CHEM2232 EXAM 2
1/64
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
65 Terms
Aromaticity criteria
cyclic, planar, fully conjugated, (4n+2) π electrons
Hückel’s rule
aromatic if (4n+2) π electrons, antiaromatic if 4n
Antiaromatic vs. nonaromatic
anti = planar + conjugated + 4n π
non = not fully conjugated or not planar
Frost circle purpose
determines aromaticity via MO energy levels; line up one corner in the middle of the circle
Frost circle rule
if all bonding MOs filled → aromatic
General EAS mechanism
electrophile attack → arenium ion → deprotonation restores aromaticity
Rate-determining step in EAS
formation of arenium ion
Activating groups
(EDG) donate electrons, increase rate, ortho/para directing
Deactivating groups
(EWG) withdraw electrons, decrease rate, meta directing
Halogens directing effect
ortho/para directors but deactivating
Ortho/para directors
-OH, -NH2, -OR, alkyl
Meta directors
-NO2, -CF3, -SO3H, carbonyls (-COR)
Blocking group purpose
control substitution position
Common blocking group
H2SO4, SO3 → SO3H (sulfonation)
How to remove SO3H
heat with H+, H2O (desulfonation)
Why protect -NH2 or -OH
too activating → causes polysubstitution
Common protection for amines
acylation (amide formation)
Kinetic product
forms faster, lower temp, less stable
Thermodynamic product
more stable, higher temp
1,2-addition
kinetic product
1,4-addition
thermodynamic product
Diels-Alder reaction
[4+2] cycloaddition between diene and dienophile
Endo rule
electron-withdrawing groups go endo (preferred)
Diene requirement
must be s-cis
Dienophile requirement
electron-poor alkene
Electrocyclic reaction
ring opening/closing via π electrons
4n system (thermal)
conrotatory
4n+2 system (thermal)
disrotatory
Halogenation of benzene
Br2/FeBr3 or Cl2/FeCl3 → substitution
Nitration
HNO₃, H₂SO₄ → NO₂ added
Sulfonation
SO₃, H₂SO₄ → SO₃H added
Hydrogenation
benzene + H2, Pd-C → cyclohexane
Friedel-Crafts alkylation
R-Cl, AlCl3 → adds alkyl group
Limitations of Friedel-Crafts alkylation
rearrangements occur
Friedel-Crafts alkylation fails when
strong deactivators present
Friedel-Crafts acylation
acyl chloride/AlCl3 → adds ketone
Advantage of Friedel-Crafts acylation
no rearrangement
Nitro → amine
reduction (H2, Pd-C or Fe or Zn, HCl)
Clemmensen reduction
carbonyl + Zn(Hg), HCl → alkane
Wolff-Kishner reduction
carbonyl + NH2NH2, EtOH, heat → alkane (basic conditions)
use when acetal is present (protected ketone/aldehyde)
Benzylic oxidation
Ar–CH₃ → Ar–COOH
Ar–CH₂R → Ar–COOH
Ar–CHR₂ → Ar–COOH
CrO₃, H₂SO₄, H₂O
PCC
alcohol (-OH) → aldehyde/ketone (no overoxidation)
Chromic acid (CrO₃, H₂SO₄, H₂O)
alcohol (-OH) → carboxylic acid (-COOH)
Ozonolysis
1) O₃, CH₂Cl₂
2) S(CH₃)₂
splits double bond → carbonyls (ketone/aldehyde)
Alkyne hydration (Markovnikov)
alkyne + HgSO₄, H₂SO₄, H₂O → ketone
Alkyne hydration (anti-Markovnikov)
internal alkyne + 1) BH₃, THF; 2) NaOH, H₂O₂, H2O → ketone
terminal alkyne + 1) BH₃, THF; 2) NaOH, H₂O₂, H2O → aldehyde
Hydride reduction (LiAlH₄, strongest/NaBH₄, mild)
LiAlH₄ reduces aldehydes, ketones, esters, and carboxylic acids → alcohols
amides/nitriles to amine
NaBH₄ reduces aldehydes, ketones → alcohols
Organometallic addition (RMgX,
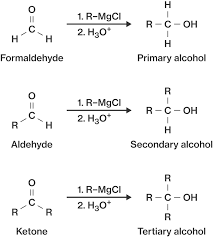
Hydrate formation
carbonyl + water → geminal diol (2 -OH groups attached to the same carbon atom)
Hemiacetal
carbonyl + alcohol (1 eq) → 1 -OH, 1 -OR
Acetal
carbonyl + alcohol (2 eq, acid) → 2 -OR
Acetal purpose
protecting group for aldehydes and ketones (acidic conditions)
Acetal hydrolysis
H++ water → carbonyl
Thioacetal formation & purpose
carbonyl + thiol (-RSH)
used as a protecting group for ketones and aldehydes (basic conditions—use when molecule is acid-sensitive)
Thioacetal removal
H2, RaNi
Imine formation
carbonyl + primary amine (acidic conditions)
Enamine formation
carbonyl + secondary amine (acidic conditions)
Imine/enamine → carbonyl
H+, H₂O
Cyanohydrin formation
carbonyl + CN⁻ → adds -CN + -OH
Wittig reaction
aldehyde/ketone + R-PPh3 → alkene
How to approach EAS problems
identify directing groups → predict position
How to approach synthesis
work backwards from product
Biggest Friedel-Crafts mistake
ignoring rearrangement
Most important carbonyl concept
protecting groups (acetals)
Most tested pattern
EAS + carbonyl combined synthesis