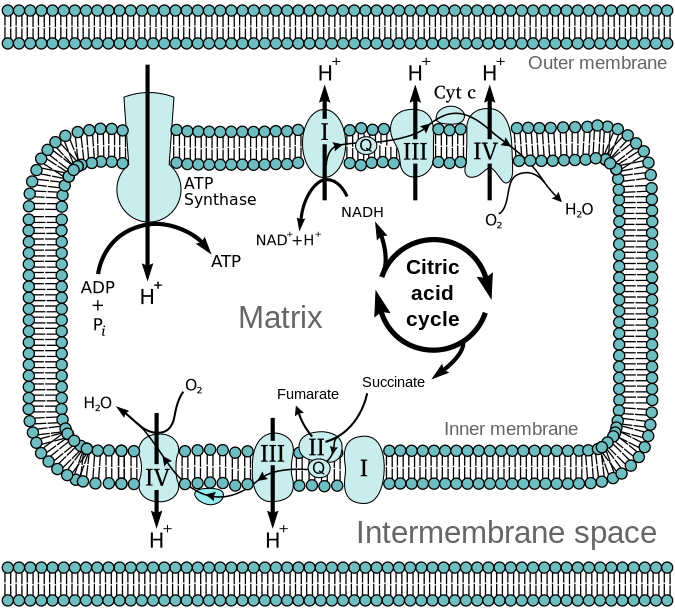
What is the chemiosmotic model?
The idea that electron transport creates a proton gradient, and the flow of protons back down that gradient drives ATP synthesis.
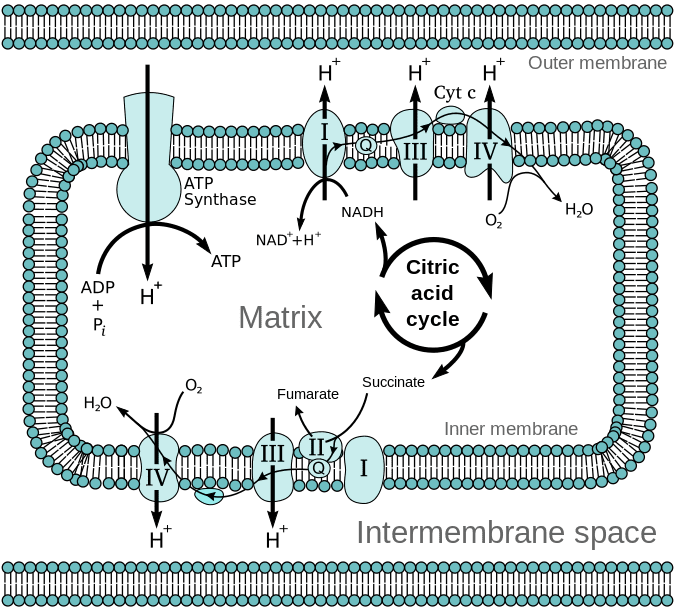
Who proposed the chemiosmotic model?
Peter Mitchell.
Why is ATP synthesis in oxidative phosphorylation not a direct group-transfer reaction?
Because ATP is made using energy from proton flow down the electrochemical gradient, not by direct transfer from a high-energy phosphate donor.
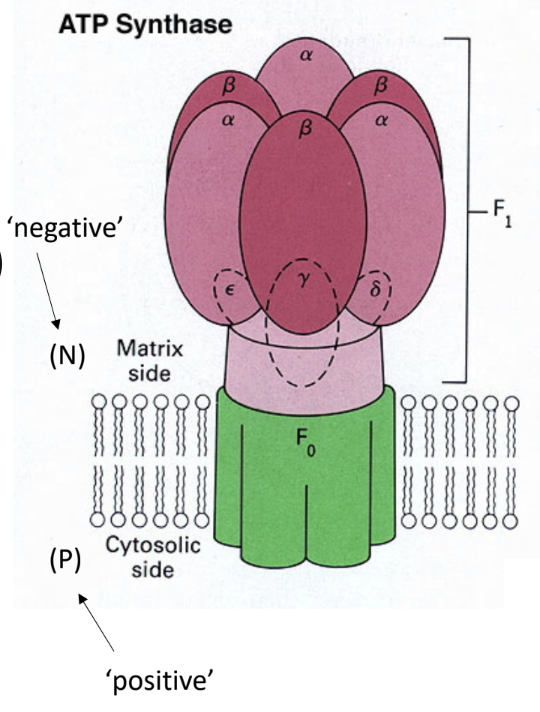
What enzyme uses the proton gradient to make ATP?
ATP synthase.
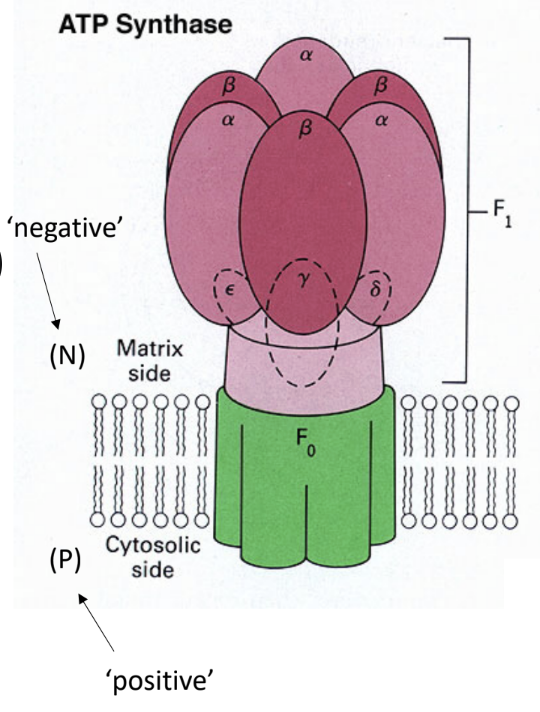
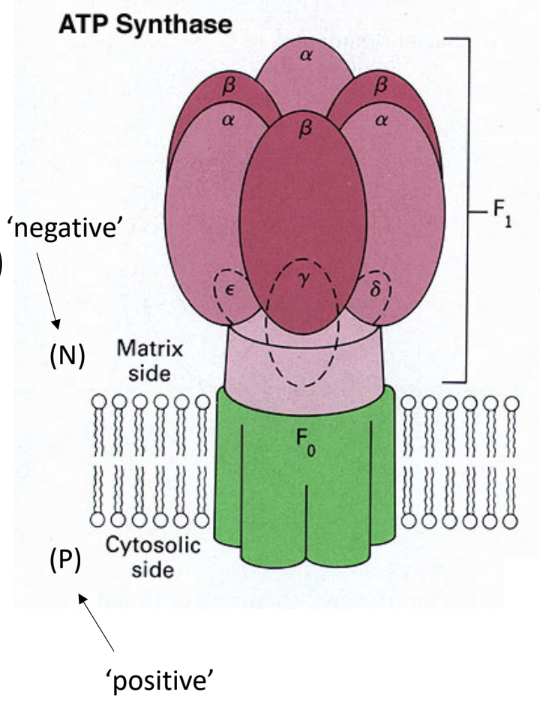
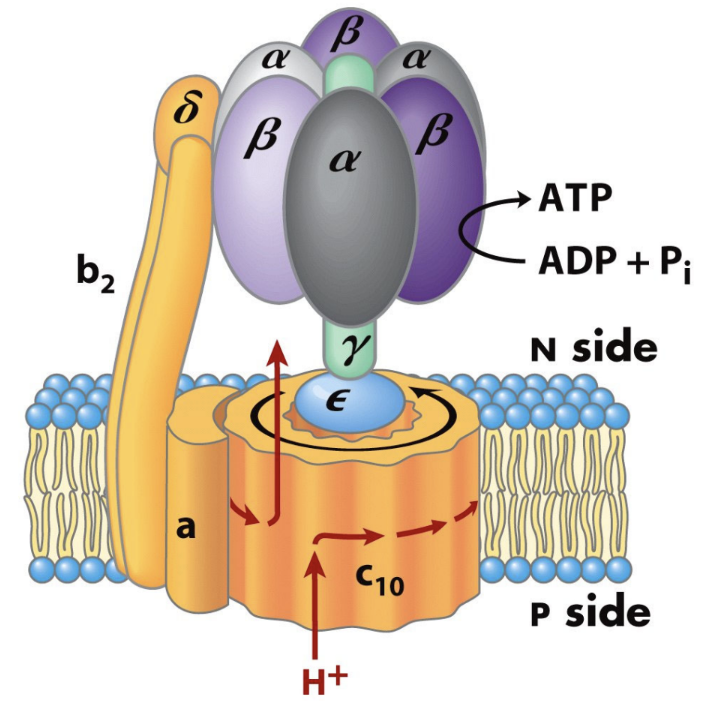
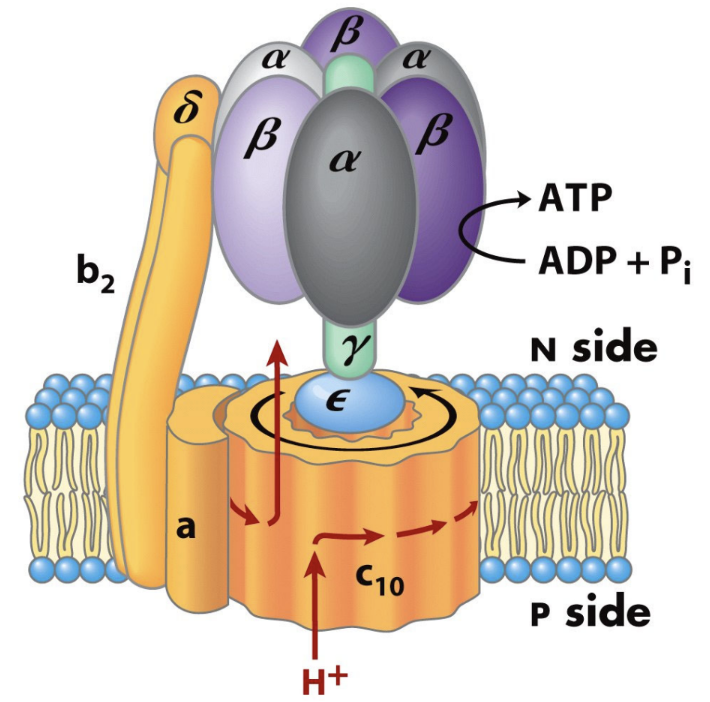
What are the two major parts of ATP synthase?
F0
F1.
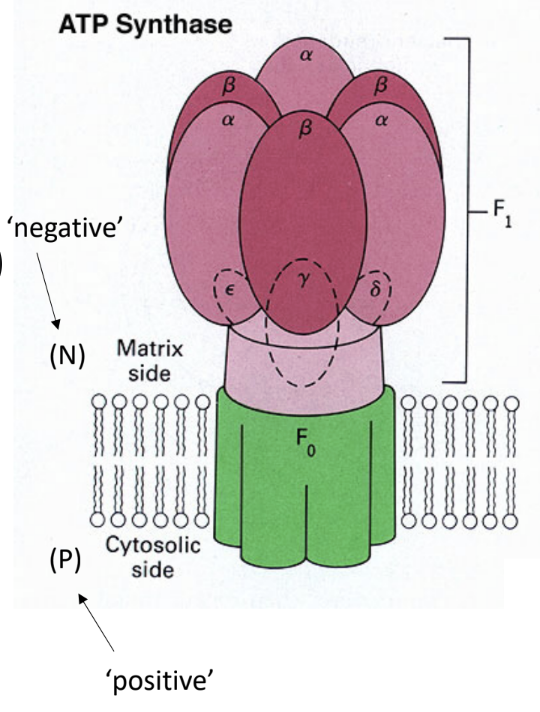
What does F0 do?
It forms the membrane proton channel/rotor.
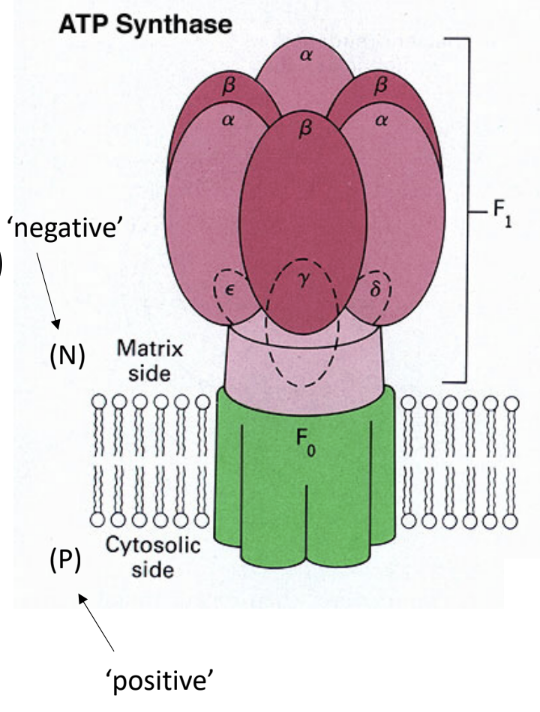
What does F1 do?
It contains the catalytic sites that synthesize ATP.
What happens when protons flow through F0?
The rotor and central shaft rotate.
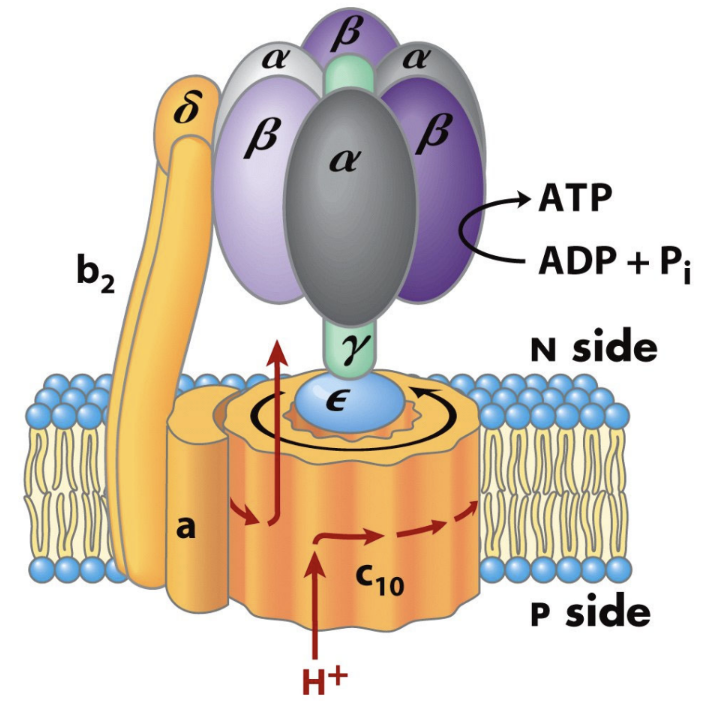
Which ATP synthase subunit acts as the rotating central shaft?
The γ subunit.
What effect does γ rotation have on F1?
It drives conformational changes in the β subunits.
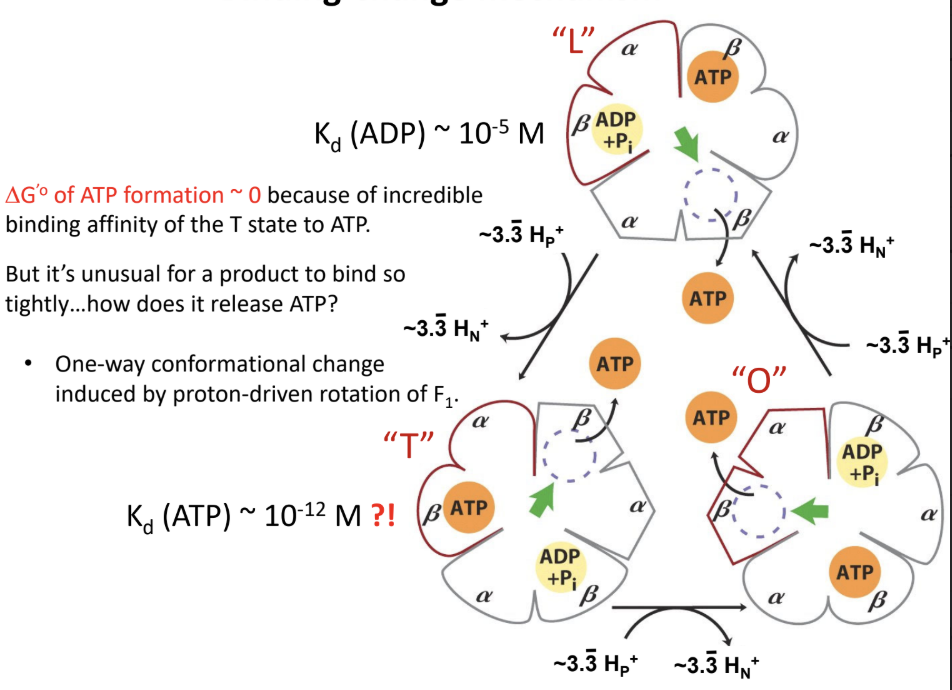
What is the binding-change mechanism?
Rotation-driven conformational changes cycle the β subunits through different states that bind substrates
Form ATP
Release ATP
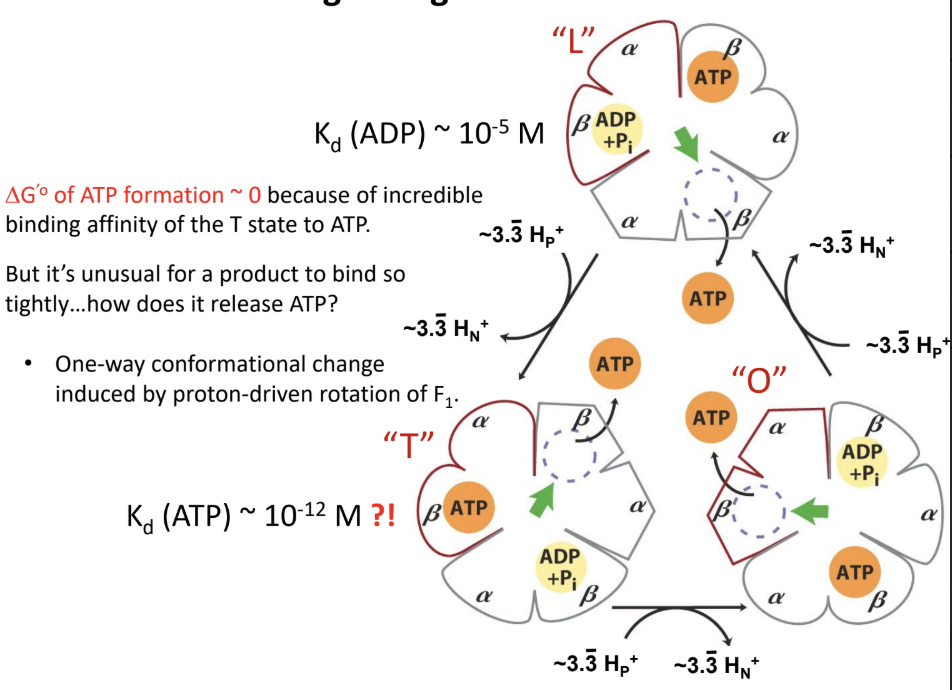
What are the three conformational states of the β subunits in ATP synthase?
L (loose)
T (tight)
O (open)
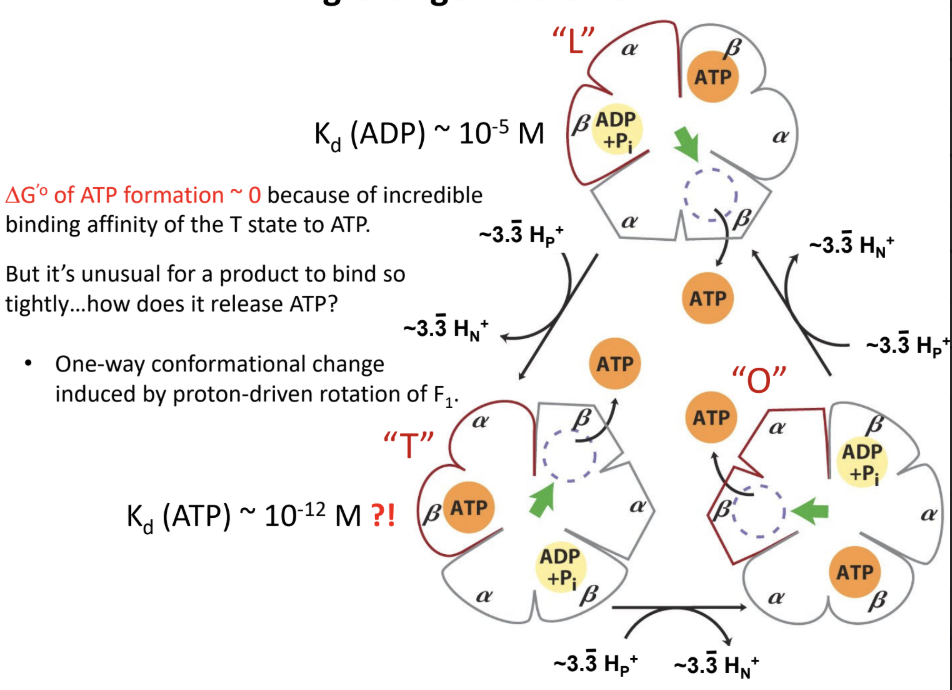
What does the L state do?
Binds ADP and Pi loosely.
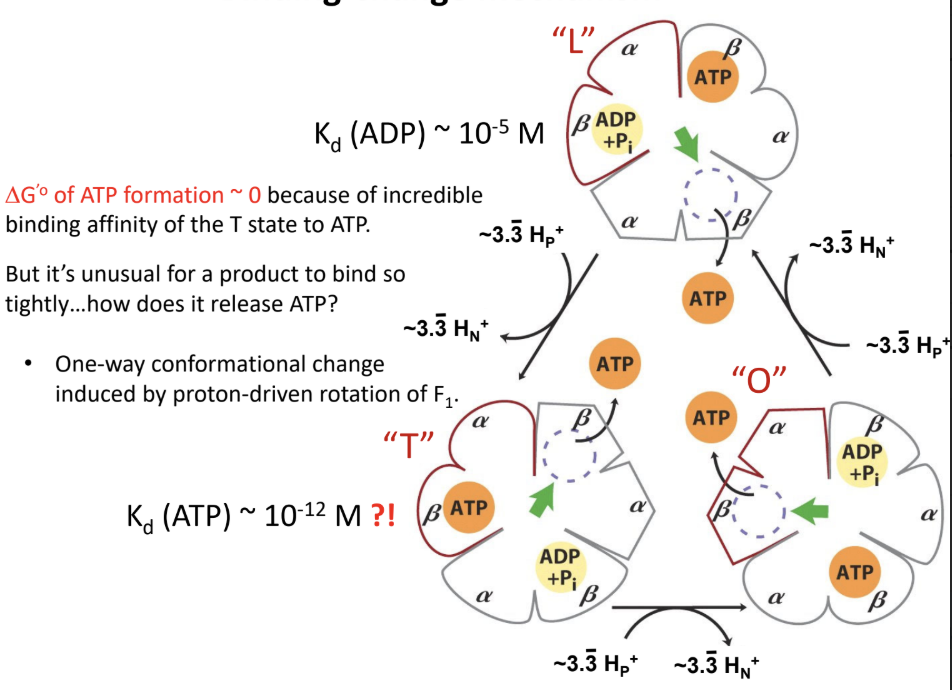
What does the T state do?
Binds ATP tightly and is the catalytic state.
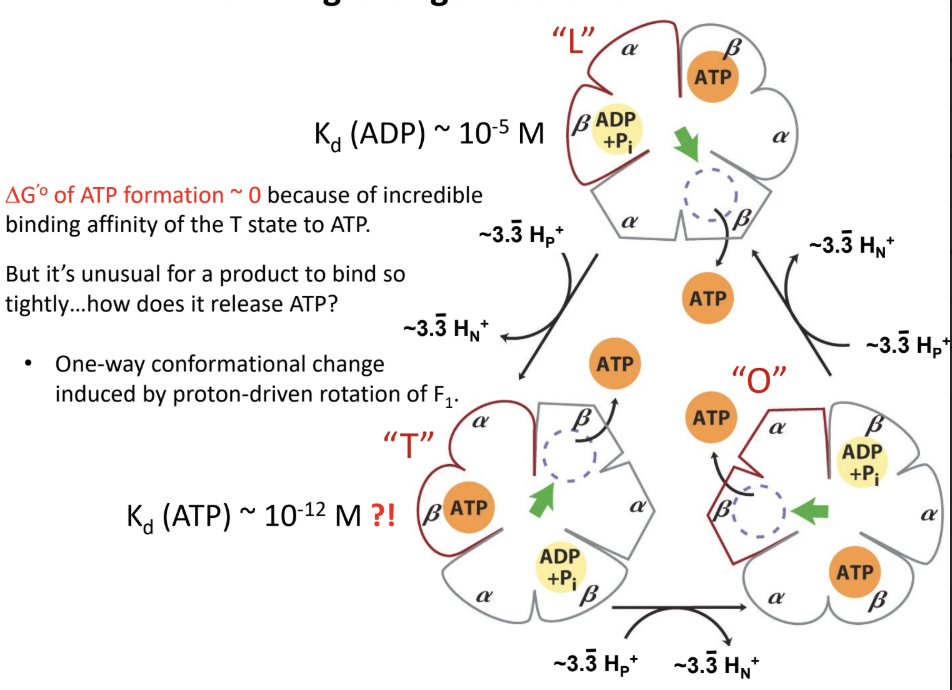
What does the O state do?
Releases ATP.
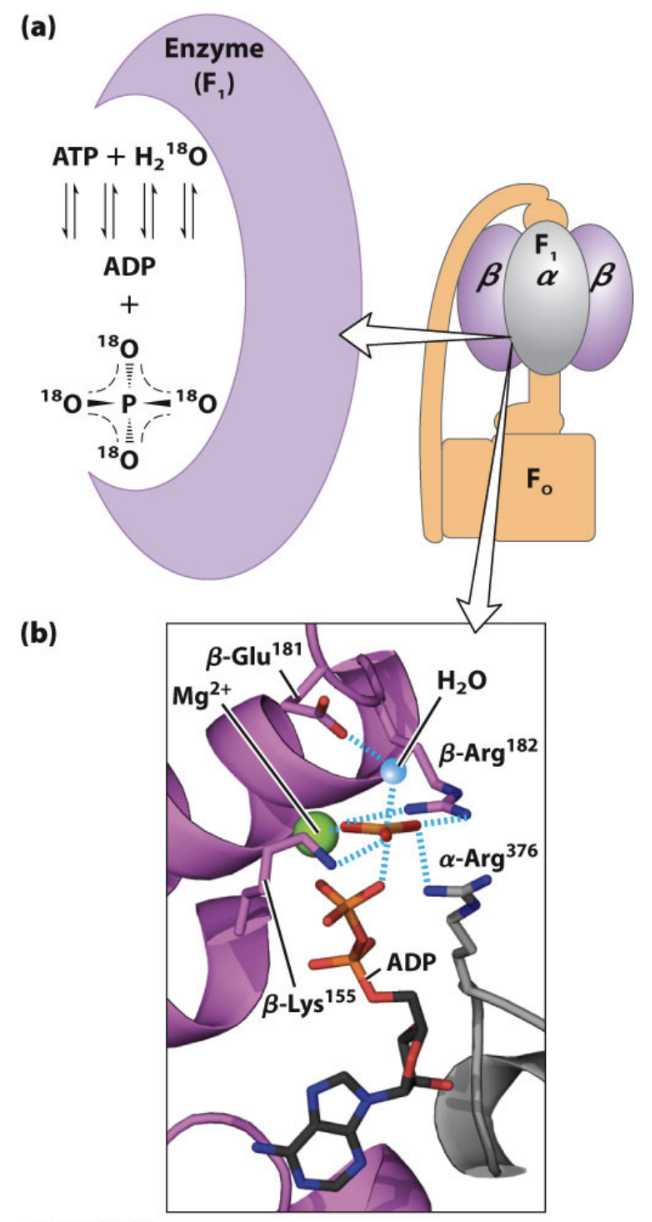
Why is ΔG°′ for ATP formation in ATP synthase approximately 0?
Because ATP formation at the active site is near equilibrium
The main energy input is used for conformational changes and ATP release
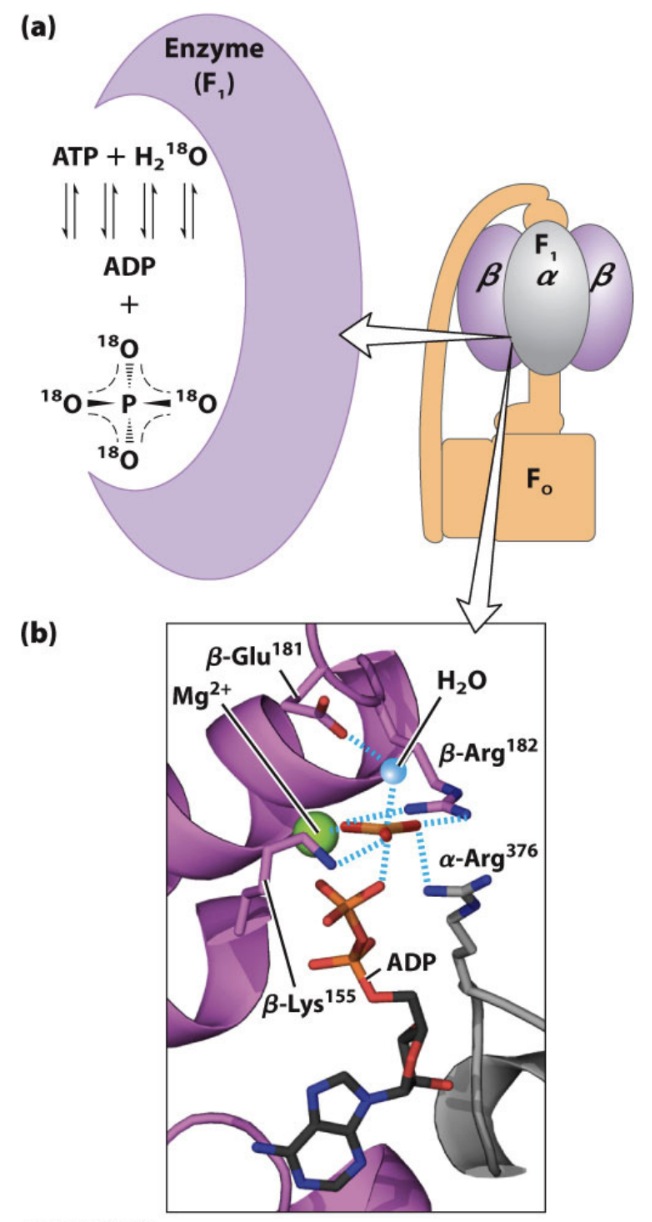
What experimental evidence suggests ATP formation is near equilibrium in ATP synthase?
H218O exchange experiments showed reversibility at the catalytic site.
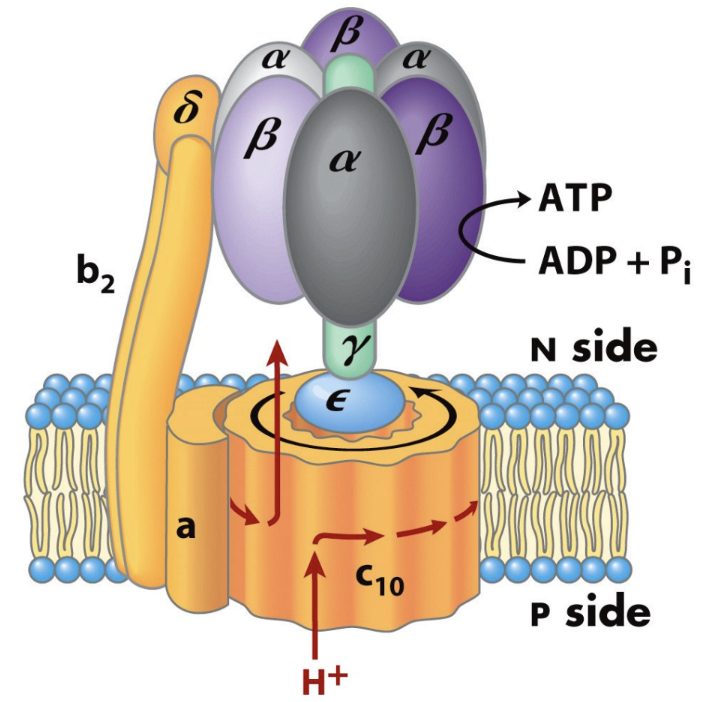
About how many ATP are made per full rotation of human ATP synthase?
About 3 ATP.
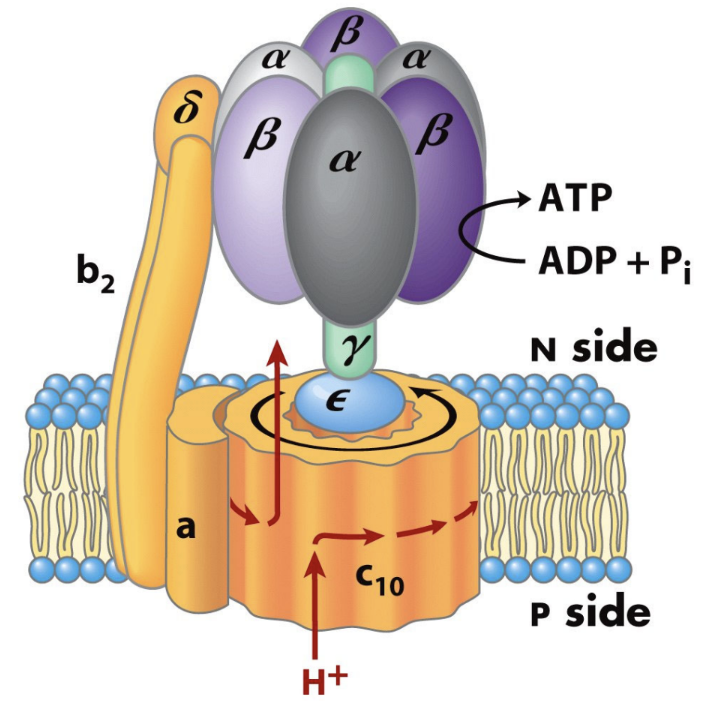
About how many protons are required per full rotation of the human c10 ring?
About 10 H+.
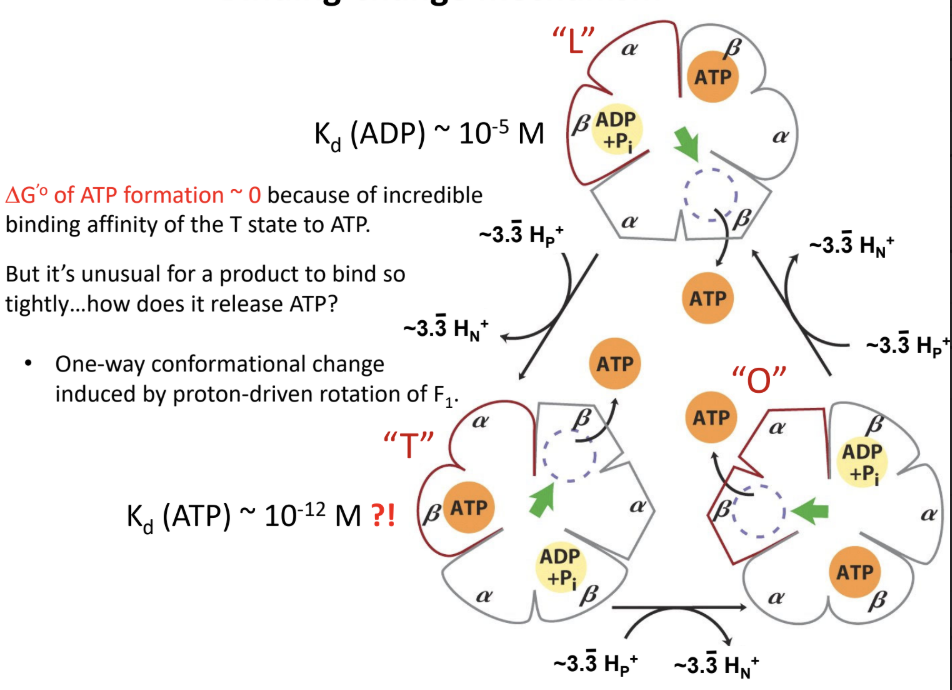
About how many protons are required per ATP in the human enzyme?
About 3.3 H+ per ATP.
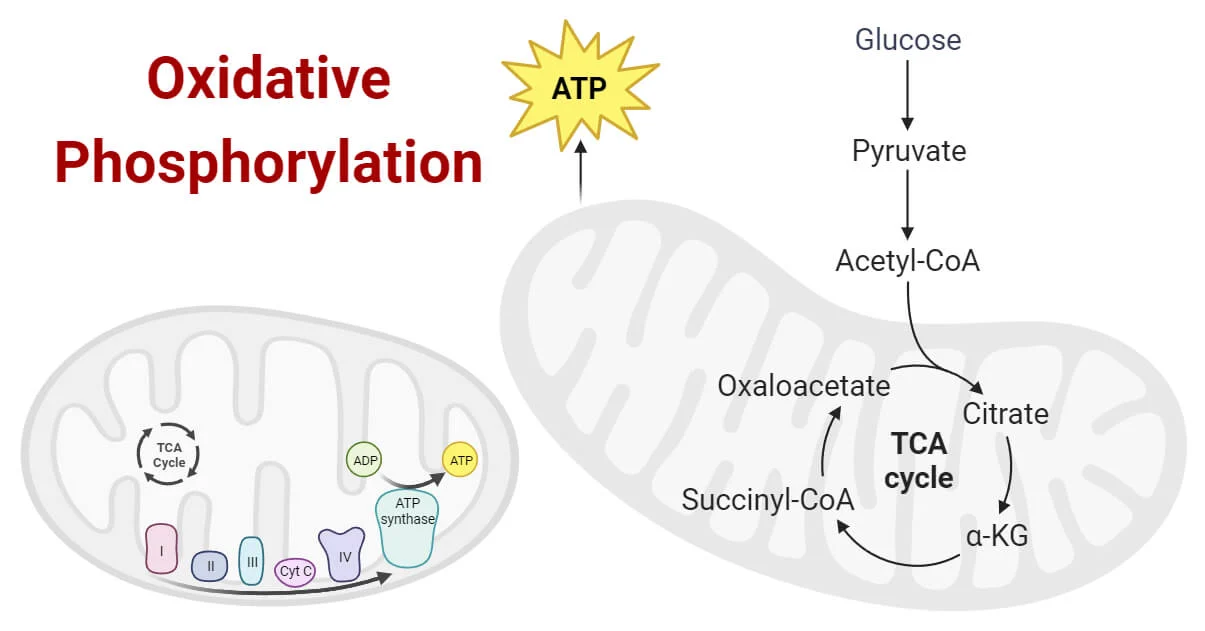
What is oxidative phosphorylation?
ATP synthesis driven by electron transport and the proton-motive force.
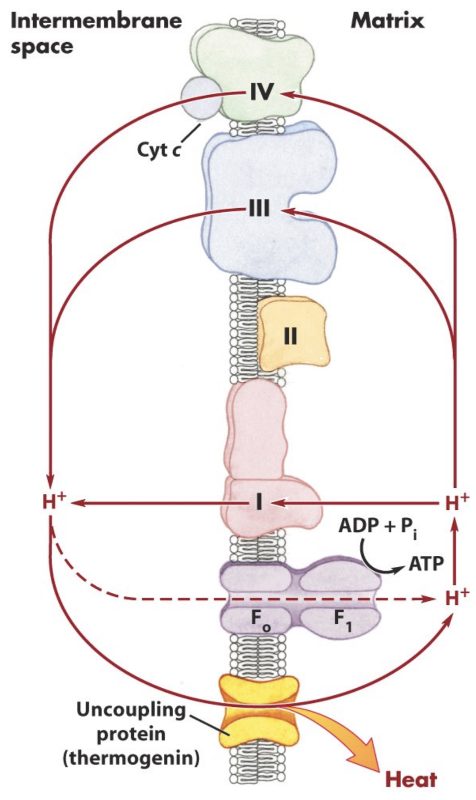
What is uncoupling of oxidative phosphorylation?
Dissipation of the proton gradient without ATP synthesis.
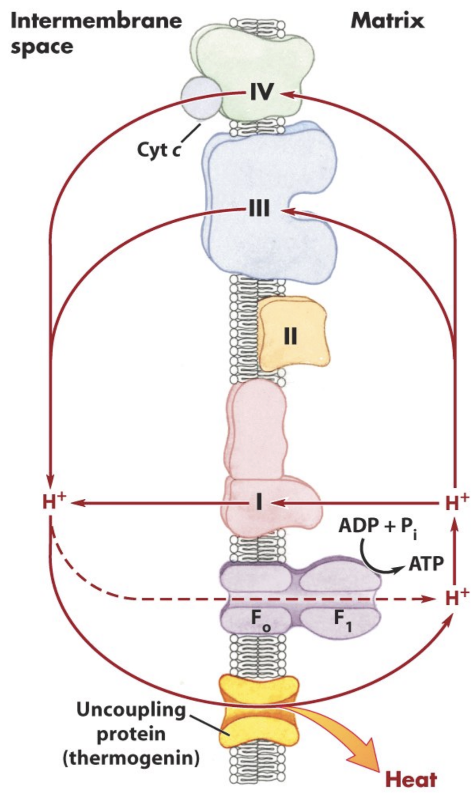
What is the effect of uncoupling on electron transport and ATP production?
Electron transport can continue, but ATP synthesis decreases and energy is released as heat.
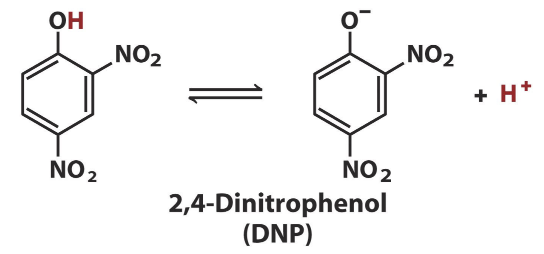
What is DNP?
A chemical uncoupler (ionophore) that carries protons across the membrane.
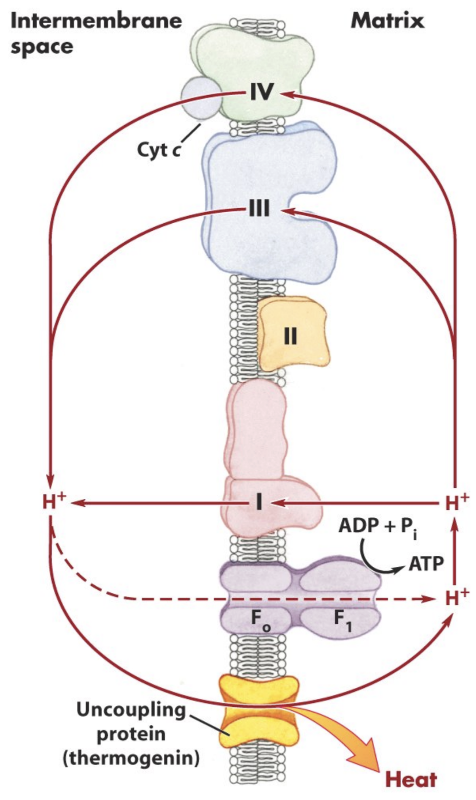
What is UCP/thermogenin?
A physiological uncoupling protein that produces heat, especially in brown fat.