Biology Exam 2 Conent
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Anabolic
small molecules assemble into large ones. consumes energy (ATP)
Ana needs energy to go uphill
reactants are more stable
overall loss of energy (because the reaction consumes it in order to proceed)
Catabolism
large molecules break down into small molecules and release energy
“cat slides down the hill”
products are more stable
overall release of energy
ΔH > 0
(Endothermic): The system absorbs energy, requiring heat input.
ΔH < 0
signifies that a reaction is exothermic, meaning it releases heat to its surroundings
kinetic energy
intrinsic, random movement, creates disorder, increases with heat
chemical energy
is a type of potential energy within chemical bonds
depends on the position of electrons within a bond
is related to bond type (nonpolar vs polar)
Polar bonds have the electrons in a lower energy state because electronegative atoms can hold those electrons better
Nonpolar bonds have a higher potential energy because they could at some point fall to a lower energy state
enthalpy
the total energy of all the products minus the total energy of all the reactants
TΔS
unusable energy in a reaction
How do you know if reactants or products are more stable?
High-entropy states are more stable because they are more statistically probable, so sides with a bunch of smaller molecules will be favored
having more stable bonds (polar bonds)
exergonic (-ΔG)
reaction is spontaneous and downhill
the reactants have more energy than the products
endergonic (+ΔG)
reaction requires energy in order to occur and is uphill
the products have more energy than the reactants
3 ways to drive endergonic reactions
increase temp
metabolic pathways
inject energy into system
metabolic pathways
instead of a single, massive reaction, pathways break processes into small, manageable steps. The product of one enzyme-catalyzed reaction becomes the reactant (substrate) for the next.
types: anabolic and catabolic
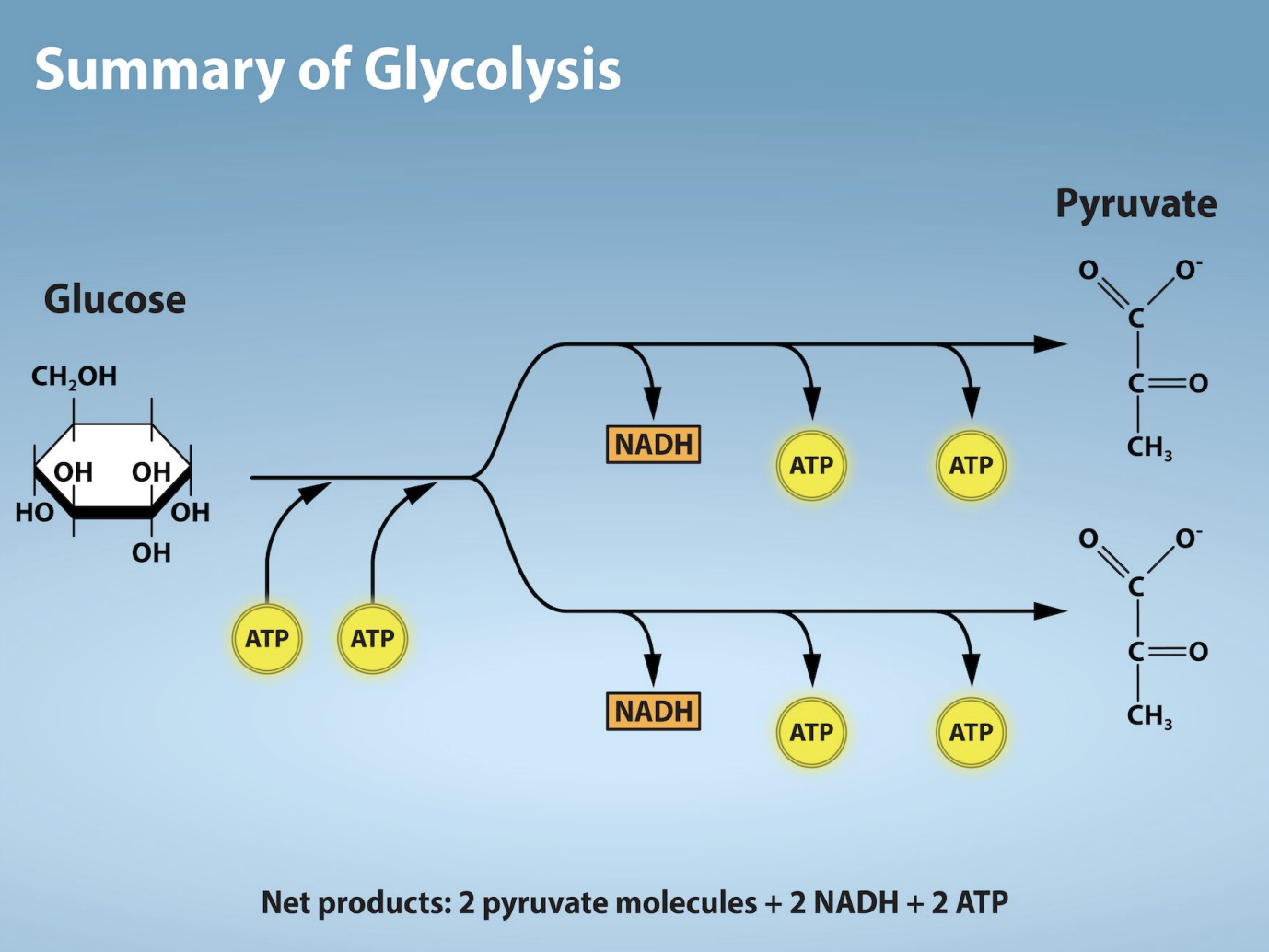
Glycolysis
What: A catabolic process in which a 6-carbon sugar molecule (glucose) is split into two 3-carbon molecules called pyruvate.
Where: In the cytoplasm (the jelly-like fluid inside the cell).
Results in: 2 pyruvate, a small amount of energy (net 2 ATP), and 2 NADH.
How: Requires an initial input of 2 ATP but does not require oxygen (anareobic).
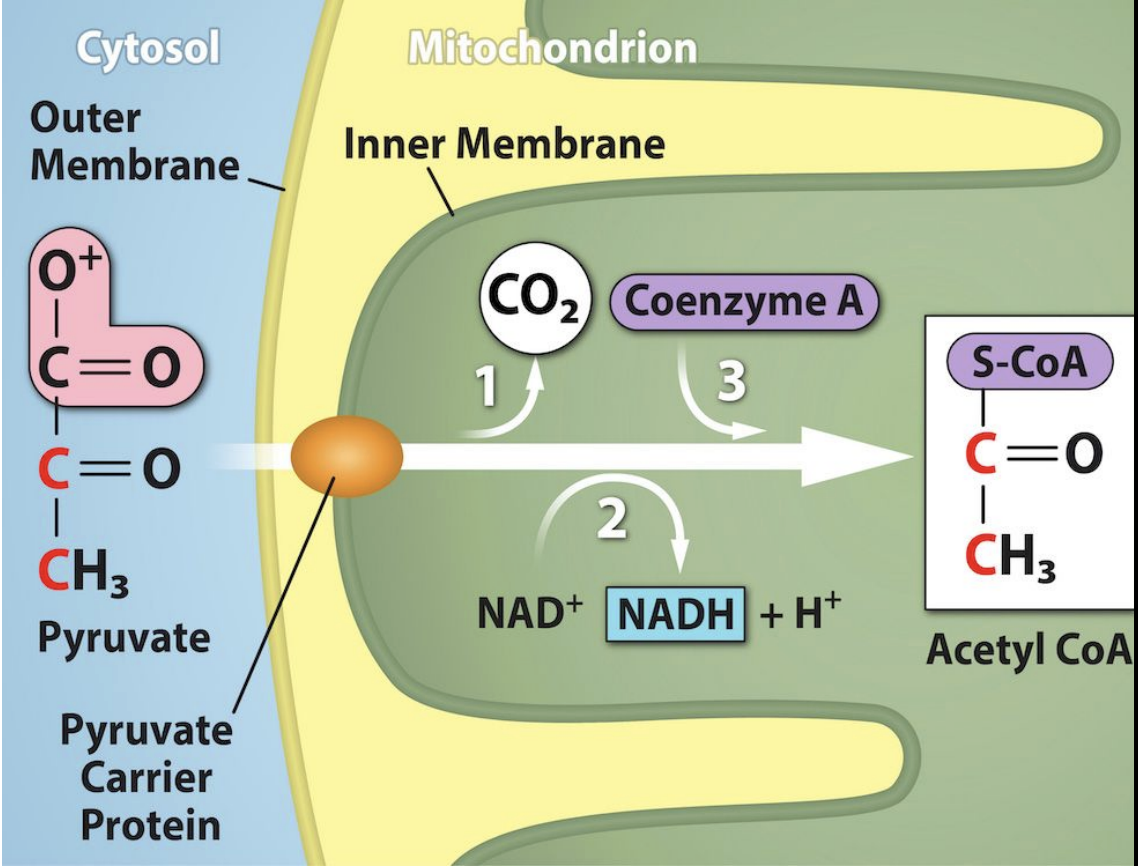
Pyruvate Breakdown
Where: Pyruvate is made in the cell’s cytoplasm during glycolysis. It must move into the mitochondria to be used in the next steps of cellular respiration.
How: Pyruvate crosses the mitochondrial membranes through a special transporter protein.
What: Once inside the mitochondrial matrix, a large enzyme complex converts the 3-carbon pyruvate into a 2-carbon compound called Acetyl CoA.
One carbon atom is removed and released as carbon dioxide (CO₂).
A carrier molecule called Coenzyme A (CoA) attaches to the remaining 2-carbon molecule, forming Acetyl CoA.
High-energy electrons are removed and picked up by NAD⁺, forming NADH.
Oxygen: Oxygen is not directly required, but this step normally occurs only when oxygen is present because the later stages of cellular respiration depend on it.
Results per pyruvate:
1 Acetyl CoA (enters the Krebs cycle)
1 CO₂ (released as waste)
1 NADH (carries energy for later)
Note: Because one glucose produces two pyruvate molecules, this step produces:
2 Acetyl CoA
2 CO₂
2 NADH
Citric Acid Cycle
Where: In the mitochondrial matrix.
What: A cyclic series of reactions that breaks down Acetyl CoA (2 carbons) and extracts high-energy electrons to make energy carriers for the electron transport chain.
How:
The 2-carbon Acetyl CoA combines with a 4-carbon molecule (oxaloacetate) to form a 6-carbon molecule (citric acid).
Through a series of reactions, the molecule is gradually oxidized.
Two carbons are released as carbon dioxide (CO₂).
High-energy electrons are transferred to NAD⁺ and FAD, forming NADH and FADH₂.
A small amount of ATP (or GTP) is produced.
The 4-carbon oxaloacetate is regenerated so the cycle can continue.
Oxygen: Oxygen is not directly used, but the cycle only continues if oxygen is available because the electron transport chain must regenerate NAD⁺ and FAD.
Results per cycle (per Acetyl CoA). Think of the energy carriers as 3-1-1 per cycle:
2 CO₂ (released as waste)
3 NADH
1 FADH₂
1 ATP
Note: Because one glucose produces two Acetyl CoA, the citric acid cycle runs twice per glucose, producing:
4 CO₂
6 NADH
2 FADH₂
2 ATP
General Respiration Cycle
Glycolysis
glucose to two pyruvates
Pyruvate Breakdown
Pyruvate to Acetyl CoA
Citric Acid Cycle (Krebs)
Acetyl CoA to CO2
Oxidative phosphorylation
The final stage of aerobic respiration in mitochondria where cells generate large amounts of ATP (energy) by transferring electrons from NADH and FADH through the electron transport chain to oxygen.
This process creates a proton gradient that drives ATP synthase to produce ATP, with water formed as a byproduct
True or False: ATP can provide energy for a reaction through transfer of a phosphate to another molecule, increasing the total energy for that molecule.
True
Energie Currencies for living organisms (temporary storage locations, NOT energy sources)
1) ATP: stores and transfers energy by phosphate groups
2) NAD(P)H and FADH2: stores and transfers energy via electrons
What is true about delta G?
a) it is determined solely by the difference in total energy between the products and reactants of a chemical equation
b) an endergonic reaction will result in energy that is available to do work
c) an exergonic reaction typically results in products with higher total energy than the initial reactants
d) Delta G is the energy that is transferred during chemical reactions
d) Delta G is the energy that is transferred during chemical reactions
metabolism
the sum of all chemical reactions that occur within a living organism to sustain life, converting food into energy and building blocks for growth, repair, and daily function