TC - UV Vis and Fluorescence Spectroscopy
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Why is UV used?
UV/Visible light photons have the right amount of energy to promote bonding/valence electrons to higher energy levels.
Sometimes called - electronic spectroscopy
Equation linking Planck’s constant, speed of light and wavelength

Define energy gap
The highest wavelength/lowest energy peak on the spectrum
Typical units of electron volts are used
Vibrational energy levels
Vibrational energy levels are approx 100 times lower in energy than electronical energy levels.
Each electronic energy level is associated with many vibrational energy.
Jablonski diagram
A molecule absorbing UV - Vis light not only causes an electron to move from the electronic ground state to an electronic excited state, the newly excited molecule can also access a higher vibrational level.
Accessing of these vibrational levels provides the continuous structure of a UV/Vis spectrum.
The accessing of these vibrational levels provides the continuous structure with a UV/Vis spectrum.
Jablonski diagram is a schematic that incorporated vibrational energy levels and electronic energy levels an vibro-electronic transitions.
Non - radiative processes
Vibrational relaxation involves the excited molecule losing energy by vibrating and ends up in the vibrational ground state (while still being in the electronic excited state)
Then, the molecule can lose further energy by vibration but also by collision and other process, summarised by internal conversion.
While each collision can occur quickly, often many collisions are required for the molecule to return all the way to the electronic ground state.
Define chrorophore
The region of the molecule most responsible for absorbing UV/Vis radiation.
What is Beer’s Law?
Quantitative analysis method of UV/Vis spec
Was determined experimentally over 100s of years
Observing effects of visible light on a liquid as a function of distance
Beer’s Law Equation

Beer’s Law Consequences
Plot of conc against absorbance will yield a straight line
Gradient will be equal to eb
Define limit of detection
The smallest amount that can be measured accurately by an instrument.
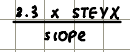
Absorbance and transmittance
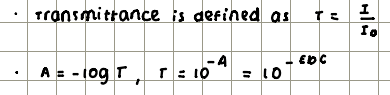
Polychromatic Deviation
Monochromator passes light into the sample only several wavelengths
Depending on the bandpass, and what wavelengths you choose to scan at, your absorbance response may vary considerably

Scattered light deviation
Although slits try to prevent stray light from reaching the detector, there will likely be a scattered light present - enhanced with a larger bandpass, and a longer b (path length)
It can also be caused by pollutants/solid matter in the sample
If sides of the cuvette get marked, light can be scattered
Ensure samples contain no solid particles/dirt
Wear gloves when placing cuvettes
Analyte/solvent molecules could also scatter light, so scattering cannot be eliminated entirely
Chemical based deviations
Beer’s law assumes that analyte molecules are far enough apart that each absorbs light independently from another.
In the high concentration limit, these assumption break down and affect e values
These can be caused by charge effects between analyte molecules
Additional deviations and fixes
In high concentrations it is possible that a molecule changes its optical properties - change in refractive index could accompany a phantom “jump“ in absorbance
All instrument-based deviations (polychromatic and scattered light) results in negative deviations
Generally most deviations can be fixed by staying in the low concentrations and low absorbance ranges, whilst keeping sample clean, and measuring at absorbance peaks with a low bandpass.
Define fluoroescence
Emission of radiation from the first excited state
Define phosphorescence
Emission of radiation from a triplet excited state
What is intersystem crossing
Where spin randomly occurs from a singlet to triplet states - slow process but can occur, also depends on the molecule.
Define stokes shift
Difference between lambda max values
Depends on molecule, and solvent system
A secondary identifying feature of an analyte
Mirror image rule
Vibrational levels in the excited ground states are similar.
Absorption spec involves vibrational of S1
Emission spectrum involves the vibrational levels of S0
Energy difference between S0V0 - S1V0 and S0V0 - S1V1 absorptions is similar enough to the difference between S1V0 - S0V0 and S1V0 - S0V1 emissions.
However, because fluorescence can only occur from S1 (V0), any spectrum that contains multiple electronic transitions. - breaks mirror image rule.
Even if fluorescence was notable from S2-S0, there still wouldn’t be a mirror image because energies of these transitions would be shorter un wavelength than S1-S0.