542 - Kwon Sterile product requirement (copy)
1/33
Earn XP
Description and Tags
KWON
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
top 10 excipients in injectable products
water
cosolvent
oils
surfactant
antimicrobial agent
buffer
antioxidant
tonicity agent
cryoprotectant/lyoprotectant
competitive binding
Top 7 requiremnts of injectable products
purity
sterility
sterile filtration
particulates
antimicrobial agents (such as perservatives)
pyrogens
isotonicity
sterility
complete destruction of all living organisms and their spores or complete removal
what sterile products are susceptible to microbial contamination?
aq solution
emulsions
suspensions
D-value
time in mins required to destroy 90% of microbes udner a standard set of conditions
D= 1 min —> 10^6 organisms goes to 10^5 after 1 min
biological indicator
specific microorganisms resistant to a particular method of sterilization
Validation of sterility
add at known level and known D value and sterilize - survival should be 10^-6 or less
List the Sterilization techniques (4)
steam
dry heat
radiation
Gas
Steam sterilization
heating under pressure in the presence of water to generate steam
moist heat causes irreversible denaturation of essential proteins
this can damage oils, proteins, and powders
D = 1 min at 121 C
Dry heat sterilization
done in sterilizing ovens
mech of microbial destruction is dehydration, denaturation, and slow burning or oxidation
D = 5 mins at 160 C
Radiation sterilziation
gamma radiation
mech: ejects electrons nad creates free radical that reacts iwth biological macromolecules, esp DNA< leading to cell damage and death
ionizing radiation can damage biologic drugs
gas sterilization
Ethylene oxide (EO) + heat + moisture
EO = alkylating agent + reacts with amine groups of DNA
Residual EO must be allowed to dissipate after sterilization and before use of sterile product
Sterile filtration
physical removal of particles and microorganisms form solution
Depth filter
substance that trap particles in channels
Screen filter
surface filtration - cellulose esters
Mcirobial filter
0.22uM
particulate - free
particles can be emboli (matters carried in blood) that can get stuck in a BV, obstructing it and resulting in a blockage —> embolism
list antimicrobial agents
benzyl alcohol
IV can cause death of neonates given preserved saline
Intrathecal can increase risk of neurological events
epidural toxicity
Pyrogens
fever causing organic molecules
primary source of pyrogens
water
Sources of microbial contamination (7)
Raw material
water
air
processing operations
equipment
employees
environment
Approaches to sterility (4)
initial control
terminal sterilization (product in primary packaging)
aseptic processing and sterile filtration for biologics
antimicrobial agents
Sterility assurance level (SAL)
1/10 ^6
Major factors of mcirobial growth and death (7)
temperature
time of storage
C_o
Chemical (Nutrients, ph)
Gaseous environment (oxygen)
Moisture content
radiation
Limulus amebocyte lysate test
Type of pyrogen test
a gel forms in the presence of pyrogen
Rabbit febrile reaction test
type of pyrogen test
elevation of >0.6 C above normal is the basis for failure of the test (from animals rectal temp
Lipopolysaccharide (LPS)
cell wall of grame negative bacteria aka endotoxin
maybe remain thermostable even after steam sterilization or bacterial filtration
List situations where you wouldn’t need an antimicrobial agents
hydroalcoholic preperations, >15% alcohol
hypertonic solutions, 67% sucrose in syrups (microorganisms cannot grow)
pH < 3 (pickling)
pH >9(too basic)
multiple dose products
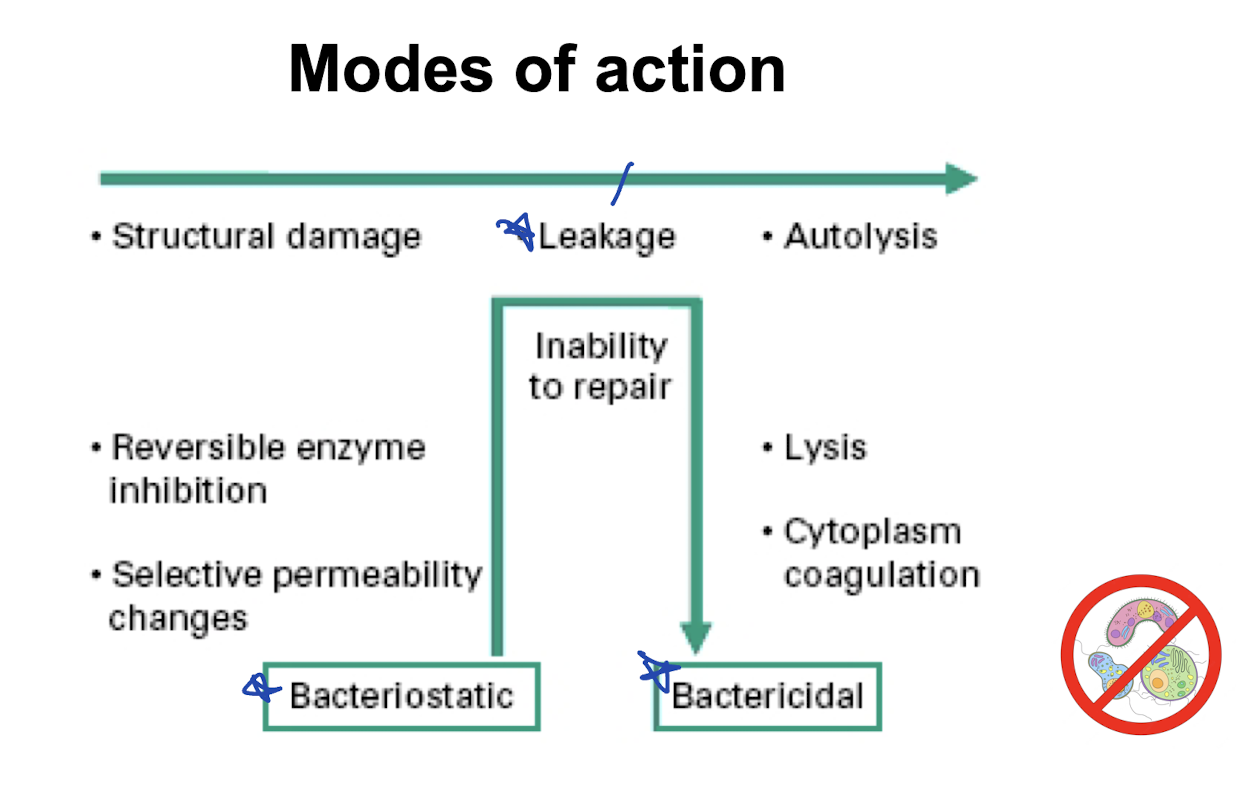
Bacteriocidal
Kills organisms
Bacteriostatic
Prevents growth
Modes of action
Sources of Particulate - free? (7)
bacteria/fungi
protein aggregation/ precipitation
incompletely dissolved drug
precipitate from incompatible admixtures
glass particles from ampoules
particles from rubber stoppers
particles form plastic
when are incidence of phlebitis reduced?
when IV infusions are filtered (final filter)
Phlebitis
inflammation of vein
pain and tenderness along course of vein