Chemical formulae, equations, calculations (1e)
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
Law of Conservation of Mass
Total mass of reactants is always equal to total mass of products
RFM Calculation
multiply each Ar by how much there is
add them together
Avogadro’s Number
6.023 × 1023
Moles and Mass
n = M/Mr
Calculating Molar Mass/ no. of moles
calculate Mr
divide mass given by Mr
Calculating Reacting Mass
find reacting moles (given or calculate with M / Mr)
find ratio between given substance and substance to find
find no. of moles
use no. of moles to find mass
Balancing Equations using Reacting Mass
write unbalanced equation
write down masses
calculate moles using mass and Mr
use moles to find ratio
use ratio to balance equation
Reasons for not getting 100% yield in a reaction
some reactants left behind
reaction may be reversible- high yield is impossible since products are continually turning back into reactants
some products lost during purification or separation stages like filtration or distillation
side reactions: substances reacting with gas in the air or impurity in reactant
products lost during transfer between containers
Thoretical Yield
amount of product that would be obtained under perfect practical and chemical conditions
calculated from balanced equation and the reacting masses
Actual Yield
recorded amount of product obtained
always less than theoretical yield
Percentage Yield
compares actual yield to theoretical
for economic reasons, objective of every company is to have yield % as high as possible → to reduce costs & wastes and increase profits
good way to measure how successful a chemical process is
Calculating Percentage Yield
find actual yield (usually given)
find theoretical yield (calculate using moles and mass)
use equation: (actual/theoretical) x 100
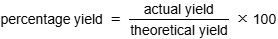
Experiment (Finding Formulae of Simple Compounds): Aim
to determine formula of hydrated copper sulfate: CuSO4.xH2O
Experiment (Finding Formulae of Simple Compounds): Method
measure mass of evaporating dish
add known mass of hydrated salt
heat over bunsen burner, gently stirring
stop when salt turns from blue to white (all water lost)
record mass of dish and contents
Experiment (Finding Formulae of Simple Compounds): Overheating the salt
decomposes and gives a larger change in mass
Experiment (Finding Formulae of Simple Compounds): Results
mass of white anhydrous salt
measure mass of white anhydrous salt (mass of salt remaining)
mass of water
subtract mass of white anhydrous salt from mass of known hydrated salt
divide mass of the salt and water by masses (moles)
simplify the ratio (multiply by 2 if decimal)
find ratio as 1:water
represent ratio as ‘salt.xH2O)
Practical (Determining Formula of Magnesium Oxide): Aim
To determine the empirical formula of magnesium oxide by combustion of magnesium
Practical (Determining Formula of Magnesium Oxide): Diagram
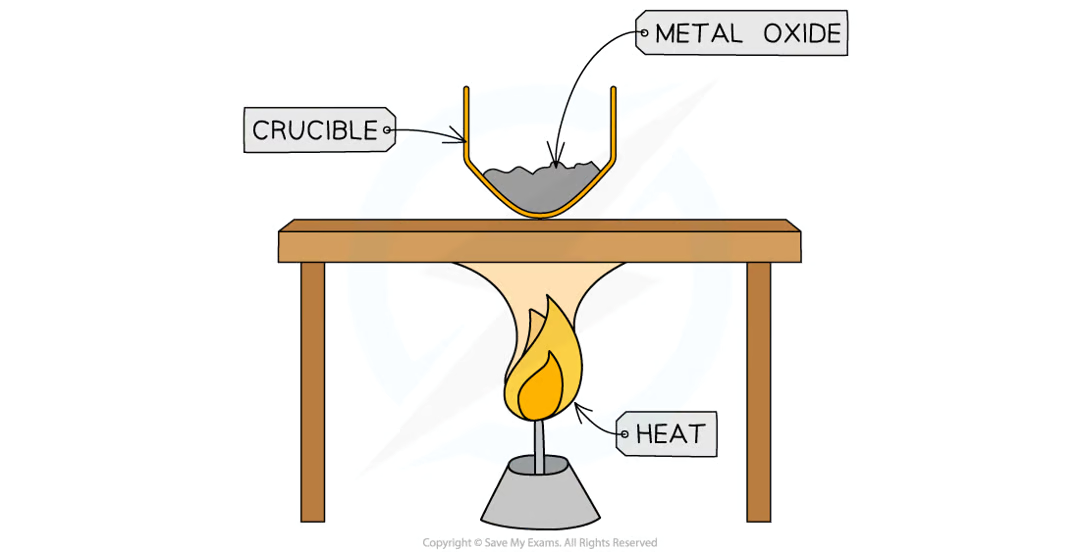
Practical (Determining Formula of Magnesium Oxide): Method
measure mass of crucible with lid
add Mg sample to crucible and measure mass with lid
measure
strongly heat crucible over Bunsen burner for several minutes
lift lid frequently to allow sufficient air into crucible for Mg to oxidise without letting MgO smoke escape
continue heating until mass remains constant (max mass) → reaction is complete
measure mass of crucible and contents
calculate mass of crucible and contents by subtracting mass of empty crucible
Practical (Determining Formula of Magnesium Oxide): Results
find mass of metal by subtracting mass of crucible from Mg and mass of empty crucible
subtract mass of Mg used from mass of MgO
divide each mass by Ar,
simplify ratio (multiply by 2 if decimal)
represent as MxOy
Practical (Determining Formula of Copper(II) Oxide): Aim
To determine the formula of copper(II)oxide by reduction with methane
Practical (Determining Formula of Copper(II) Oxide): Diagram

Practical (Determining Formula of Copper(II) Oxide): Method
measure mass of empty boiling tube
place metal oxide into a horizontal boiling tube and measure mass again
support tube in horizontal position by clamp
natural gas(methane) is passed over copper(II) oxide and excess gas is burned off
copper(II) oxide is heated strongly with a Bunsen burner
heat until metal oxide fully changes colour (all oxygen removed)
measure mass of remaining powder in the tube and subtract mass of tube
Practical (Determining Formula of Copper(II) Oxide): Results (Empirical Formula)
measure mass of powder to find mass of metal
divide masses by Ar
simplify ratio
represent ratio as MxOy
Molecular Formula
Formula showing number and type of each atom in a molecule. Ex: ethanoic acid is C2H4O2
Empirical Formula
Simplest whole number ratio of atoms of each element present in one molecule or formula unit of the compound. Ex: ethanoic acid is CH2O
Ionic compounds are always _________ formulae
Empirical
Calculating Empirical Formulae
write element
write value given (% or mass)
write Ar
calculate moles by m/Mr
calculate ratio of moles (multiply to make all values whole numbers)
write final empirical formula
Calculating Molecular Formula
find Mr of empirical formula (add masses of all atoms in the empirical formula
divide Mr of molecular formula by Mr of empirical formula
multiply each number in empirical formula by answer to find molecular formula
Calculating Concentration of Solutions in mol/dm3
number of moles (mol) / volume (dm3)
Avogadro’s Law
At the same conditions of temperature and pressure, equal amounts of gases will occupy the same volume of space
Molar Gas Volume at RTP
24dm3 or 24000 cm3
RTP
room temperature and pressure (20oC and 1atm)
Gas Volume Equations
volume = moles x 24 (dm3/mol)
volume = moles x 24000 (cm3/mol)
Metals + Cold Water Reaction Speeds
K : violent
Na: quick
Li and Ca: less strong
Fe: slow rust
Mg, Zn, Cu: no reaction/very slow
Metal + Water Reaction Format
metal + water —→ hydroxide + hydrogen gas
Metals and Acids Reaction Rate
only metals above Hydrogen
more reactive metal = more vigourous reaction
K and Na are very dangerous and react explosively
Metal + Acid Reaction Format
Metal + Acid → Salt + Hydrogen