Bio 231 - Cell Cycle Control, Death, and Cancer
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
What is the cell cycle?
The orderly sequence of events in which a cell duplicates its contents + divides in two. Occurs in two major phases:
Mitotic phase
Mitosis - nuclear division
Cytokinesis - cytoplasmic division
Interphase
G1
S phase
G2
What are the different stages of interphase? What are their major functions?
G1 (first gap)
Cell grows in size and mass
Macromolecule synthesis
Organelles duplicated
Duration is variable: Short (min-hr) in rapidly dividing cells, such as embryo or stem cells; Long (hour-week-year) in slowly or non-dividing cells
S phase
DNA synthesis
Required prior to cell division (mitosis)
G0 phase
Cells exit cell cycle + arrest in non-mitotic state
May occur in either G1 or G2 (perhaps G1 more common)
Virtually absent in highly mitotic cells, such as stem cells (haematopoietic, epithelial) or germ cells
Temporary in some cells, such as liver cells or resting leukocytes
Permanent in some cells, such as brain or muscle
What is the importance of cell cycle regulation? How does the G0 phase help out a cell?
Ensures events required during each cell cycle phase are carried out
Ensures cell has ability to respond to external conditions: growth factors; nutrients
Some cells need a time out - cell cycle phase is not complete or cell may have repairs to make
G0 phases give cells a chance to catch a breath before carrying on to either cell cycle progression or programmed cell death
What are the three checkpoints of cell cycle regulation? What allows a cell to move forward past each checkpoint (what qualifiers must they have)?
Restriction point (G1 checkpoint): near the end of G1 phase
Cells check:
Size (halts cycle for further growth if necessary)
DNA damage (halts if too badly damaged to replicate)
Nutrients (halts if insufficient)
Growth factors (checks for extracellular signal proteins that stimulate cell division)
If cells do not pass G1 checkpoint, they enter G0
G2-M transition (G2 checkpoint): near end of G2
Cells check:
Completion of DNA synthesis (halts if incomplete)
DNA damage (halts until repairs are complete)
Available space (halts if no room to divide)
If cells do not pass G2 checkpoint, they enter G0
Metaphase-Anaphase transition (Mitosis checkpoint)
Cells check:
Chromosomes; must be properly attached to spindle fibres before separation begins
If cells do not pass checkpoint ?
What regulatory proteins control the cell cycle?
Passage through cell cycle checkpoints controlled by key regulatory proteins
Protein kinases regulate protein phosphorylation (ATP → Pi → protein)
Phosphatases regulate protein dephosphorylation (remove Pi from a protein)
Both are cativated at critical times (such as the checkpoints), and then deactivate
What do Cyclins and Cyclin-dependent kinases contribute to the cell cycle?
Cyclins:
Concentration increases gradually and peaks just before checkpoint
Responsible for controlling Cdk activity
Cyclin-dependent kinases (Cdk):
Cdk control passage of cell through checkpoints
Cdk concentration remains constant throughout cell cycle
Cdk activation dependent upon cyclin level; occurs just before checkpoints
What cellular components does Cdk target to trigger mitosis? (phosphorylate)
Cell cycle progression is controlled by checkpoints; checkpoints are controlled by Cdk; Cdk control cell cycle by phosphorylating proteins required for mitosis
Lamin proteins in nuclear lamina help provide structure to nucleus
Phosphorylation results in lamin depolymerisation
Facilitates degradation of nuclear envelope
Tubulin phosphorylation triggers formation of mitotic spindles
Histone phosphorylation promotes DNA condensation into chromosomes
Condensin complex protein phosphorylation triggers chromosomal condensation
What controls a cells entry into the S-phase? What signal transduction pathway mediates the activation of Cdk-cyclin?
G1 checkpoint
Restriction point is key entry way into cell cycle
Growth factors (ie. epidermal growth factor, EGF) often act via RTK signal transduction via the Ras-MAPK pathway
Explain the 8 steps of the Ras-MAPK pathway, and how it leads to G1 Cdk.
EGF binds to RTK
Receptor dimerization + autophosphorylation
Recruitment of proteins with an SH2 domain: GRB2 (intermediate protein with SH2 domain + Sos binding domain)
Sos recruited by GRB2
Sos activates Ras (by GDP/GTP replacement)
Ras-GTP triggers phosphorylation cascade (Raf → Raf-P →MEK → MEK-P-P → MAPK → MAPK-P-P → C-Myc → gene expression
c-Myc is a transcription factor; promotes the transcription of a G1 cyclin (cyc D)
G1 Cdk can now be turned on!
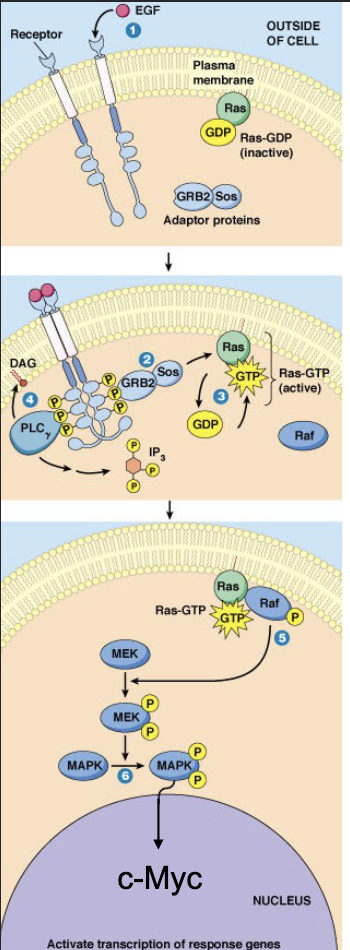
How does the Ras-MAPK pathway play a role in DNA replication?
At G1 checkpoint, the cell is making a decision to replicate DNA - if a cell replicates DNA, it will likely proceed to mitosis
Therefore, growth factors (such as EGF) help drive the decision to replicate DNA
The Ras-MAPK pathway results in cyclin (cyc D) expression
With cyclin D expression, G1 cdk is turned on
Recall the function of cdk (a kinase) is to phosphorylate proteins → G1 cdk target protein is retinoblastoma
What protein does cdk target? What is its functional role?
Rb protein acts as a molecular switch that controls another transcription factor (E2F); E2F activates transcription of genes required for DNA replication
In a de-phosphorylated state (with no phosphate), Rb binds to E2F
E2F becomes inactive and cannot promote expression of S-phase genes - meaning cells remain arrested (G0 lock)
Once G1 Cdk is active, Rb is phosphorylated → Rb can no longer bind to E2F, so E2F is free to activate S-phase gene - meaning cell passes through G1 checkpoint into S phase
What inhibits cyclin-cdk after it has done its job?
Cyclins ubiquitinated (marked for destruction) + degraded by proteasomes immediately after cell passes through checkpoint
Cdk subsequently returns to inactive state
What are the two important areas where apoptosis (programmed cell death) matters? What function do they play?
Embryonic development
Cells with DNA damage (ie. defective cells)
Removal of excess cells (ie. embryonic nerve cells are produced in huge excess and compete for target cells to innervate; extra cells programmed to die (apx. 50%)
Removal of tissue to open mouth and eyes
Elimination of tissue as fingers, toes (separation)
Organogenesis and organ sculpting to size
Elimination of self-reactive immune cells (T cells)
Important in adult tissues
Organ size maintenance (organs adjusted to size)
Cells at end of life span (Hayflick number reached)
Bone marrow and intestine (billions per hour)
Cells infected with pathogen (e.g. virus)
Immune cells no longer needed
Cells with DNA damage (could become cancerous)
Cancer cells - in early stages (most of the time)
What proteolytic enzyme catalyzes apoptotic cell death? How are they triggered into action? What are their primary targets?
Caspases - Proteolytic enzymes that cleave at aspartic acid residues. Present as inactive procaspases in all normal cells, which get activated by proeolytic cleavage (post-translational processing)
Caspase activation triggers caspase cascade; results in caspase-3 activation (the executioner caspase) → initiates apoptotic cell death
Executioner caspases degrade a select group of essential cellular proteins:
Protein kinases controlling cell adhesion
Lamins of the nuclear lamina
Cytoskeleton components
DNA repair enzymes
Regulatory protein that inhibits ladder DNAase (enzyme responsible for DNA fragementation
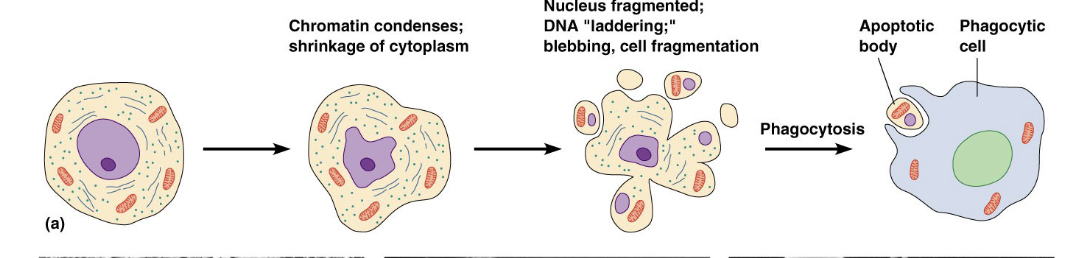
Explain the general steps of apoptotic cell death. How does the cell disassemble?
Cell quietly disassembles itself from within - DNA condenses + fragments (DNA laddering); cytoskeleton collapses; cell shrinks (cytoplasmic volume decreases)
Cell fragments into blebs, but does not leak! Blebs are pieces of a plasma membrane containing cellular constituents (aka apoptotic bodies), meaning there is nothing to stimulate inflammation.
Blebs are quietly removed by phagocytic cells, ie. macrophages
Phagocytes recognize newly exposed ‘eat me’ signals on the apoptotic bodies (blebs) which are typically not present on healthy cells
The ‘eat me’ signal is phosphatidylserine (PS), which is regularly maintained in the plasma membrane inner leaf by phospholipid translocases (flippases) that make sure it stays inside
During apoptosis, the flippase is inactivated, causing PS to accumulate on the outer leaf of the cells membrane (“EAT ME!”)
How does necrotic cell death differ from apoptotic cell death?
Necrotic cell death:
Damaged cells swell and rupture (lyse), causing a cells content to leak
Intracellular contents are ‘foreign’ to other cells = inflammation → more tissue damage
In contrast, apoptotic death is non-inflammatory (which is important since inflammatory responses can be highly damaging)
How is the apoptosis pathway activated?
Apoptosis is normally suppressed in cells
Apoptosis is activated only in response to signals engaged by different pathways, including:
Extracellular triggers
Survival withdrawal triggers (withdrawal of survival signals / trophic factors - informs cell to suppress apoptosis)
Intracellular triggers
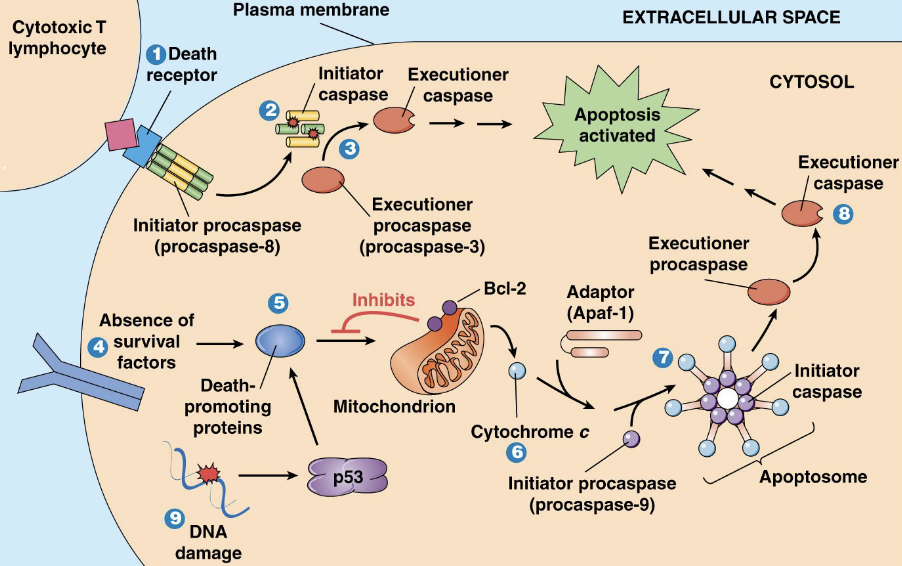
How can apoptosis be extracellularly triggered?
Extracellular signal molecule Fas ligand (FasL) is expressed on the surface of lymphocyte (leukocyte - WBC)
FasL binds to apoptosis-specific death receptor Fas (CD95), which is found in the plasma membrane of target cells
Fas-FasL interaction directly activates caspase cascade:
Inactive procaspases → active caspases
Caspases act in a series to activate final executioner caspase (caspase-3), which initiates apoptosis in cell
How can apoptosis be intracellularly triggered?
Intracellular triggers:
DNA damage (very strong activator)
Early stages of conversion to cancer cell
Severe oxidative stress (ie. free radical production)
Pathogen infection (virus, bacteria, protozoan)
Generally (though not always), apoptosis is initiated intracellularly by a cytoplasmic protein known as p53 - which activates pro-apoptotic proteins (ie. Bad/Bax)
In normal, healthy cells, Bcl-2 (associated with mitochondrion) inhibits apoptosis - as it is an anti-apoptotic protein
When pro-apoptotic Bad / Bax accumulates in mitochondrial outer membrane, apoptosis is initiated
Pro-apop Bad / Bax overcome anti-apoptosis Bcl-2
Bad/Bax binding causes mitochondria to release cytochrome c
Cytochrome c release triggers mitochondrial Ca2+ release
Ca2+ activates initiator procaspase 9 → caspase cascade
Caspase 9 activates executioner procaspase 3
What causes cells to be labelled cancerous? What are the two major groups of genes that can be very harmful if mutated?
Cancer results from mutations that liberate cells from usual controls
If cell death is controlled → cells that should die, live instead
If the cell cycle is controlled → prolonged cell proliferation (harmful cells)
Two major groups of genes where mutations can have disastrous consequences: Oncogenes and tumour suppressor genes