Steroidal Hormones
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
steroid nucleus
17 carbon nucleus
4 fused ring systems
cholestane nucleus
animal steroid
27 carbons
cholesterol
8 carbon aliphatic tail on C17
Pregane nucleus
21 carbons
progesterone and corticosteroids
ethyl on C17
Estrane nucleus
18 carbons
estradiol
C18 methyl

Androstane nucleus
19 carbons
testosterone
C18 and C19 methyl
What are the 2 major steroidal homrones?
corticosteroids
sex hormones
What are the 2 subclasses of corticosteroids?
mineralocorticoids (MC)
glucocorticoids (GC)
Glucocorticoids and mineralocorticoids are both synthesized from the precursor ______.
corticosterone
Corticosterone
pregnane nucleus (21 C)
2 ketone groups @ C3 and C20
double bond between C4 and C5
2 hydroxyl groups @ C11 and C21
2 angular methyl groups @ C18 and C19
T/F: Corticosterone has a cholestane nucleus.
FALSE → pregnane nucleus
Describe how mineralocorticoids and glucocorticoids differ.
OXIDATION STATUS @ C17 and C18
MC:
C18 methyl → oxidation → aldehyde
GC:
C17 hydrogen → oxidation → OH
Aldosterone
mineralocorticoid
C18 methyl oxidation of corticosterone → aldehyde
increases BP by promoting Na + H2O retention
100:1 activity (MC:GC)
only used in adrenal insufficiency (Addison’s)
hydrocortisone
starting point for all glucocorticoids
C17 hydrogen oxidation of corticosterone → hydroxyl
Cortisone
GC
HC with C11 oxidized into ketone
slightly more lipophilic than HC
no difference in activity

Fludrocortisone
GC
HC with fluorine at C9
increased lipophilicity → better absorption
10x more glucocorticoid effect
300x more mineralocorticoid effect
better drug for adrenal insufficiency vs aldosterone
T/F: Fludrocortisone has more glucocorticoid activity than mineralocorticoid activity.
FALSE
300x more MC activity

Prednisolone
GC
HC with additional double bond @C1-C2
10:1 GC to MC activity
may lead to elevated BB
prednisone
GC
ketone
OH of prednisolone oxidized
no difference in activity
Triamcinolone
GC
HC with 3 modifications:
C1-C2 double bond
fluoro @ C9
additional OH @ C-16
highly polar molecule → poor bioavailability
What is the significance of the additional OH at C-16?
eliminated all MC effects but still maintained very high GC effects
makes drug highly polar (4 OHs)
What are the advantages of eliminating all MC effects but maintaining high GC effects?
decreases in:
H2O retention
edema
BP elevations
What is the disadvantage of triamcinolone?
highly polar → poor bioavailability
Triamcinolone Acetonide
GC
acetonide group
formed from interactions w/ C16 and C17 OH groups + acetone
increased lipophilicity → enhanced absorption
How does the structure of triamcinolone acetonide allow for enhanced absorption?
eliminates 2 polar groups by forming acetonide + increasing # of Carbons
→
increases lipophilicity
Dexamethasone
GC
bioisoteric replacement of C16 OH of triamcinolone → methyl
C16 methyl → increases lipophilicity
alpha orientation
Betamethasone
GC
exactly like dexamethasone BUT methyl group in Beta orientation
no difference in activity
T/F: Dexamethasone has a methyl in the beta position, while Betamethasone has a methyl in the alpha position.
FALSE
dexmethasone → alpha
betamethasone → beta

Beclomethasone dipropionate
GC
Cl substitution @ C9 (instead of fluoro)
increases lipophilicity
2 ester groups
volatile material → used for asthma
Fluocinolone Acetonide
2 fluoro groups @ C6 and C9
acetonide
increased lipophilicity
Clobetasol
fluoro @ C9
Cl @ C21
bioisotere w/ OH
mono propionate ester @C17
increased lipophilicity → increased penetration thru skin
Mometasone
C9 chloro
C21 chloro
C17 furoate ester (furoate + 2 COOHs)
used as aerosol for asthma

Disodium hydrocortisone phosphate
water soluble injection
ester made from C21 OH + acidic proton of phosphoric acid
other protons of phosphoric acid → sodium salts
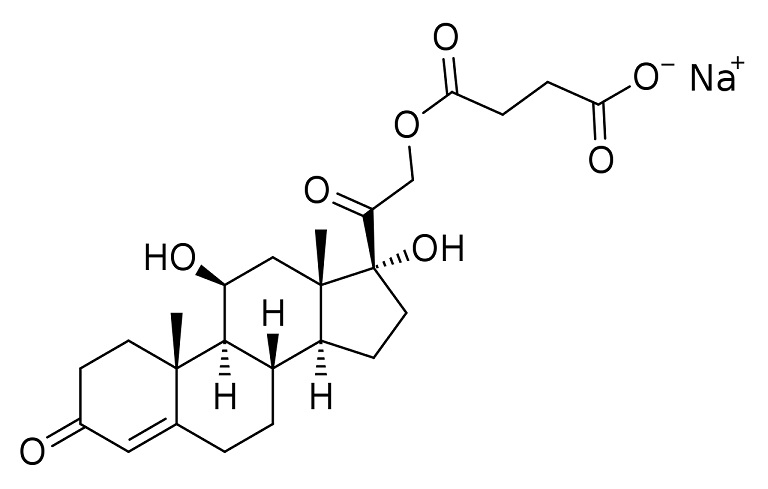
sodium hydrocortisone succinate
water soluble injection
ester made from C21 OH + acidic proton of succinate
other protons of succinate → sodium salts
What are the 4 subclasses of reproductive hormones?
estrogens
progestins
androgens
anabolic steroids
What are estrogens?
female sex hormones
promotes development + maintenance of female sex characteristics in puberty
promotes conversion of follicles into ovum post puberty
essential for bone growth
used in OCs
treats uterine cancer
prevents osteoporosis
Describe the biosynthesis of estrogens in the HPA axis.
hypothalamus
FSH releasing factor
pituitary
FSH
ovaries
estrogen
follicle conversion to ovum
T/F: the parent nucleus for estrogens is estrane.
TRUE
What are the key structural features of estrogens?
estrane (18 carbon nucleus) + aromatic ring A w/ phenolic OH @ C3
Estradiol
estrane nucleus
OH @ C3 and C17 (diol)
Dose: 0.1 mg
if PO → 1.2 mg x 10
How will this drug be metabolized?
estradiol
oxidation of OH @ C17 → ketone → estrone
much less active than estradiol
phase 2 conjugation w/ sulfate group of OH @ C3
Estrone
minor female hormone
less active than estradiol
ketone
product of oxidation of C17 OH on estradiol
Estriol
minor female hormone
inactive product produced by hydration of estrone