Molecular Geometry Chart - AP Chemistry
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
26 Terms
Linear (0 lone pairs)
2 electron groups, 0 lone pairs
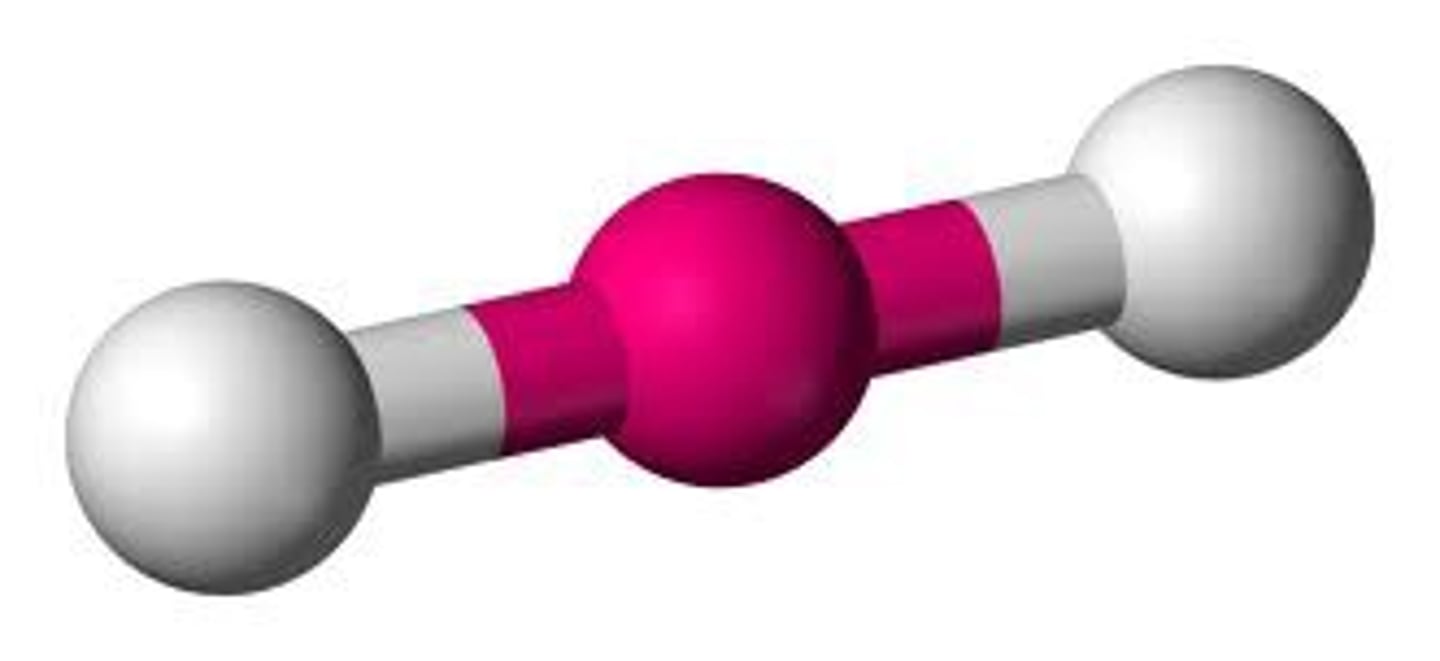
linear bond angle
180 degrees
trigonal planar
3 electron groups, 0 lone pairs
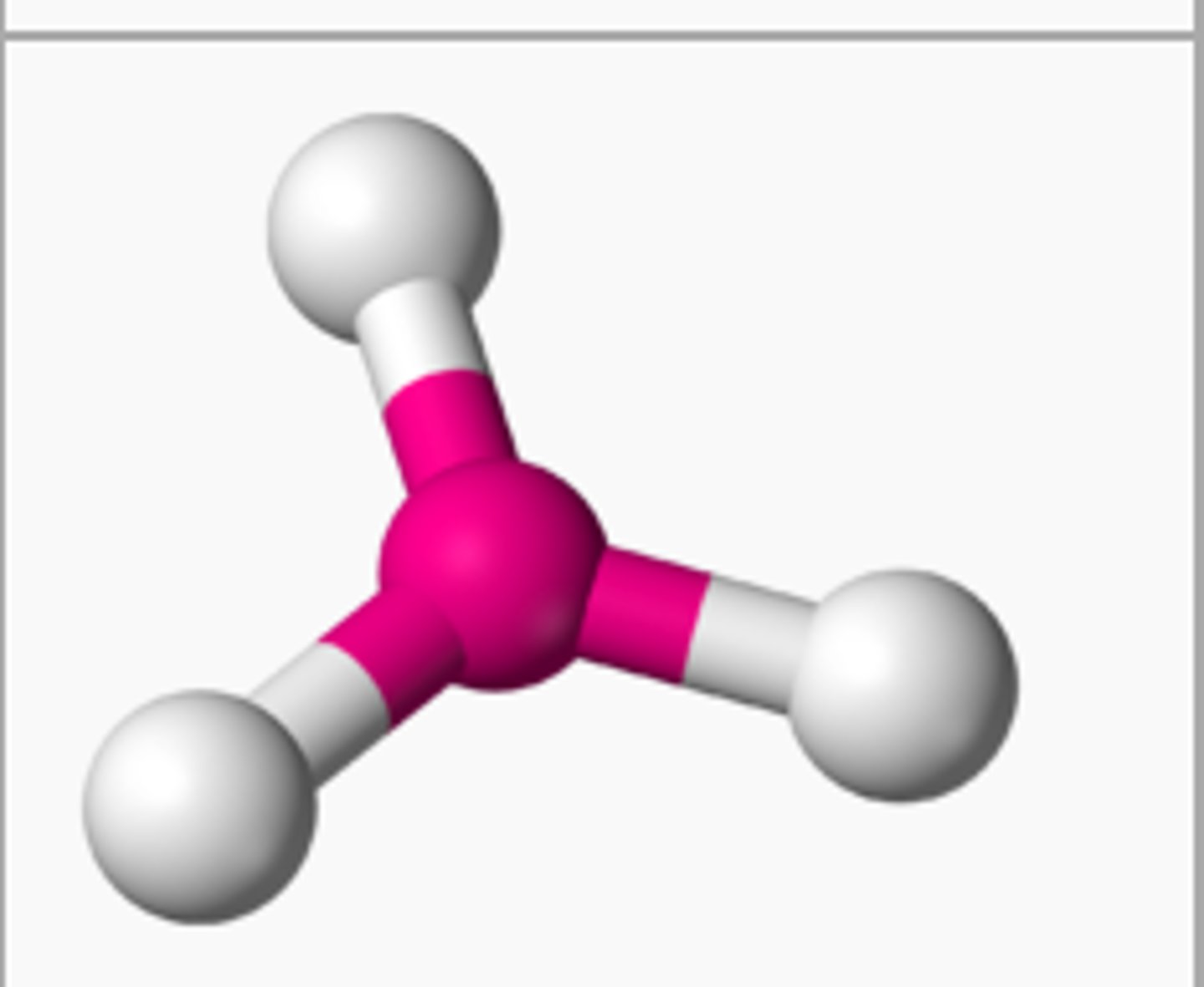
trigonal planar bond angle
120 degrees
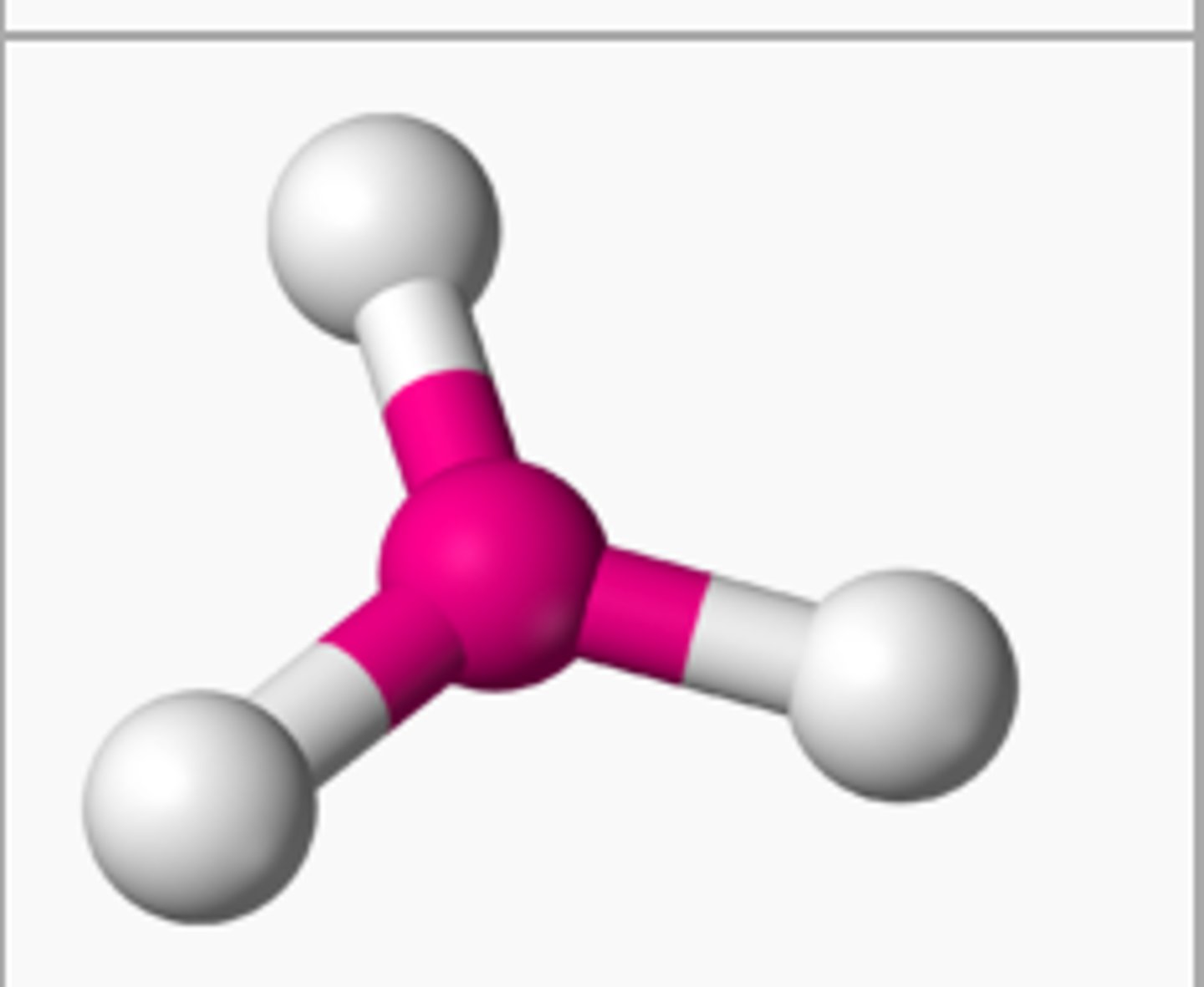
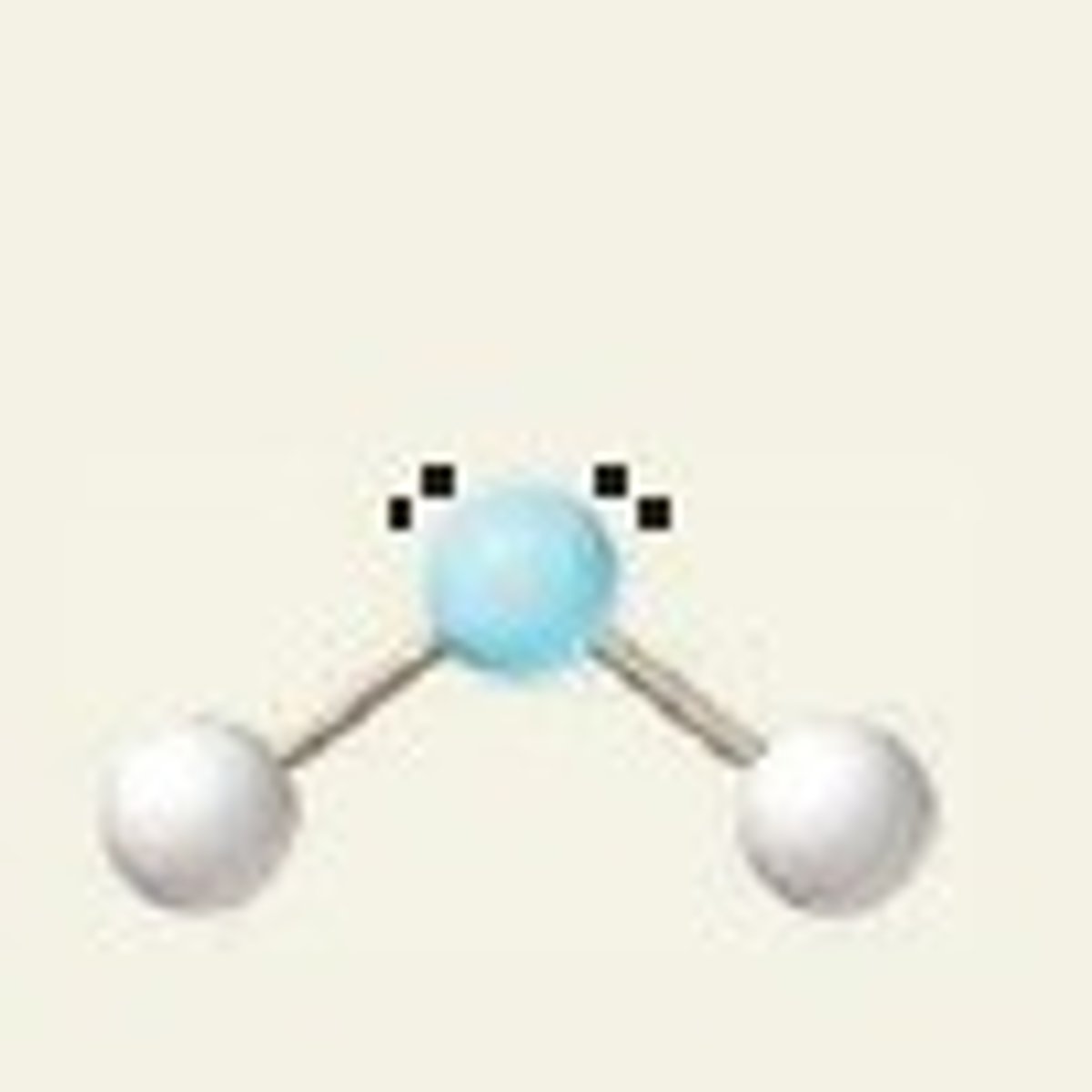
bent (1 lone pair)
3 electron groups, 1 lone pair

bent bond angle (1 lone pair)
less than 120 degrees

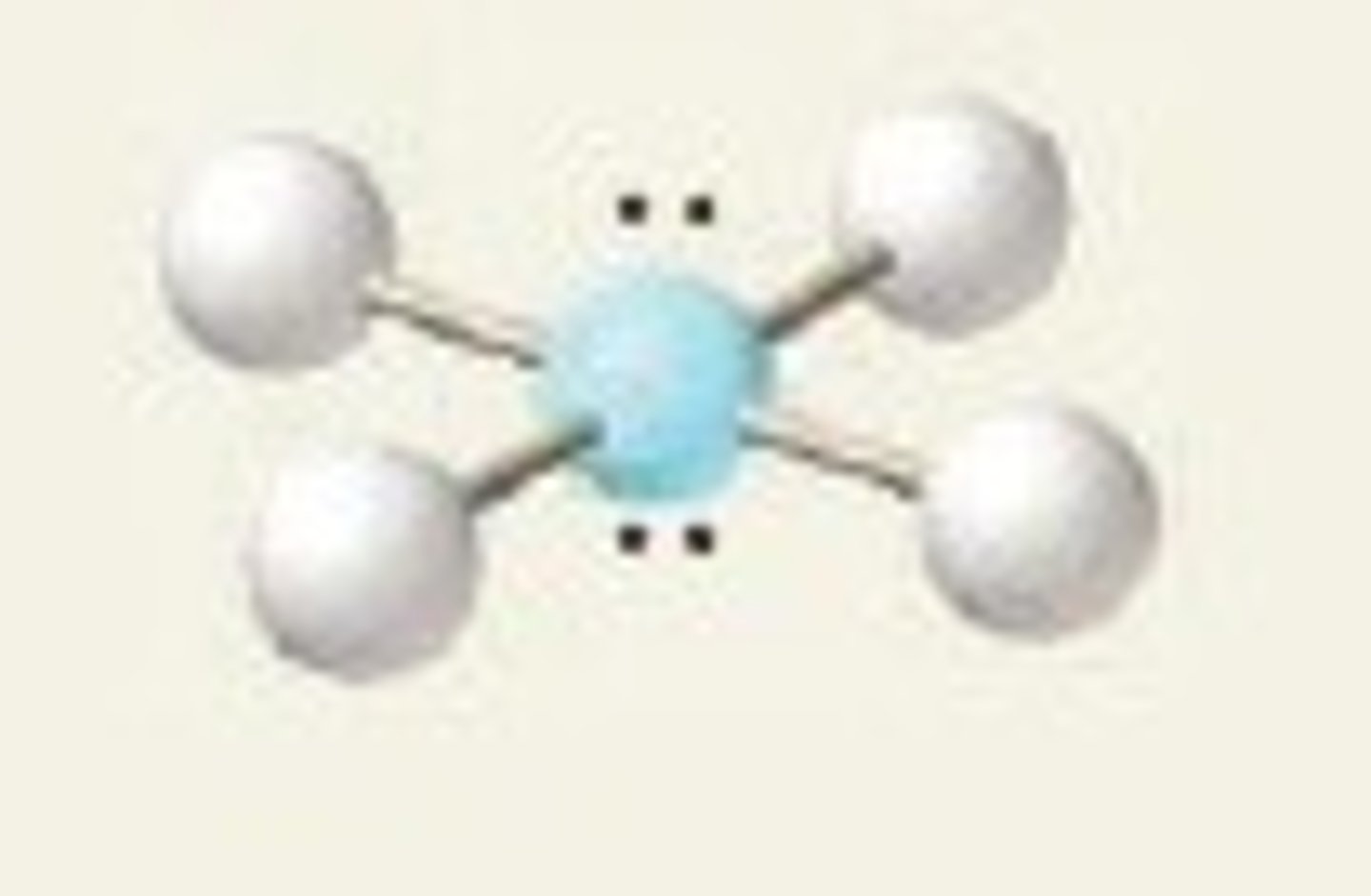
tetrahedral
4 electron groups, 0 lone pairs
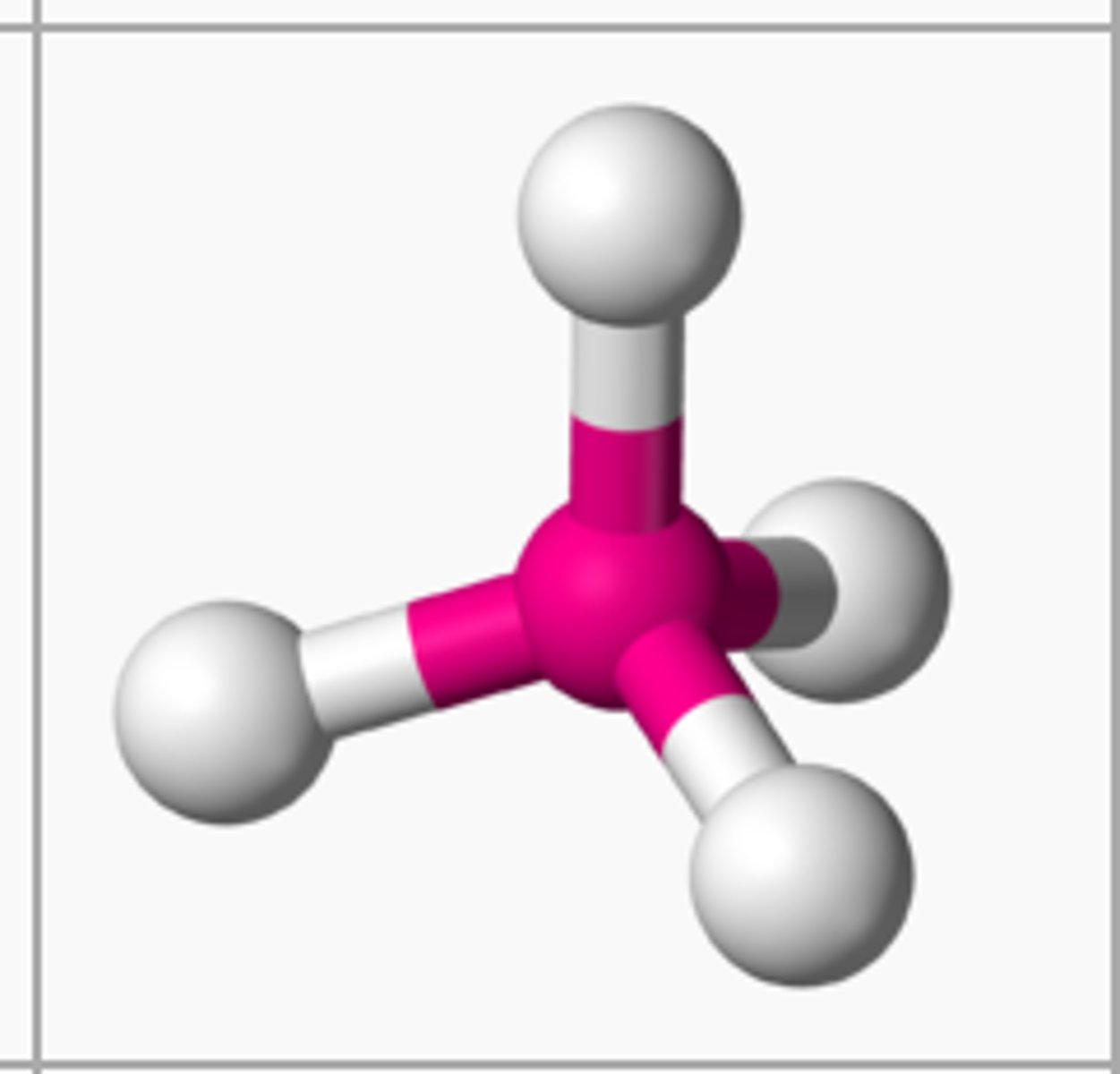
tetrahedral bond angle
109.5 degrees
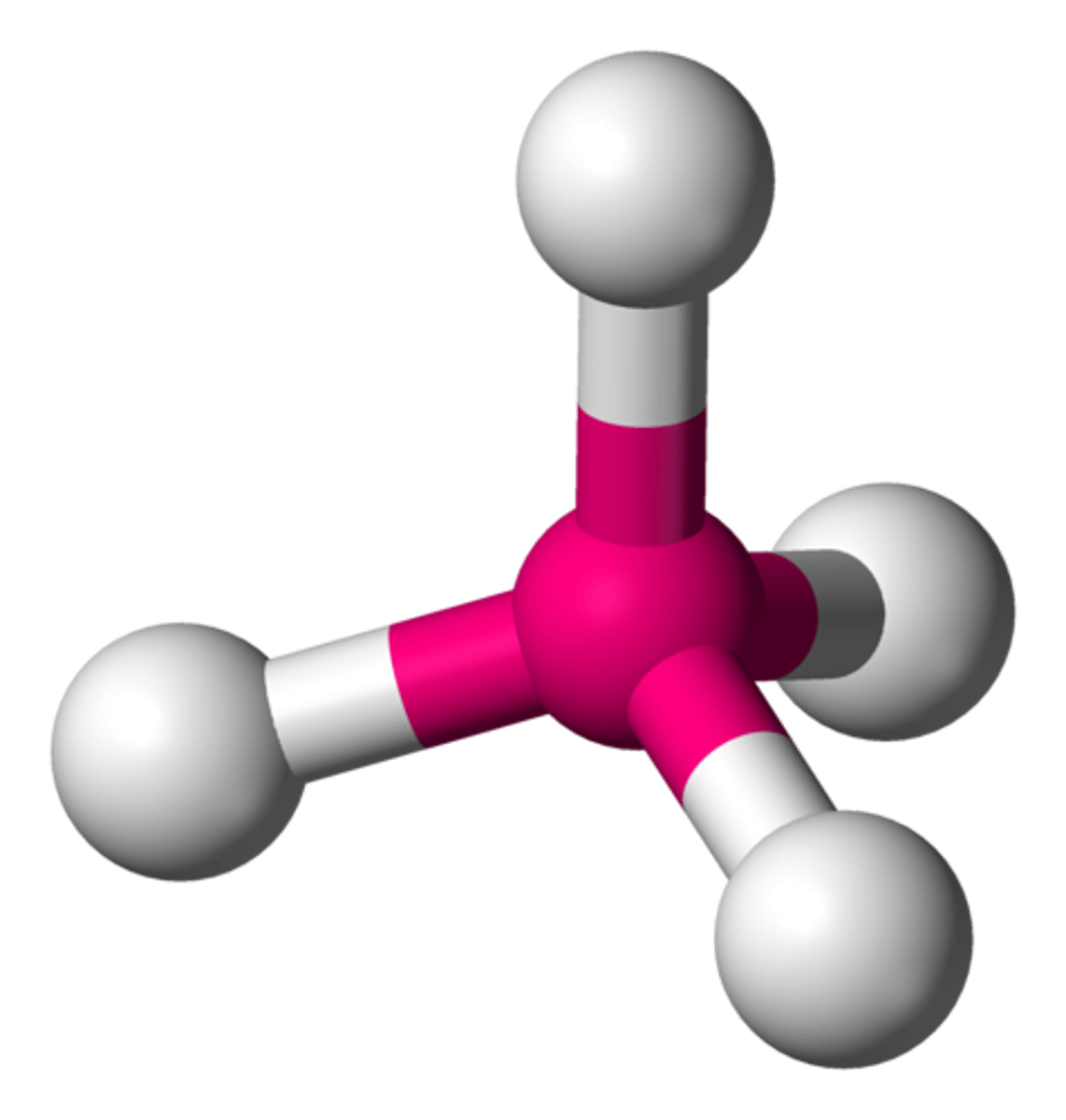
trigonal pyramid
4 electron groups, 1 lone pair

trigonal pyramid bond angle
<109.5 (~107) degrees

bent (2 lone pairs)
4 electron groups, 2 lone pairs
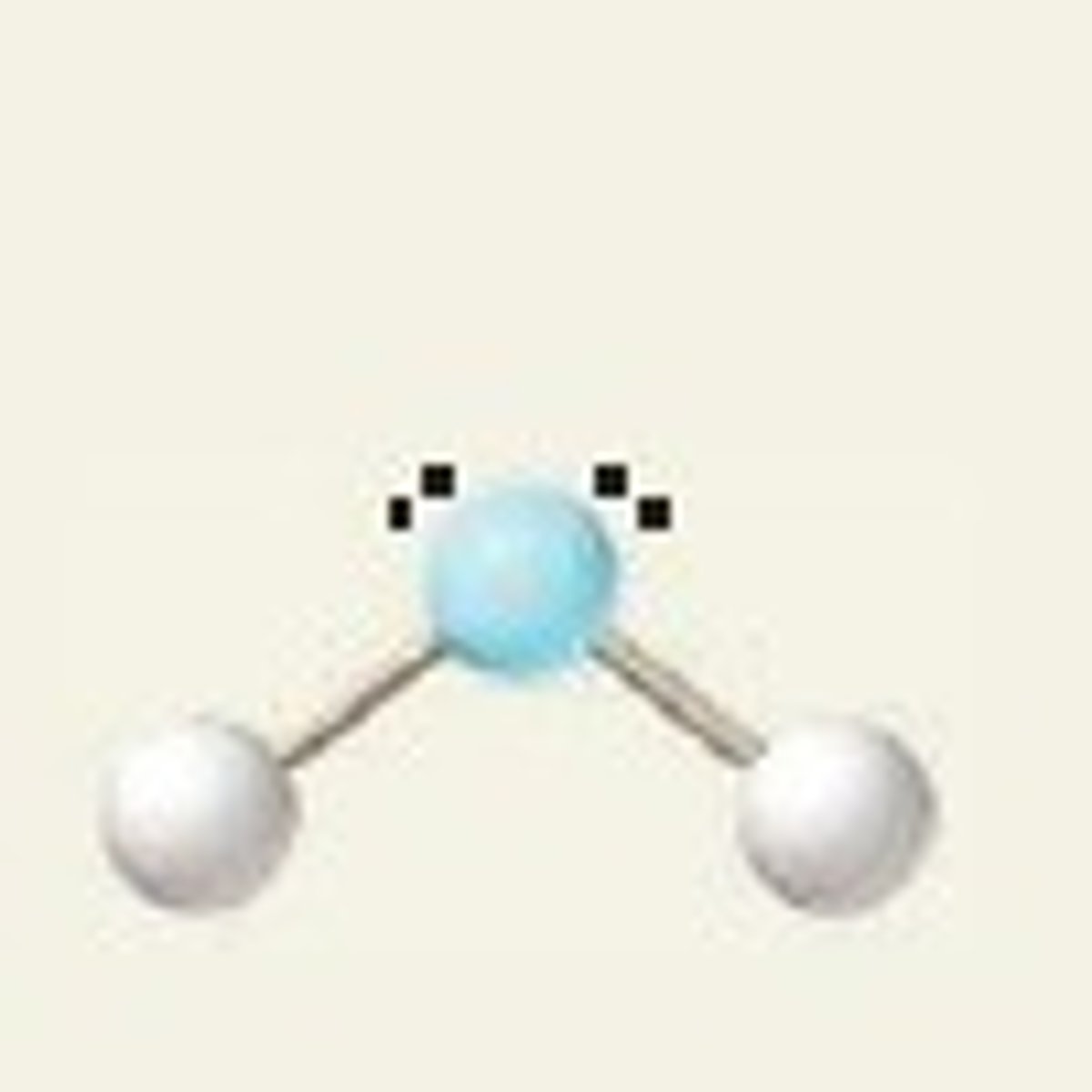
bent bond angle (2 lone pairs)
<109.5 (~105) degrees
trigonal bypyramidal
5 electron groups, 0 lone pairs
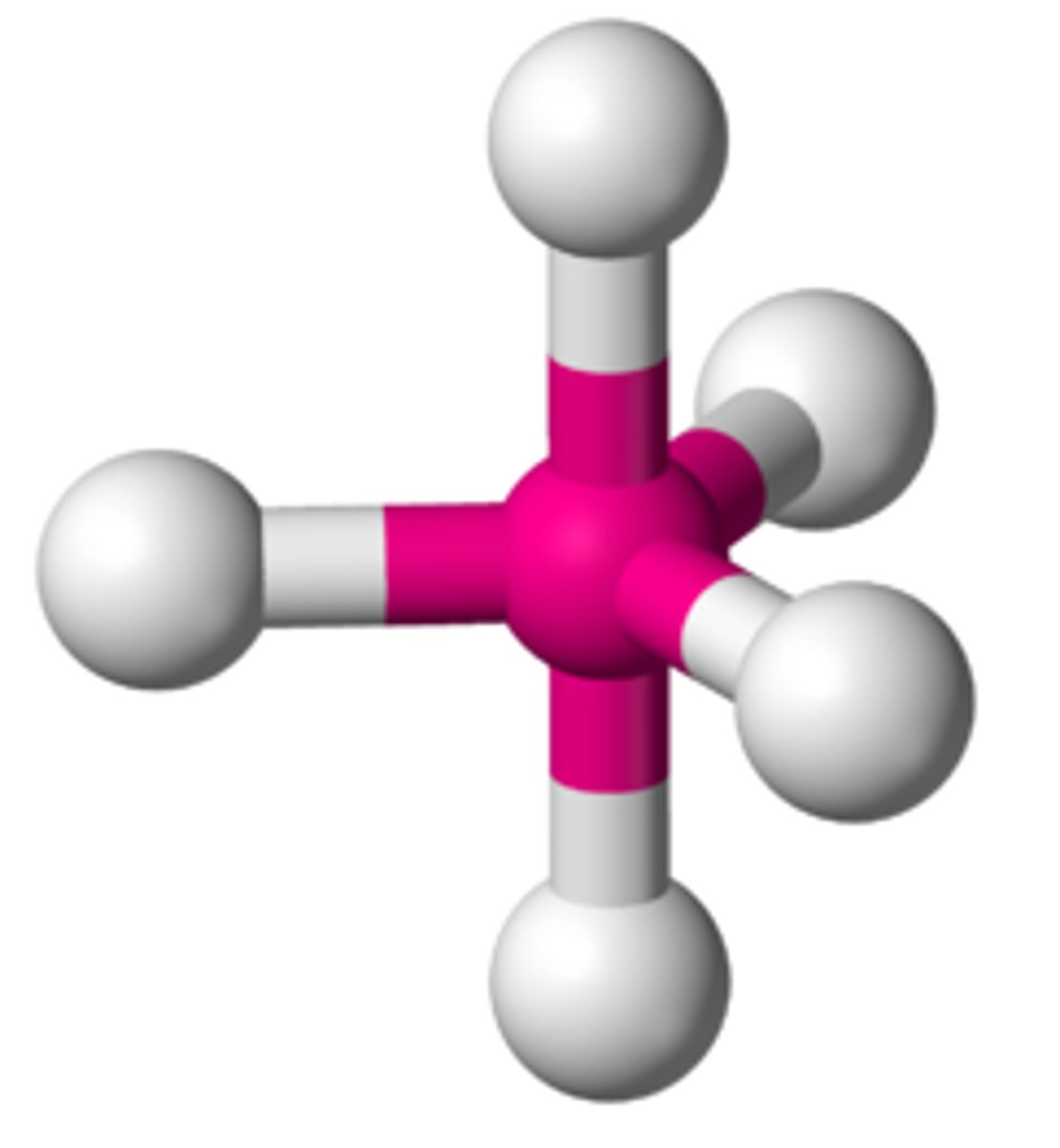
trigonal bypyramidal bond angles
90 degrees, 120 degrees
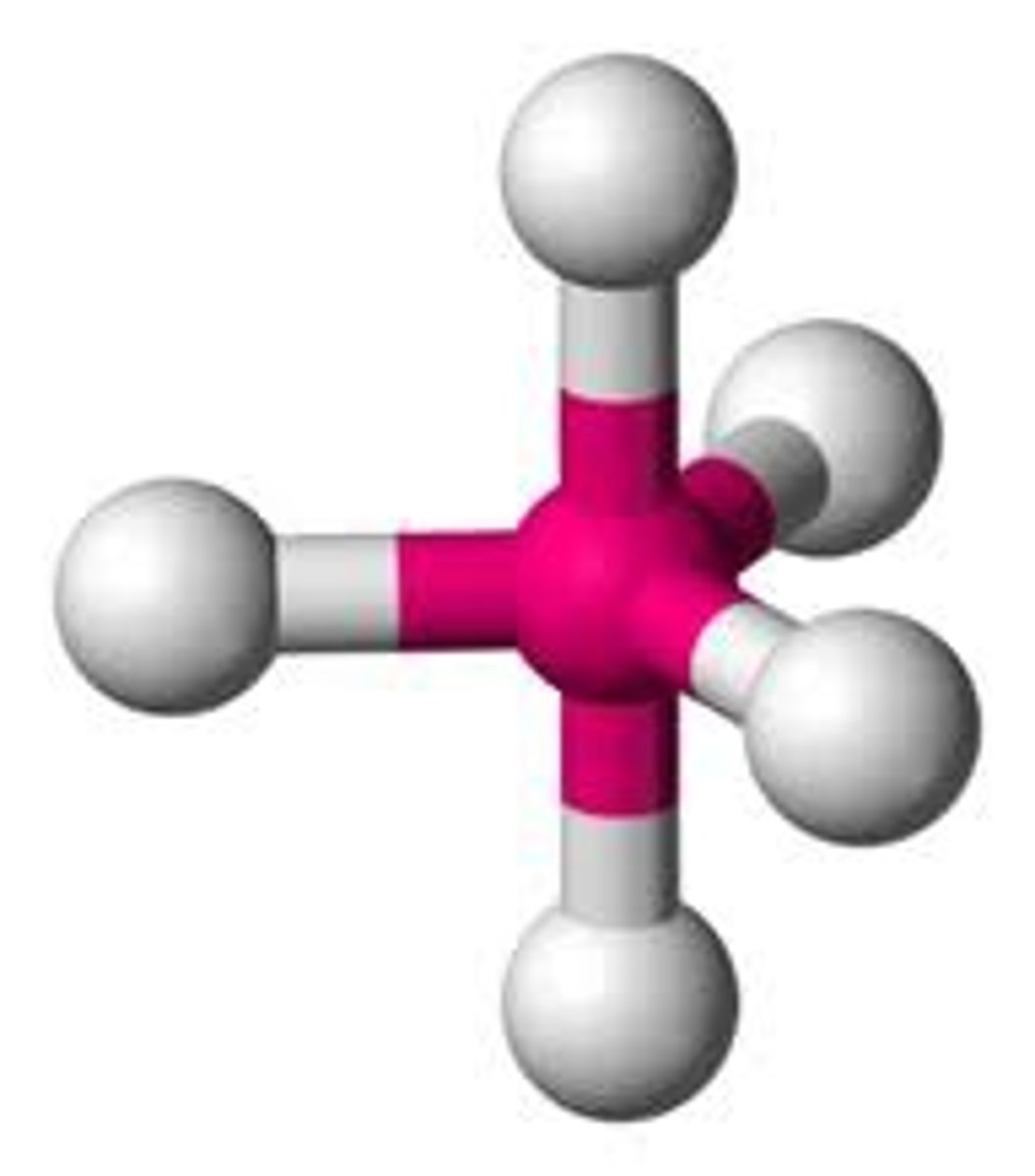
see-saw
5 electron groups, 1 lone pairs
see-saw bond angles
<90 degrees, <120 degrees
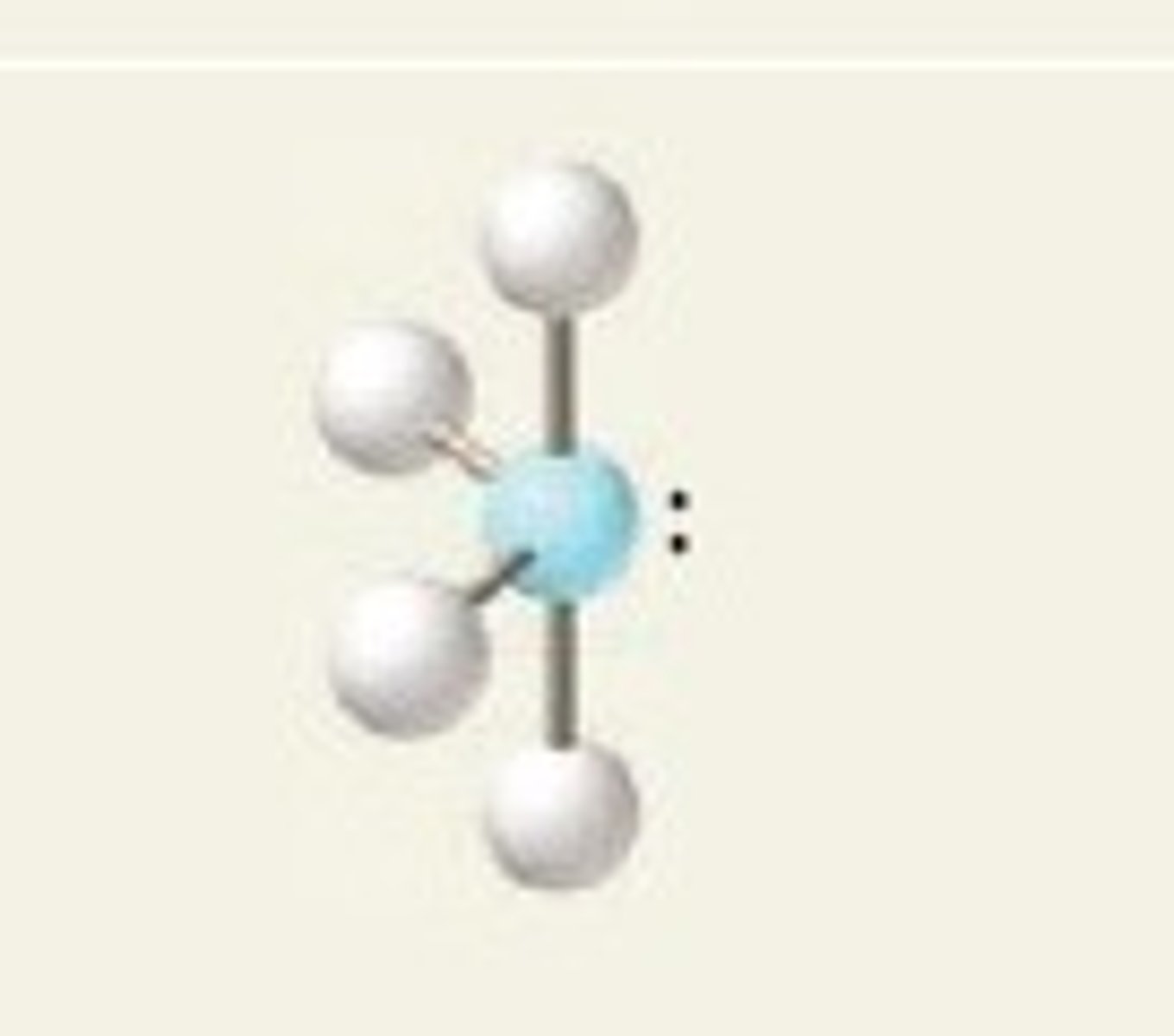
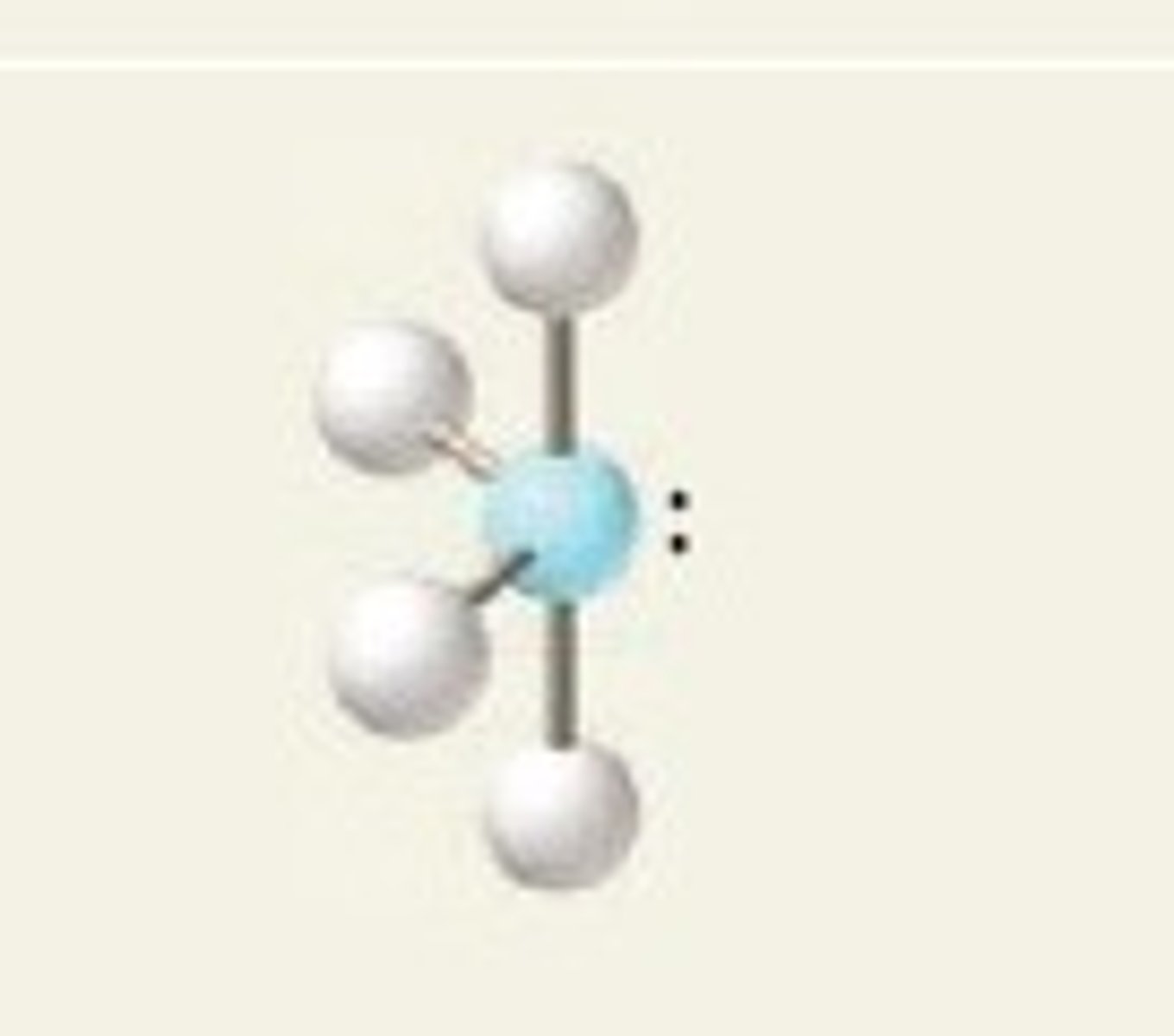
t-structure
5 electrons, 2 lone pairs
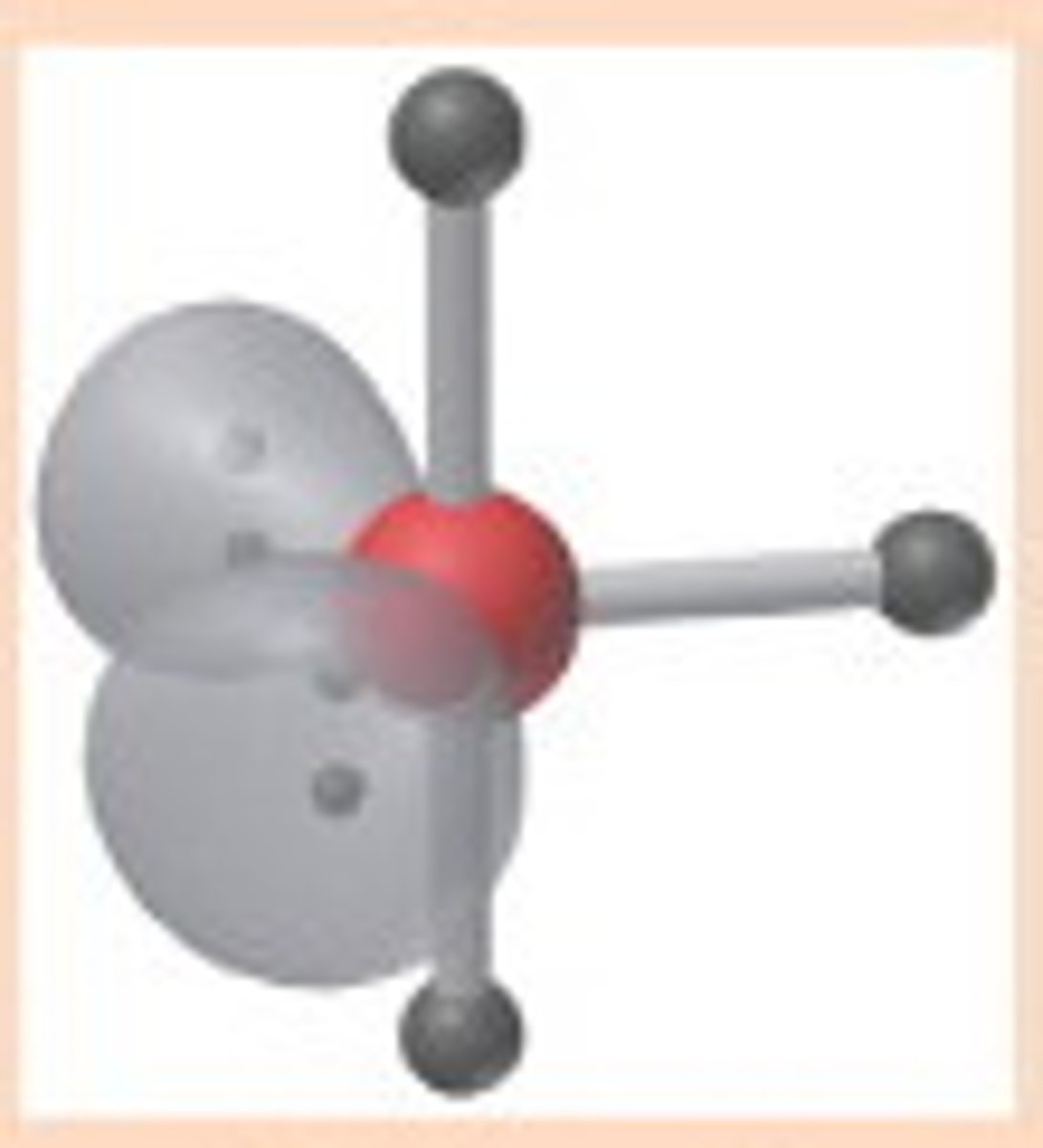
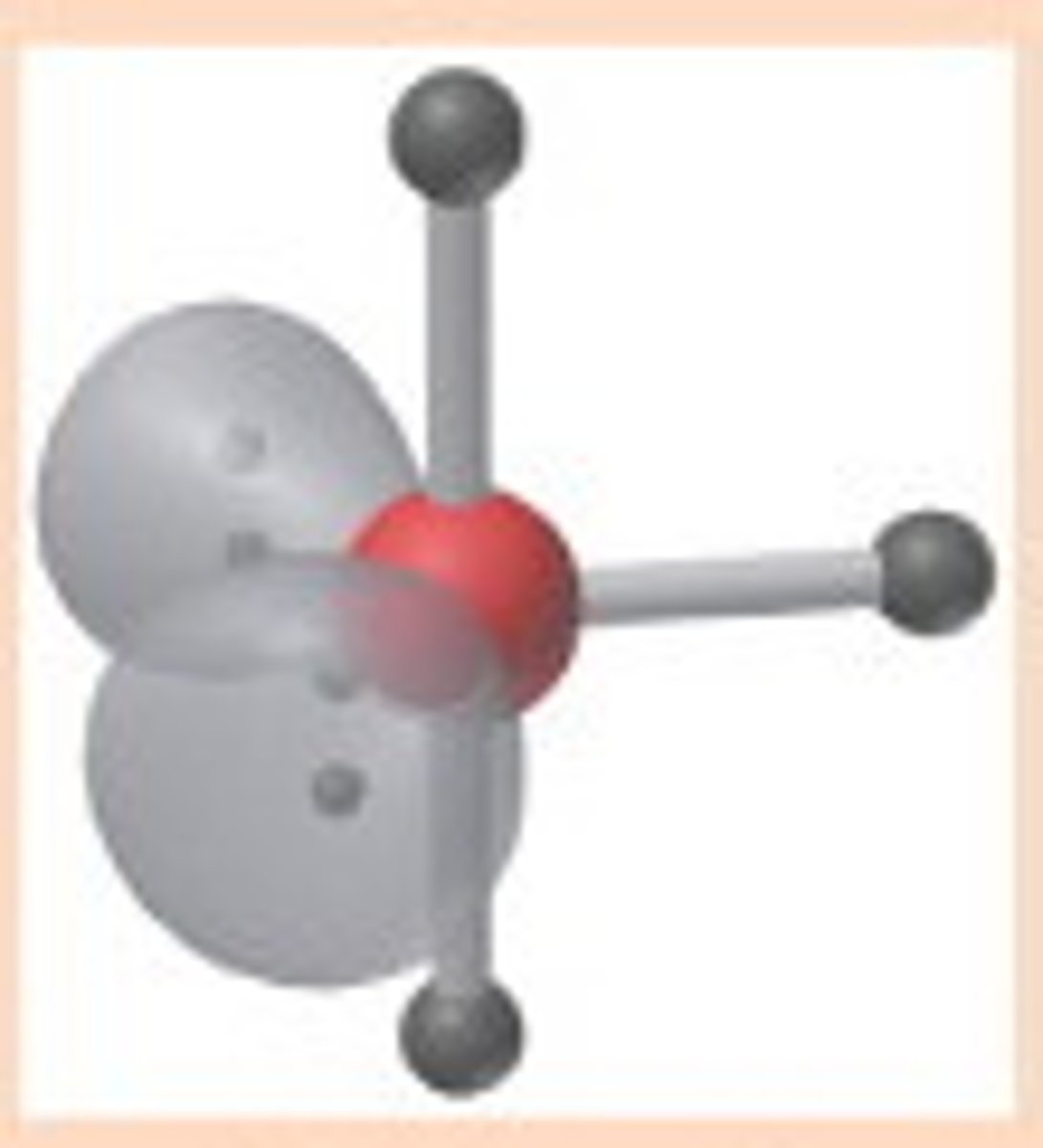
t-structure bond angles
<90 degrees
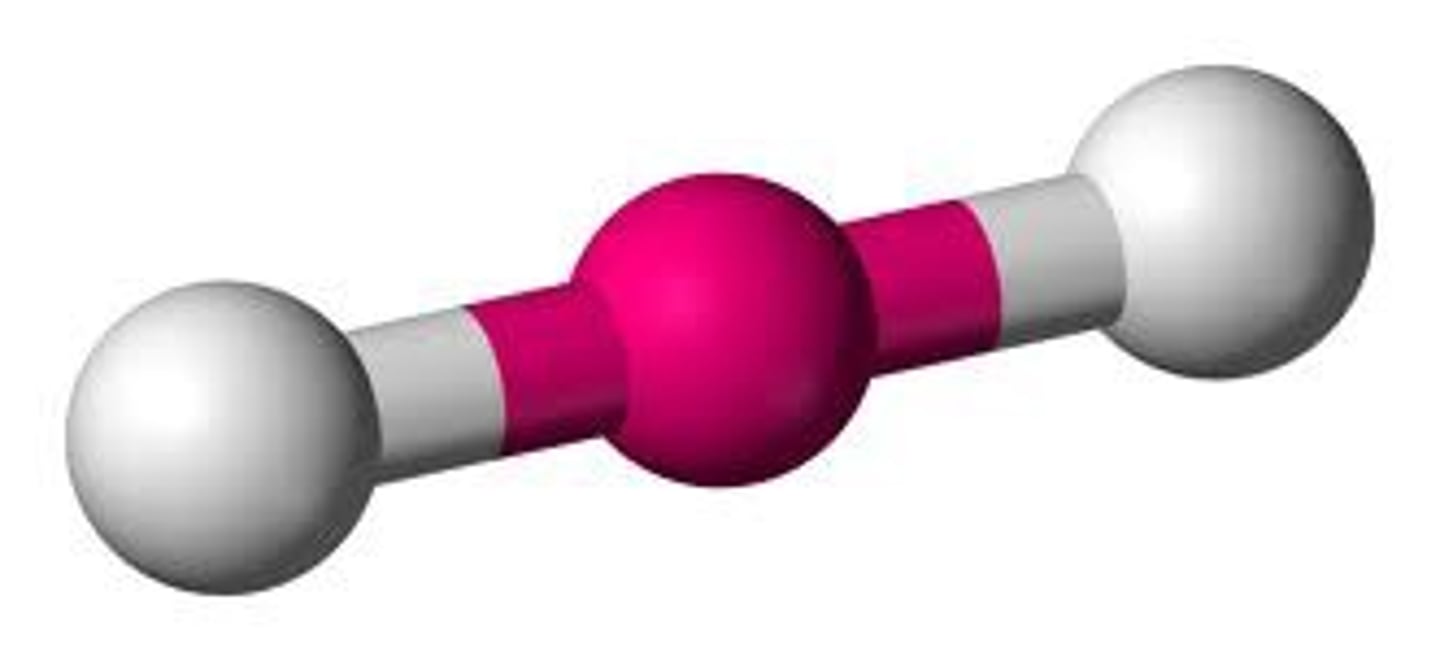
linear (3 lone pairs)
5 electron groups, 3 lone pairs
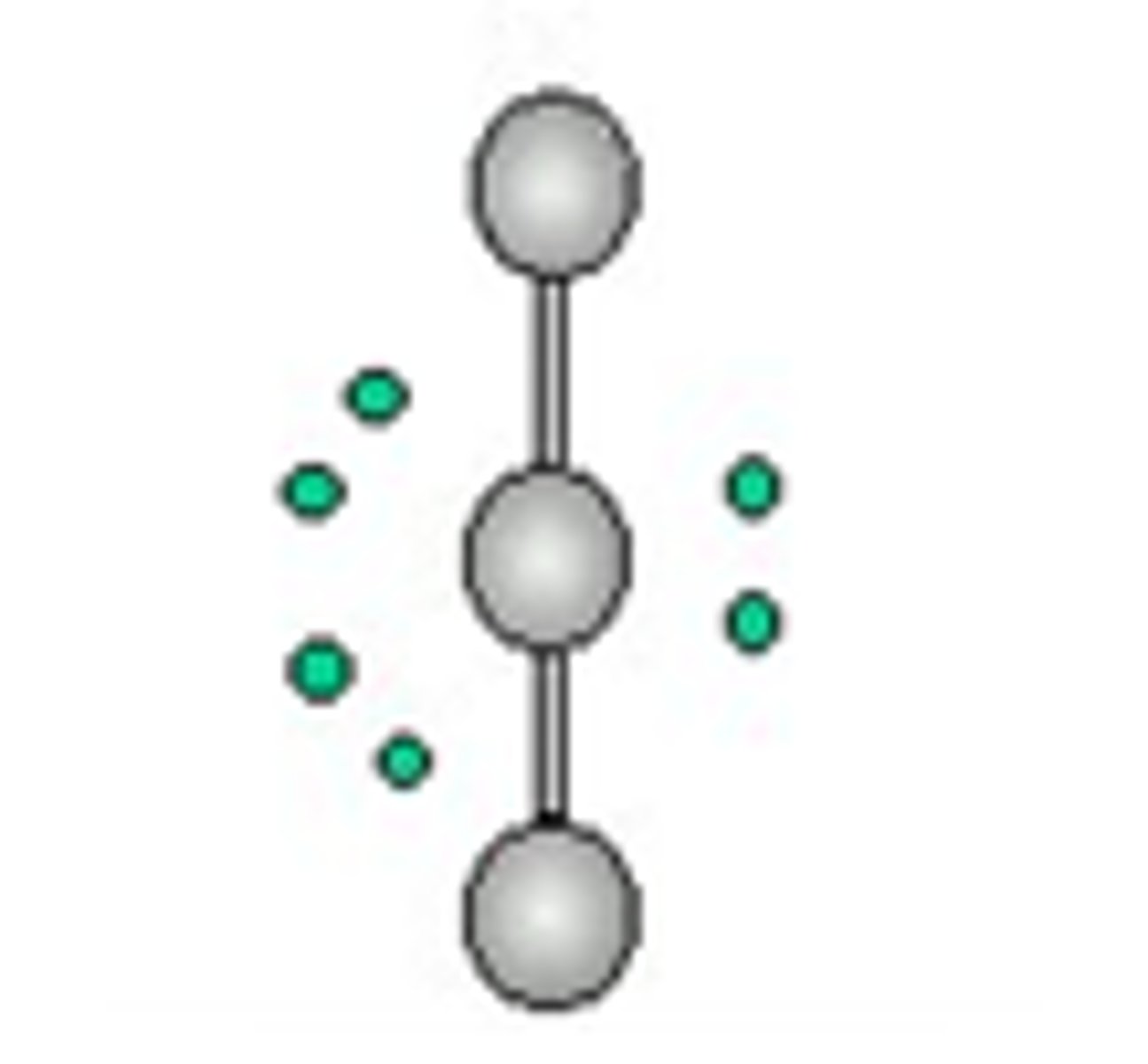
linear (3 lone pairs) bond angle
180 degrees
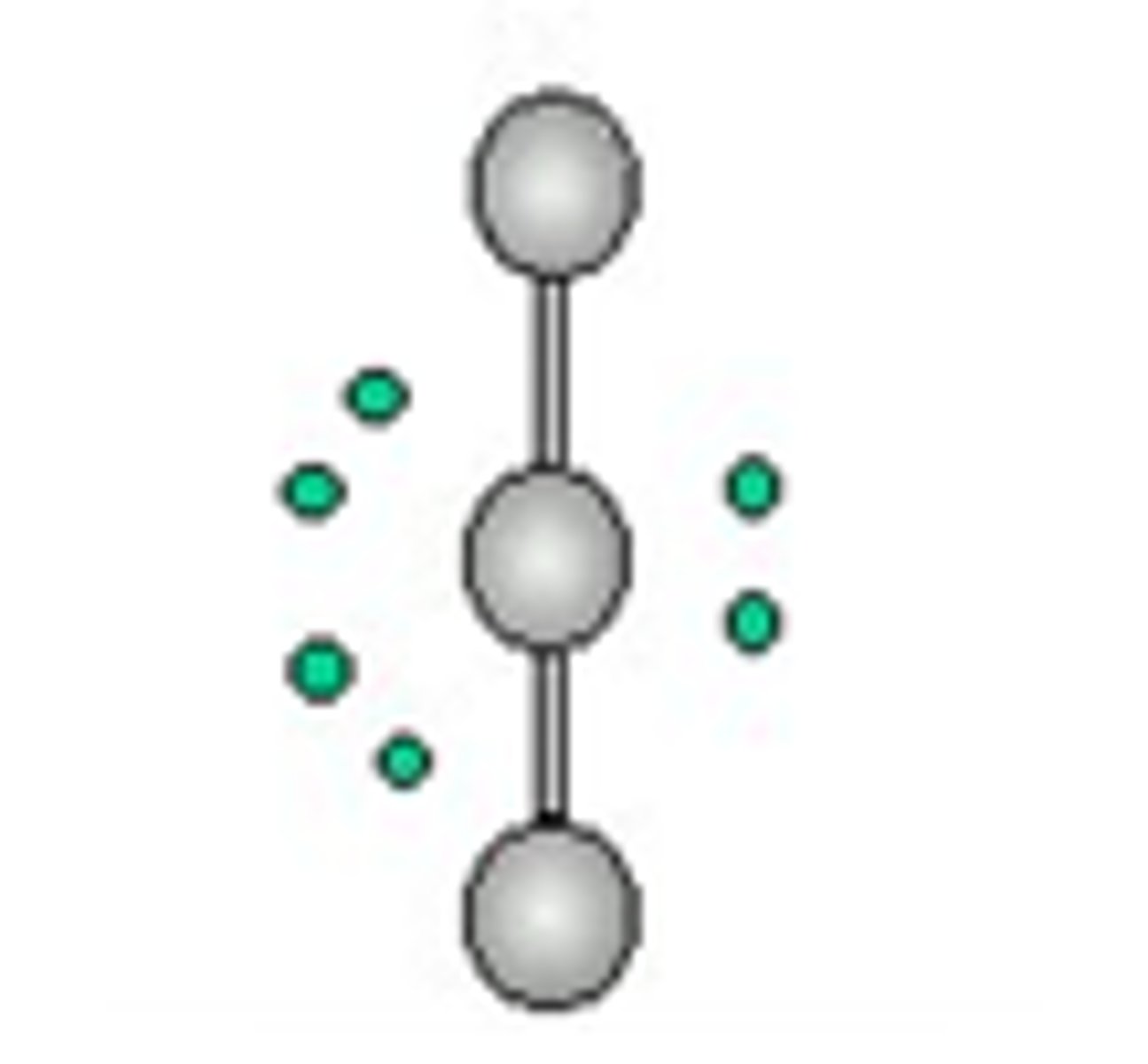
octahedral
6 electron groups, 0 lone pairs
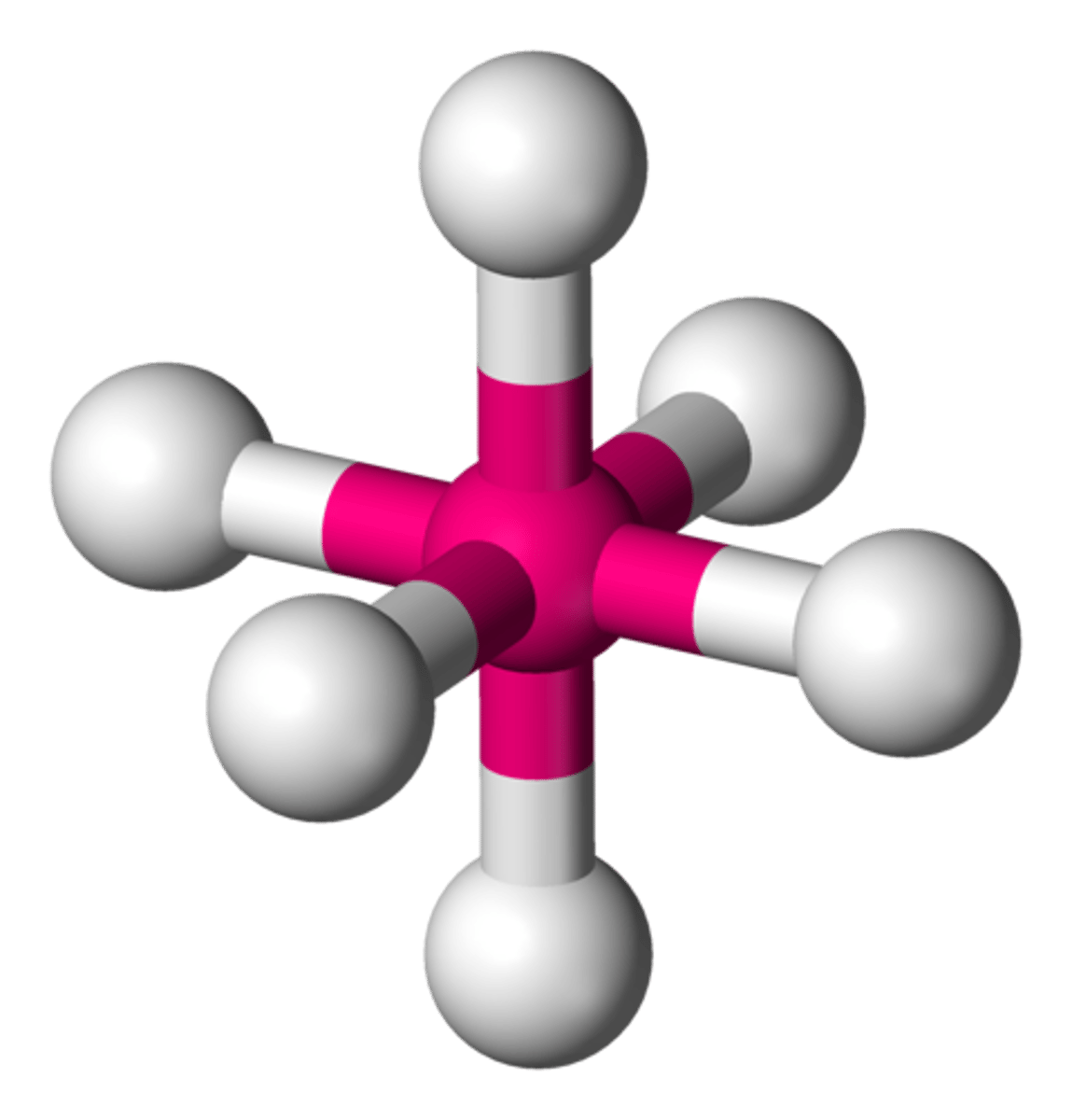
octahedral bond angle
90 degrees, 90 degrees
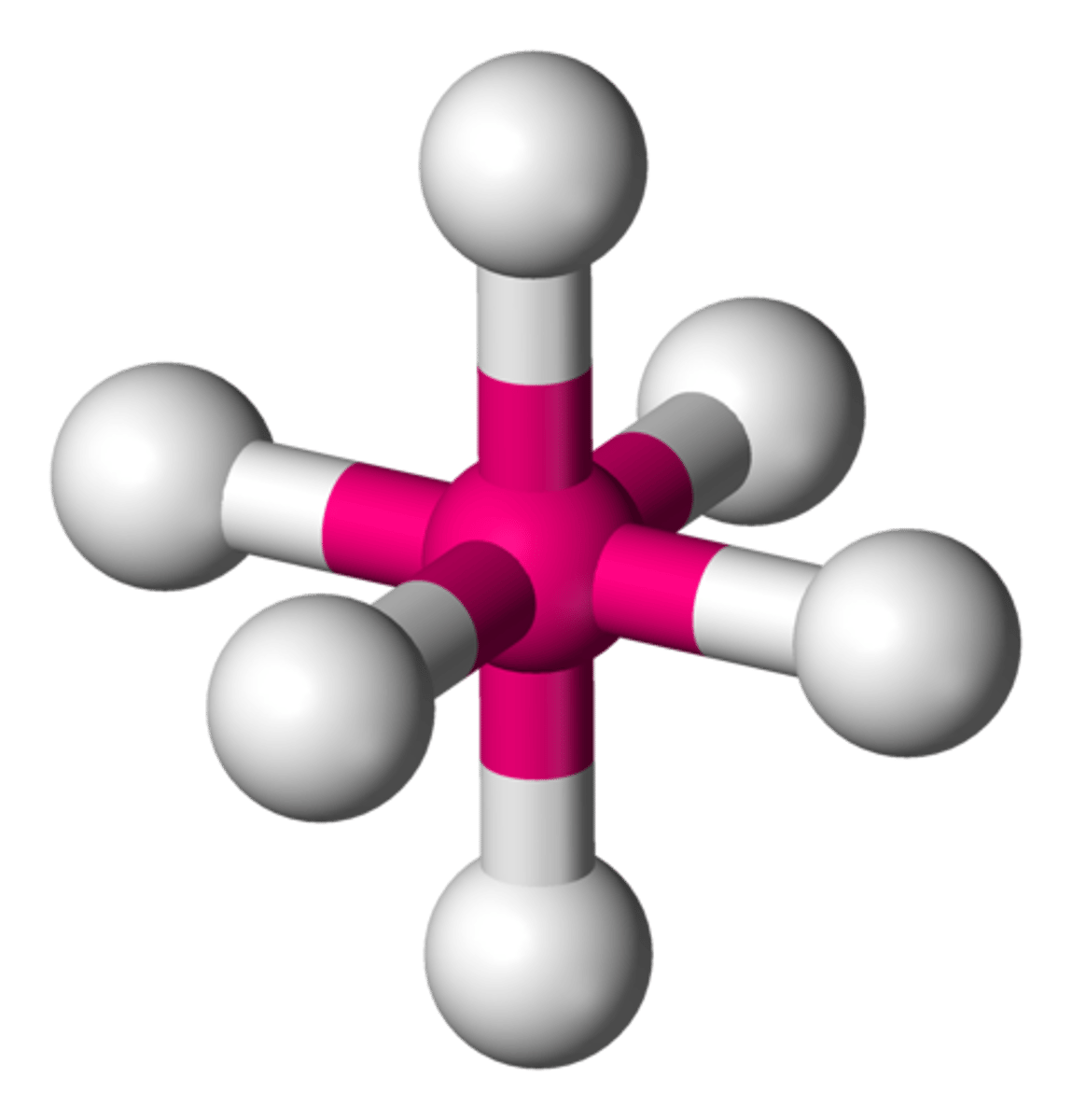
square pyramidal
6 electrons, 1 lone pair
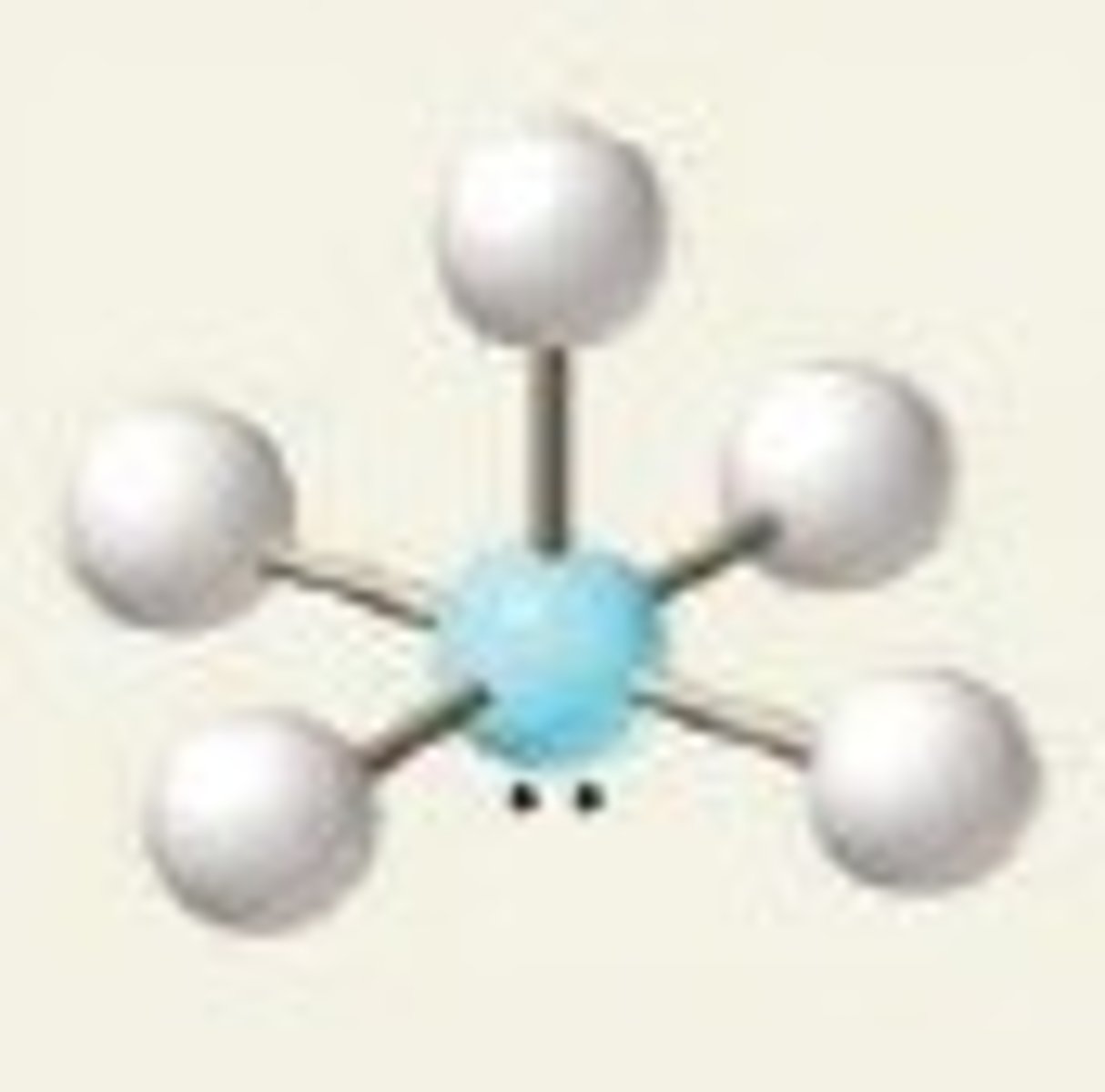
square pyramidal bond angles
90 degrees, less than 90 degrees
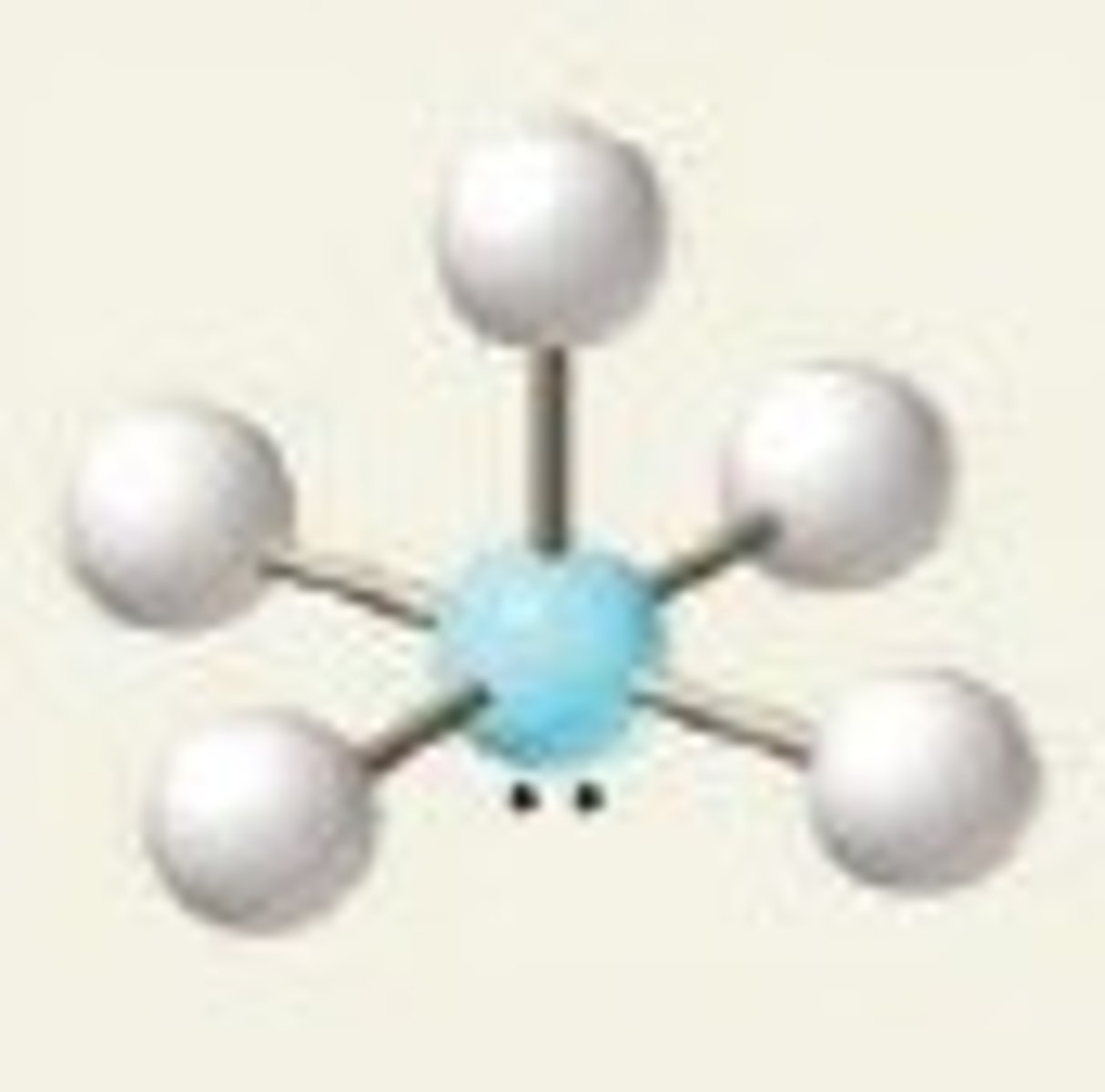
square planar
6 electrons, 2 lone pairs
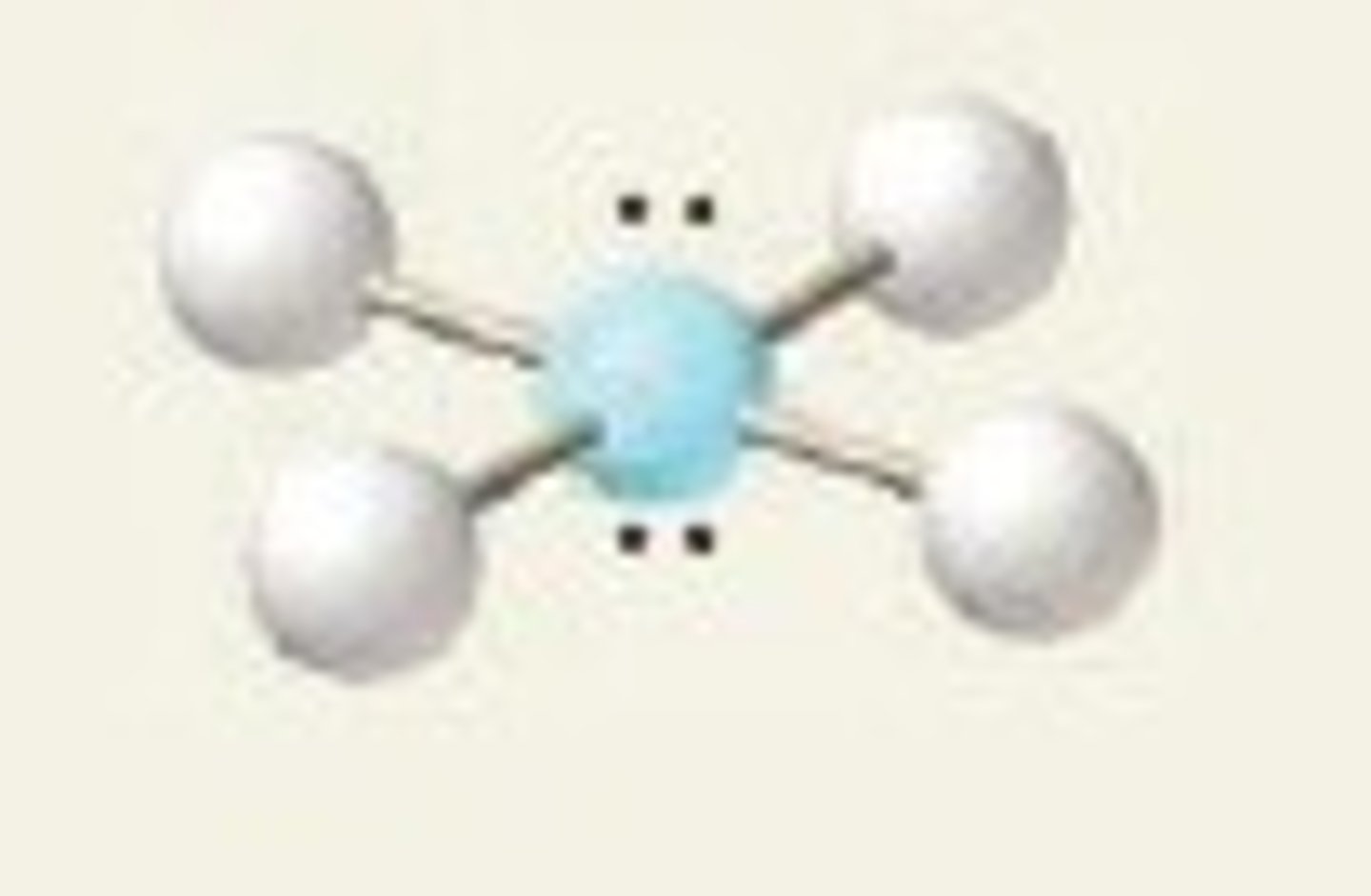
square planar bond angles
90 degrees