L1- Adrenal pharmacology HPA and RAAS
1/168
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
169 Terms
The adrenal glands consist of two functionally distinct regions. What are they?
The adrenal cortex (main target for pharmacological therapies)
The adrenal medulla
What are the three functionally distinct layers (outer to inner) of the adrenal cortex and their products?
Zona glomerulosa → Mineralocorticoids (aldosterone)
Zona fasciculata → Glucocorticoids (cortisol)
Zona reticularis → Sex steroids (androgens)
GFR
GM
FG
GS
What does the adrenal medulla produce? Give two examples
Catecholamines (e.g., adrenaline and noradrenaline)
What regulates adrenal hormone secretion?
ACTH (pituitary) → cortisol
RAAS → aldosterone
What are the 3 main functions of cortisol?
Stress response (↑ Cardiovascular responsiveness)
Anti-inflammatory and immunosuppressive effects
Metabolism regulation
C- SAM
What are the 4 metabolic effects of cortisol?
Increase gluconeogenesis in liver
Increases glyogenolysis
Increases lipolysis in muscle
Increases proteolysis in muscle
What are the 3 cardiovascular effects of cortisol?
Increases myocardial contractility
Increases CO
Increases catecholamine pressor effect
What are the 3 main functions of aldosterone?
Potassium excretion
Blood pressure regulation
Sodium retention
A-PBS
What stimulates the HPA axis? Give 5 examples
Stress
Hypoglycaemia
Hypotension
Fever
Trauma
Surgery
What is the pathway of the HPA axis?
Hypothalamus (CRH) → Pituitary (ACTH) → Adrenal cortex → Cortisol
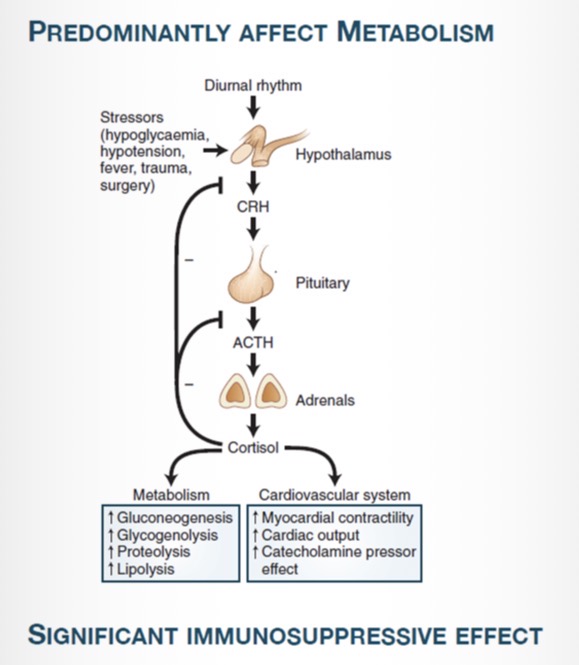
How is the HPA axis regulated?
By negative feedback- cortisol inhibits CRH and ACTH release
What is the purpose of negative feedback in the HPA axis?
To maintain stable cortisol levels
Describe the circadian rhythm of cortisol (graph).
Peak:
Lowest:
Peak: early morning (~6–9 am)
Lowest: midnight
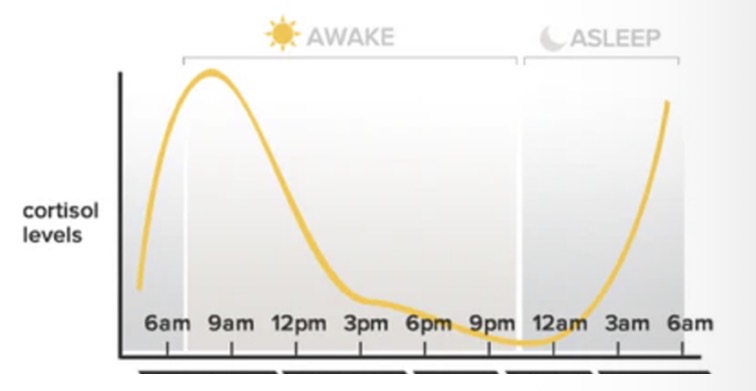
What controls cortisol’s circadian rhythm?
Hypothalamic signals and the sleep–wake cycle
Why is cortisol’s circadian rhythm clinically important?
Glucocorticoid therapy should mimic this pattern
What are 3 key HPA disorders?
Addisons disease/ Adrenal insufficiency → inadequate cortisol production
Cushing syndrome → excess cortisol production
Drug-induced suppression → risk of long-term glucocorticoid therapy
ACD
What 3 things triggers renin release?
Low blood pressure/ perfusion
Low sodium
Sympathetic activation
Outline the RAAS pathway.
Liver (angiotensinogen) → Renin → Angiotensin I → ACE (lungs) → Angiotensin II
What are the 4 effects of angiotensin II?
Aldosterone release
Vasoconstriction
↑ ADH from posterior pituitary + thirst from hypothalamus
↑ sympathetic activity
What is the main function of ADH?
Water reabsorption in the kidney
What are the 3 primary stimuli for aldosterone secretion?
Decreased blood volume and hyponatremia (via RAAS activation)
Indirectly hyperkalemia
Where does aldosterone act in the kidney?
Distal convoluted tubule and collecting duct
MOA of aldosterone
Aldosterone binds to mineralocorticoid receptor (MR / NR3C2)
The receptor-hormone complex translocates to the nucleus and alters gene transcription
It upregulates the epithelial sodium channel (ENaC)
What are the 3 effects of aldosterone at the kidney?
↑ Na⁺ reabsorption
↑ K⁺ excretion
↑ water retention (blood volume)
What is the key difference between HPA axis and RAAS/ADH systems?
HPA is a true endocrine axis; RAAS and ADH are coordinated but not a single regulated pathway- ADH and RAAS work together to maintain fluid balance and blood pressure
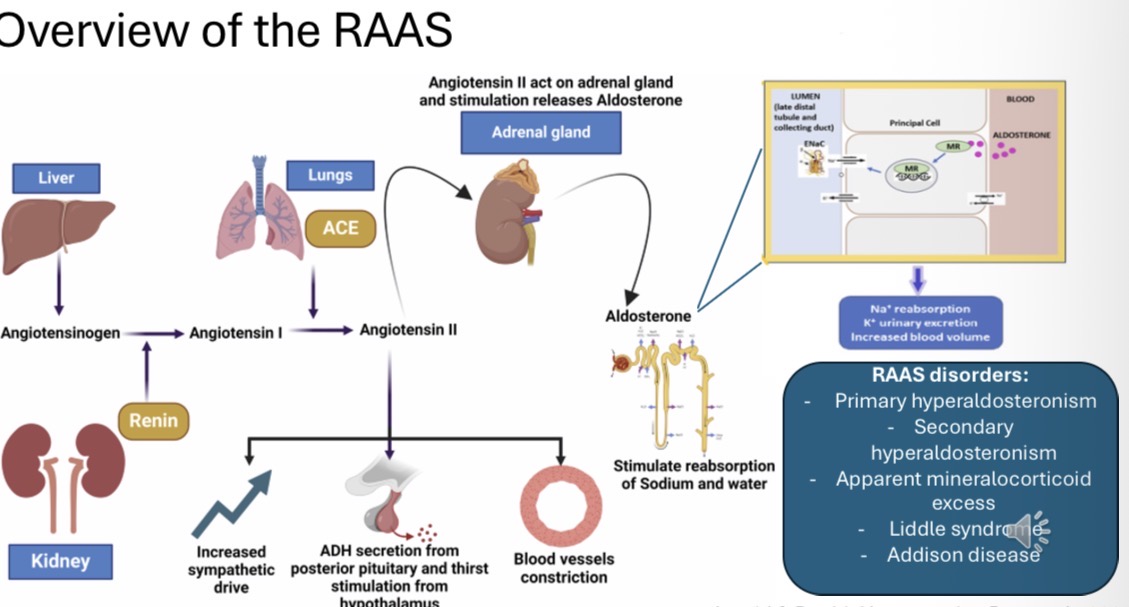
What 5 disorders are associated with RAAS?
Primary hyperaldosteronism
Secondary hyperaldosteronism
Apparent mineralocorticoid excess
Liddle syndrome
Addison disease
What is the common precursor of all adrenal steroid hormones?
Cholesterol
What is the rate-limiting step of steroidogenesis?
Transport of cholesterol into mitochondria by StAR (steroidogenic acute regulatory protein)
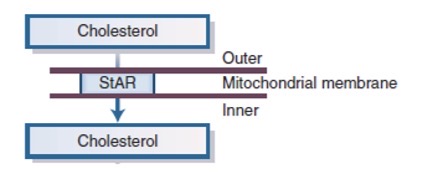
What happens to cholesterol once inside the mitochondria?
It is converted into pregnenolone
Which enzyme converts cholesterol to pregnenolone?
CYP11A1 (cholesterol side-chain cleavage enzyme)
What is the sequence of the first steps in steroidogenesis?
Cholesterol → (StAR transport) → mitochondria → CYP11A1 → pregnenolone
What is pregnenolone the precursor for?
All adrenal steroid hormones
Which enzyme families are mainly involved in steroid hormone synthesis?
Cytochrome P450 enzymes
Name 4 key enzymes involved in adrenal steroidogenesis.
CYP17
CYP21
CYP11B1
CYP11B2
Which 3 enzymes are required for cortisol synthesis?
CYP17
CYP21
CYP11B1
Which 2 enzymes are involved in aldosterone synthesis?
CYP21
CYP11B2
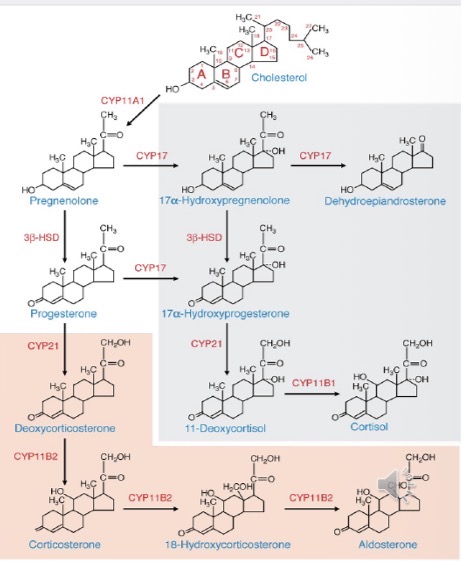
Why are steroidogenesis enzymes important in pharmacology?
They are targets for drugs treating cortisol excess
Which drug inhibits CYP11B1 and what is the effect?
Metyrapone → reduces cortisol synthesis
What type of molecules are steroid hormones?
Lipophilic hormones derived from cholesterol
Are steroid hormones stored in secretory vesicles?
No, they diffuse out once synthesized
Do steroid hormones circulate freely in blood?
No, they are mostly protein-bound to Cortisol-binding globulin (CBG)
Which other protein carries cortisol to a lesser extent?
Albumin
Which fraction of cortisol is biologically active?
The free (unbound) fraction
Where are steroid hormone receptors located?
Intracellular (cytoplasm/nucleus)
What are the 3 main adrenal steroid receptors?
Glucocorticoid receptor (GR)
Mineralocorticoid receptor (MR)
Androgen receptor (AR)
MOA of steroid hormones
Bind receptor → receptor dimerisation → enter nucleus → regulates gene transcription by binding to specific DNA sequences known as hormone response elements
What are the three main domains of nuclear receptors?
Ligand-binding domain
DNA-binding domain
Transcriptional activation domain
What is special about cortisol and MR?
Cortisol can bind MR
Why doesn’t cortisol normally activate MR in the kidney?
It is converted to inactive cortisone
What enzyme prevents cortisol from activating MR?
11β-HSD2 converts cortisol → cortisone (inactive)
What happens if 11β-HSD2 is inhibited? Give 2 effects
Cortisol activates MR → hypertension and hypokalaemia
What substance inhibits 11β-HSD2?
Glycyrrhizinic acid (found in liquorice)
What are the 3 effects of structural modifications on glucocorticoids?
Increase glucocorticoid activity (duration)
Alter mineralocorticoid activity
Increase potency
What is the effect of a double bond in the A ring?
Increased glucocorticoid activity
What is the effect of a CH₃ group at position 6?
Increased glucocorticoid activity
What is the effect of a CH₃ group at position 16?
Increased glucocorticoid activity
What is the effect of fluorine at position 9?
Increased glucocorticoid AND mineralocorticoid activity
What is the effect of CH₃ at C16 + F at C9?
Increased glucocorticoid activity (reduced mineralocorticoid effect)
What is the effect of double bond + CH₃ (C16) + F (C9)?
Increased glucocorticoid activity with almost no mineralocorticoid activity → strong anti-inflammatory effect with minimal salt-retaining effect
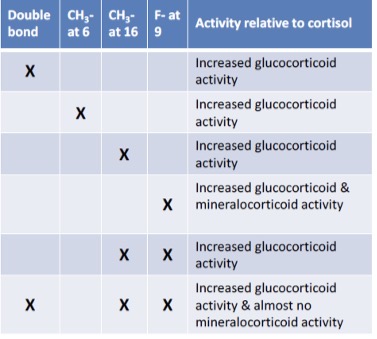
Which drug is an example of strong glucocorticoid activity with minimal mineralocorticoid effect?
Dexamethasone
What is the reference glucocorticoid for potency comparison?
Cortisol (potency = 1)
How potent are prednisone and prednisolone compared to cortisol?
~4 times more potent
Which 2 glucocorticoids have ~25× the anti-inflammatory potency of cortisol?
Dexamethasone and Betamethasone
Which corticosteroid has very strong mineralocorticoid activity (Sodium retention)?
Fludrocortisone
What is fludrocortisone mainly used for?
Mineralocorticoid replacement in adrenal insufficiency
Why are dexamethasone and betamethasone preferred in many conditions?
Dexamethasone and Betamethasone have strong anti-inflammatory effect without fluid retention (minimal mineralocorticoid activity)
Which 2 glucocorticoids are short-acting?
Cortisol and cortisone
Which glucocorticoids are intermediate-acting?
Prednisone and methylprednisolone
Which 2 glucocorticoids are long-acting?
Dexamethasone and betamethasone
What is the relationship between potency and duration of action in glucocorticoids?
Higher potency drugs are generally longer-acting
What are glucocorticoids used for? Give 3
•At high-dose (supraphysiologic) anti-inflammatory / immunosuppressive
•Used in inflammatory conditions both systemically and topically
•Immunosuppressive post-transplantation - avoid rejection
Name 4 common conditions treated with glucocorticoids.
Asthma
Rheumatoid arthritis
Inflammatory bowel disease
Allergic disorders
What are 8 major side effects of glucocorticoids?
•Long-term use - Iatrogenic Cushing’s Syndrome
•Thin Skin / Bruising
•Hypertension
•Osteoporosis
•Proximal Myopathies (muscle weakness)
•Drug-induced Diabetes Mellitus / Insulin Resistance
•Opportunistic Infections
• HPA axis suppressed → Secondary Adrenal Insufficiency if long term glucocorticoids suddenly stopped
What are the three main anti-inflammatory mechanisms of glucocorticoids?
Transactivation → ↑ anti-inflammatory proteins
Transrepression → ↓ inflammatory gene transcription
Post-transcriptional → ↓ cytokine mRNA stability
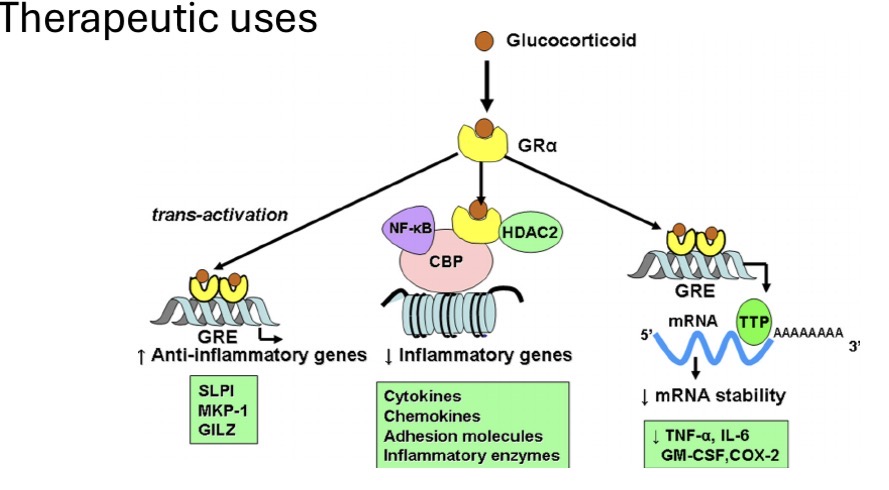
How do glucocorticoids exert anti-inflammatory effects via transactivation?
Glucocorticoids diffuse into the cell → bind GRα →
complex enters nucleus →
GR homodimer binds GRE →
↑ transcription of anti-inflammatory genes (e.g., SLPI, MKP-1, GILZ)
How do glucocorticoids suppress inflammatory gene transcription (transrepression)?
GR interacts with transcription factors (e.g., NF-κB) and coactivators (CBP)
→ recruits HDAC2 →
histone deacetylation →
↓ transcription of inflammatory genes (cytokines, chemokines, adhesion molecules, inflammatory enzymes)
How do glucocorticoids reduce cytokine production post-transcriptionally?
Glucocorticoids ↑ expression of tristetraprolin (TTP) →
TTP binds AU-rich regions of cytokine mRNA →
promotes mRNA degradation →
↓ cytokine synthesis (TNFa, IL6, GMCSF, COX2)
How is adrenal insufficiency classified?
Primary, secondary, or tertiary (based on level of defect in HPA axis)
What is primary adrenal insufficiency?
Failure of the adrenal gland → ↓ glucocorticoids + ↓ mineralocorticoids (↓ cortisol + ↓ aldosterone)
Name 5 causes of primary adrenal insufficiency.
Genetic disorders
Autoimmune adrenalitis
Metastatic cancer
Adrenal hemorrhage
Tuberculosis
What is the most common form of primary adrenal insufficiency?
Addison's disease
What is the treatment for primary adrenal insufficiency (Addison’s Disease)?
Lifelong hormone replacement therapy
Glucocorticoid replacement
Hydrocortisone (15–25 mg/day in divided doses)
Mineralocorticoid replacement
Fludrocortisone (0.05–0.2 mg/day)
When must glucocorticoid doses be increased in treatment of primary adrenal insufficiency?
During stress (infection, surgery, trauma)
What are the 10 symptoms of Addison’s disease?
Hyperpigmentation
Low blood pressure
Weakness
Weight loss
Nausea
Diarrhea
Vomiting
Constipation
Abdominal pain
Vitiligo
Why does hyperpigmentation occur in Addison’s disease?
↑ ACTH (from lack of cortisol feedback) → stimulates melanocytes
Why do patients with Addison’s disease have low blood pressure?
↓ Aldosterone → ↓ sodium and water retention → ↓ blood volume
What is adrenal crisis?
Life-threatening acute deficiency of cortisol
What can trigger an adrenal crisis? Give 3
Severe illness
Trauma
Abrupt withdrawal of glucocorticoids
What is the 3 step emergency treatment of Adrenal Crisis (Acute Adrenal Insufficiency)?
IV hydrocortisone (100 mg)
IV normal saline
Correction of electrolyte imbalance
What are the 6 symptoms of an Adrenal Crisis (Acute Adrenal Insufficiency)/ Addisonian crisis?
Fever
Syncope (severe hypotension)
Convulsions
Hypoglycemia
Hyponatremia (electrolyte imbalance)
Severe vomiting and diarrhea
What causes secondary and tertiary adrenal insufficiency?
Impaired stimulation of the adrenal cortex due to dysfunction in the HPA axis
What is secondary adrenal insufficiency?
Anterior pituitary failure → ↓ ACTH → ↓ cortisol
What is tertiary adrenal insufficiency?
Hypothalamic failure → ↓ CRH → ↓ ACTH → ↓ cortisol
What is the most common cause of tertiary adrenal insufficiency?
Chronic exogenous glucocorticoid use → atrophy
How do long-term glucocorticoids cause adrenal insufficiency?
Negative feedback → ↓ CRH & ACTH → adrenal atrophy
What happens to aldosterone in secondary/tertiary adrenal insufficiency?
Usually preserved (RAAS intact)
What replacement therapy is needed in secondary/tertiary adrenal insufficiency?
Glucocorticoids only (no mineralocorticoids)
What is the main drug used for replacement therapy in secondary/tertiary adrenal insufficiency?
Hydrocortisone- modified-release hydrocortisone formulations to mimic physiological cortisol rhythm
Name 2 modified-release hydrocortisone formulations used for replacement therapy in secondary/tertiary adrenal insufficiency
Plenadren
Chronocort
What is an alternative glucocorticoid for replacement therapy used for replacement therapy in secondary/tertiary adrenal insufficiency?
Prednisolone