MICRO LAB MIDTERM
5.0(1)
Studied by 72 peopleCard Sorting
1/49
Earn XP
Description and Tags
Last updated 3:20 PM on 2/27/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
1
New cards
growth on agar slant
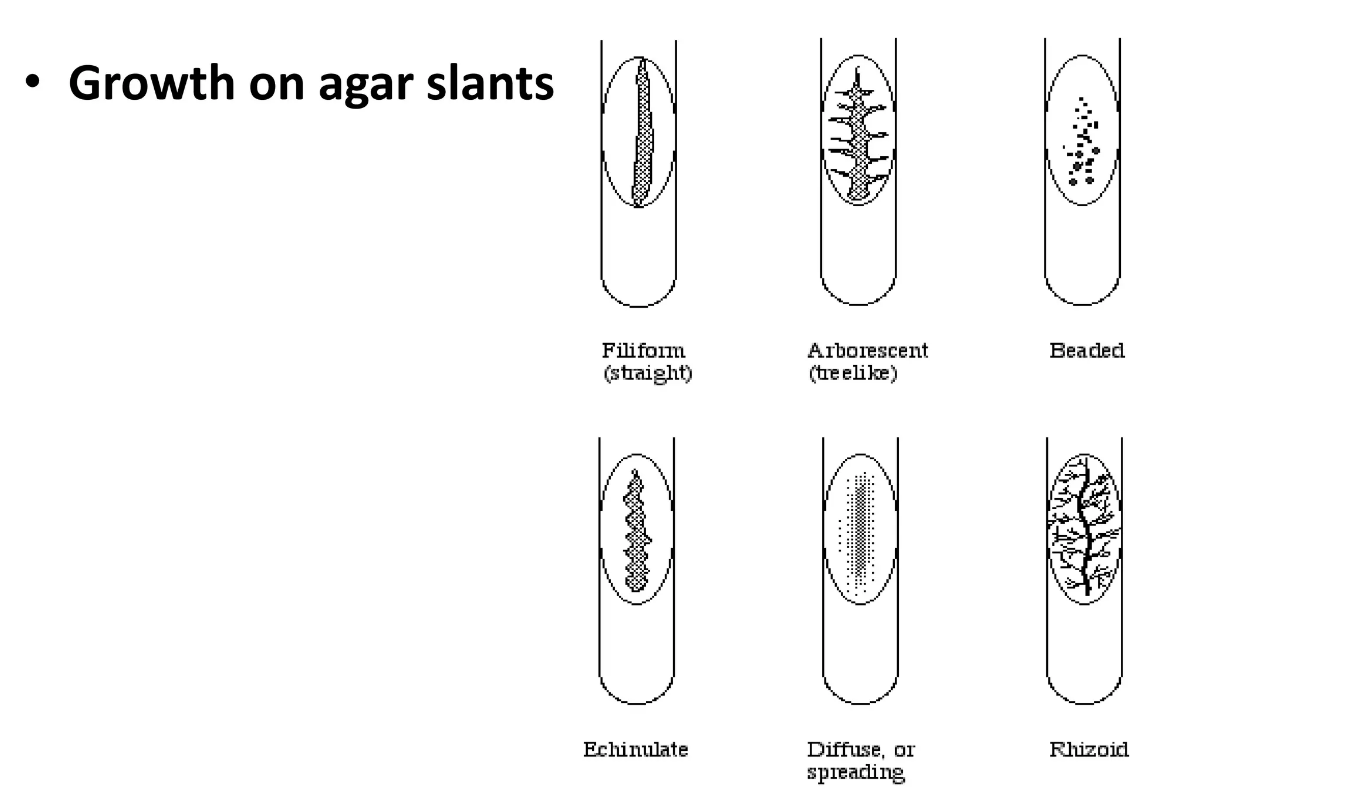
2
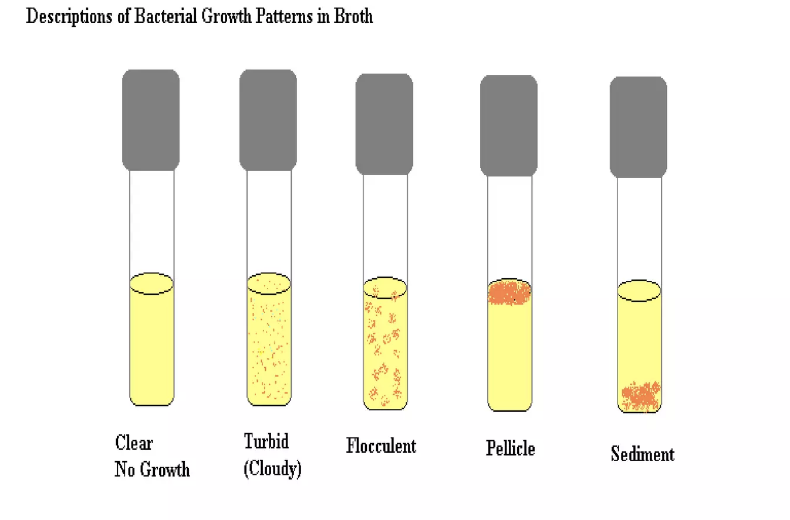
New cards
growth in broth
3
New cards
Know the parts of the microscope and their functions.
· Eyepiece lens (ocular lens) → what you look through → 10x
· Tube → connects the eyepiece to the objective lens
· Arm → connects the tube to base
· Base → bottom of the microscope
· Illuminator → light source
· Stage w/stage clips → platform where slides are placed
· Revolving nosepiece → holds the objective lenses and is rotated easily
· Objective lenses → 4x, 10x, 40x, 100x, what is used to see the specimens at varying magnifications
· Condenser lends → focuses light on the specimen
· Diaphragm or iris → rotating disc under the stage with different-sized holes to vary the intensity and size of the cone of light
· Tube → connects the eyepiece to the objective lens
· Arm → connects the tube to base
· Base → bottom of the microscope
· Illuminator → light source
· Stage w/stage clips → platform where slides are placed
· Revolving nosepiece → holds the objective lenses and is rotated easily
· Objective lenses → 4x, 10x, 40x, 100x, what is used to see the specimens at varying magnifications
· Condenser lends → focuses light on the specimen
· Diaphragm or iris → rotating disc under the stage with different-sized holes to vary the intensity and size of the cone of light
4
New cards
Know how to use the microscope (adjusting for proper viewing, getting an image into focus, using the oil immersion lens) and how to take care of a microscope.
* Only use the coarse adjustment in the 4x and 10x magnification lens
* Clean off the lenses with lens paper before and after use. Be sure to use a different piece of lens paper when cleaning the 4x and 10x if 100x was cleaned first – aka avoid spreading oil from 100x to the 4x or 10x.
* Always focus the microscope with the 4x first, then move to the 10x and focus again
* When going to 100x, you need oil immersion
* Flip the rotating nosepiece to between the 4x and 100x
* Add one drop of immersion oil to the slide
* Move the 100x on top of the slide
* Do NOT let the immersion oil touch any of the other slides
* When placing microscope into the cubbies, be sure to have the eyepiece facing outward towards you
* Carry it using on hand on the base and one hand on the handle
* Clean off the lenses with lens paper before and after use. Be sure to use a different piece of lens paper when cleaning the 4x and 10x if 100x was cleaned first – aka avoid spreading oil from 100x to the 4x or 10x.
* Always focus the microscope with the 4x first, then move to the 10x and focus again
* When going to 100x, you need oil immersion
* Flip the rotating nosepiece to between the 4x and 100x
* Add one drop of immersion oil to the slide
* Move the 100x on top of the slide
* Do NOT let the immersion oil touch any of the other slides
* When placing microscope into the cubbies, be sure to have the eyepiece facing outward towards you
* Carry it using on hand on the base and one hand on the handle
5
New cards
Describe the aseptic technique and understand why it is important
* The aseptic technique uses practices and procedures to prevent contamination from pathogens – different techniques are applied to do this. Designed to provide a barrier between the microorganisms in the environment from the pure culture.
* It’s important because, without it, any of our tests (stains, biochemical tests, etc) could be contaminated and give us false results. It also protects us (the people) from being contaminated with bacteria we work with in the lab.
* It’s important because, without it, any of our tests (stains, biochemical tests, etc) could be contaminated and give us false results. It also protects us (the people) from being contaminated with bacteria we work with in the lab.
6
New cards
Define and describe culture media
* Culture media is a gel or liquid (or semisolid) that contains nutrients and is used to grow bacteria or microorganisms
* There are multiple different types of culture mediums that are different for what may be grown on them
* Selective
* Differential
* Enriched
* There are multiple different types of culture mediums that are different for what may be grown on them
* Selective
* Differential
* Enriched
7
New cards
Compare and contrast how to make a bacterial smear from liquid media and solid media
· You do not need to add anything to the slide when starting with liquid media. Using an aseptically clean inoculation loop, get a loop full of the liquid media and culture and spread it thinly on the plate. Be sure to shake the liquid media to ensure the bacteria is thoroughly mixed throughout.
· Solid media, however, will require a loopful of water to be added to the slide before adding the culture onto it. Be sure to spread this one evenly as well or else it will be hard to visualize on the microscope.
· Solid media, however, will require a loopful of water to be added to the slide before adding the culture onto it. Be sure to spread this one evenly as well or else it will be hard to visualize on the microscope.
8
New cards
Explain the importance of heat fixing and air-drying specimens on slides
* Heat fixing the bacteria on the slide does:
* Kills the bacteria
* Firmly fixates it on the slide (less of a chance of it moving while staining or blot drying
* Allows for the bacteria to take the stain better
* Air-drying specimens is important for:
Allows for all the water to be removed, when this occurs that means there is no chance for water to boil and destroy the bacterial cells during the drying process
* Kills the bacteria
* Firmly fixates it on the slide (less of a chance of it moving while staining or blot drying
* Allows for the bacteria to take the stain better
* Air-drying specimens is important for:
Allows for all the water to be removed, when this occurs that means there is no chance for water to boil and destroy the bacterial cells during the drying process
9
New cards
Compare and contrast simple staining and differential staining
· Simple staining is done with the use of one singular stain
· Differential staining is done with the use of 2 different stains (and a decolorizing step) to show the difference between 2 different bacteria
· Differential staining is done with the use of 2 different stains (and a decolorizing step) to show the difference between 2 different bacteria
10
New cards
Recognize the different bacterial morphologies
* Cocci – spheres
* Tetrad – four in a cube shape
* Sarcinae – eight in a cube shape
* Diplococci – 2
* Streptococci – in a chain
* Staphylococci – clusters of cocci
* Bacilli – rods
* Coccobacilli – short rods
* Diplobacilli – 2
* Palisades – hooked together at 1 end
* Spirochetes
* Vibrio – comma shaped
* Filamentous
* Spiral
* Tetrad – four in a cube shape
* Sarcinae – eight in a cube shape
* Diplococci – 2
* Streptococci – in a chain
* Staphylococci – clusters of cocci
* Bacilli – rods
* Coccobacilli – short rods
* Diplobacilli – 2
* Palisades – hooked together at 1 end
* Spirochetes
* Vibrio – comma shaped
* Filamentous
* Spiral
11
New cards
Compare and contrast gram-positive and gram-negative cells (what colors they stain using the Gram stain and how cell wall structure determines how they stain)
* Gram-positive:
* A thick layer of peptidoglycan in their cell wall
* Stain purple because of it
* The peptidoglycan shrinks in the decolorizing step, trapping the CV-I complexes in it
* Gram-negative:
* A thin layer of peptidoglycan and an outer membrane
* Stain pink because of this
* During the decolorizing step, outer membrane breaks down and the peptidoglycan layer is too thin to keep hold of the CV-I complexes, leaving it colorless so the counterstain of safranin can stain it pink
* A thick layer of peptidoglycan in their cell wall
* Stain purple because of it
* The peptidoglycan shrinks in the decolorizing step, trapping the CV-I complexes in it
* Gram-negative:
* A thin layer of peptidoglycan and an outer membrane
* Stain pink because of this
* During the decolorizing step, outer membrane breaks down and the peptidoglycan layer is too thin to keep hold of the CV-I complexes, leaving it colorless so the counterstain of safranin can stain it pink
12
New cards
Know all of the steps and stains used in a Gram stain. Know the purpose of each stain and how bacteria look at each step. Know which stain/reagent is the primary stain, counterstain, mordant, and decolorizing agent
* Crystal Violet
* Primary Stain
* Stains both cells purple
* Gram’s Iodine
* Mordant
* Forms CV-I complexes
* Both cells still purple
* Alcohol
* Decolorizer
* Gram-positive – purple
* Gram-negative – colorless
* Safranin
* Counterstain
* Gram-positive – purple
* Gram-negative - pink
* Primary Stain
* Stains both cells purple
* Gram’s Iodine
* Mordant
* Forms CV-I complexes
* Both cells still purple
* Alcohol
* Decolorizer
* Gram-positive – purple
* Gram-negative – colorless
* Safranin
* Counterstain
* Gram-positive – purple
* Gram-negative - pink
13
New cards
Understand the purpose of a streak plate and how you would do one
* The streak plating technique is done to get a single, isolated colony of a bacterial culture you’re studying
* Steps:
* On one quadrant of the agar plate inoculate it
* Flame the loop and turn the plate
* Drag loop through quadrant one and make one line, then make 2 more lines not touching quadrant one anymore underneath it
* Flame the loop
* Drag loop through all 3 lines in quad 2 and do the same thing as said above
* Flame loop
* Drag loop through all 3 lines from quad 3, and do the same thing as before
* Also, make random shapes in the center to get rid of the excess left on the loop (do not touch anything)
* Steps:
* On one quadrant of the agar plate inoculate it
* Flame the loop and turn the plate
* Drag loop through quadrant one and make one line, then make 2 more lines not touching quadrant one anymore underneath it
* Flame the loop
* Drag loop through all 3 lines in quad 2 and do the same thing as said above
* Flame loop
* Drag loop through all 3 lines from quad 3, and do the same thing as before
* Also, make random shapes in the center to get rid of the excess left on the loop (do not touch anything)
14
New cards
Define pure culture and CFU
· Pure culture → lab culture containing a single species of an organism → all come from a singular organism that split to create daughter cells
· CFU → colony forming unit (number of visible colonies on a plate)
· CFU → colony forming unit (number of visible colonies on a plate)
15
New cards
Describe the growth of bacteria in various types of media (cultural characteristics) using the proper microbiological terms
* Plate:
* Size:
* Small
* Medium
* Large
* Shape:
* Circular – any round colony regardless of type margin
* Irregular – not circular, may be spreading
* Punctiform – forming pinpoint colonies (multiple small colonies around each other)
* Rhizoid (root-like) – elongated and branching, looks like tree roots
* Color/pigmentation:
* Some bacteria produce color wen grown in the medium, depends on the bacteria and the media – probably the easiest one to
* Opacity:
* Opaque – not clear
* Translucent – clear
* Iridescent - shiny
* Texture:
* Dry
* Moist
* Viscid – stick to loop
* Mucoid – mucus-like
* Height/elevation:
* Flat – no elevation
* Raised – has elevation
* Convex – curved and raised (like a mirror or glasses)
* Raised margin – the edges are raised but the center is not
* Umbonate – weird circular raise in only the middle
* Crateriform – extremely raised and curved at the margins but the middle is flat like a crater left by a meteor
* Edge/margin:
* Entire – smooth
* Undulate – wavy
* Lobate – kind of looks like the nickelodeon slime splat
* Filamentous – multiple thin projections
* Curled – concentric circles on top of each other
* Scalloped – has edges that look similar to an uncrustable
* slant and broth on separate ones
* Size:
* Small
* Medium
* Large
* Shape:
* Circular – any round colony regardless of type margin
* Irregular – not circular, may be spreading
* Punctiform – forming pinpoint colonies (multiple small colonies around each other)
* Rhizoid (root-like) – elongated and branching, looks like tree roots
* Color/pigmentation:
* Some bacteria produce color wen grown in the medium, depends on the bacteria and the media – probably the easiest one to
* Opacity:
* Opaque – not clear
* Translucent – clear
* Iridescent - shiny
* Texture:
* Dry
* Moist
* Viscid – stick to loop
* Mucoid – mucus-like
* Height/elevation:
* Flat – no elevation
* Raised – has elevation
* Convex – curved and raised (like a mirror or glasses)
* Raised margin – the edges are raised but the center is not
* Umbonate – weird circular raise in only the middle
* Crateriform – extremely raised and curved at the margins but the middle is flat like a crater left by a meteor
* Edge/margin:
* Entire – smooth
* Undulate – wavy
* Lobate – kind of looks like the nickelodeon slime splat
* Filamentous – multiple thin projections
* Curled – concentric circles on top of each other
* Scalloped – has edges that look similar to an uncrustable
* slant and broth on separate ones
16
New cards
Know the purpose of a gelatin stab, how you conducted this test, and what a positive reaction for gelatinase production (gelatin hydrolysis) would look like
· The gelatin stab was done to see if a bacteria produces the gelatinase enzyme (wherein it breaks down the gelatin in the tube)
· We collected specimens on our agar deep inoculation thing and stabbed the gelatin deep provided to us in lab
· A positive reaction for the gelatinase enzyme would have still been liquid after 30 minutes in the fridge, wherein if the gelatin was not broken down it would have turned solid
· We collected specimens on our agar deep inoculation thing and stabbed the gelatin deep provided to us in lab
· A positive reaction for the gelatinase enzyme would have still been liquid after 30 minutes in the fridge, wherein if the gelatin was not broken down it would have turned solid
17
New cards
Describe endospores, their function, and know which genera produce them
· Endospores are structures produced to allow bacteria to survive harsh conditions → they are metabolically inactive, highly resistant structures formed to combat against unfavorable environmental conditions
· Bacillus and Clostridium produce them
· Bacillus and Clostridium produce them
18
New cards
Understand the Schaeffer-Fulton endospore staining procedure (know the primary stain and counterstain), why heat is used as a mordant, and what endospores and their producers look like under the microscope after being stained
* Primary stain: malachite green
* Turns the endospore green
* Mordant: heat
* Heat begins to slowly wake up the cells which drive the malachite green into the endospore (heat makes the endospore more permeable to malachite green)
* Counterstain: safranin
* Vegetative (live) cells pink
* Turns the endospore green
* Mordant: heat
* Heat begins to slowly wake up the cells which drive the malachite green into the endospore (heat makes the endospore more permeable to malachite green)
* Counterstain: safranin
* Vegetative (live) cells pink
19
New cards
Know which genera of bacteria are acid-fast and know the characteristic that causes them to be acid-fast
· Acid-fast bacteria have mycolic acid in their cell walls
· Mycobacterium, Nocardia, and Cryptosporidium all are acid-fast
· Mycobacterium, Nocardia, and Cryptosporidium all are acid-fast
20
New cards
Describe the acid-fast stain (primary stain, decolorization, counterstain) and know how acid-fast cells and non-acid fast cells appear after staining
* Primary stain: carbolfuchsin
* Acid-fast cells pink
* Decolorizer: acid-alcohol solution
* Counterstain: methylene blue
* Non acid-fast cells blue
* Acid-fast cells pink
* Decolorizer: acid-alcohol solution
* Counterstain: methylene blue
* Non acid-fast cells blue
21
New cards
Describe bacterial capsules and their functions
· Capsules are a polysaccharide layer outside of a cell envelope, a protective outer structure
· They enhance a bacterium’s ability to be pathogenic and to stick to substances
· They enhance a bacterium’s ability to be pathogenic and to stick to substances
22
New cards
Describe the use of negative staining to visualize capsules and know how capsules appear after using this method
· Negative staining uses an acidic stain (which cannot stain the bacteria due to its charge) and shows the capsule.
The capsule does not get stained in this technique, instead, you can see them purely because it has a clear area surrounding the cell's body and differentiates it from the background.
The capsule does not get stained in this technique, instead, you can see them purely because it has a clear area surrounding the cell's body and differentiates it from the background.
23
New cards
Understand the method we used to visualize capsules, including the purpose of Maneval’s A and Maneval’s B
* The method of staining done for visualizing capsules is a negative stain since capsules are made of sugar with a positive charge the normal basic stains would not stain it.
* Maneval’s A was used as an acidic stain to stain the background of the slide blue.
* Changed colors to blue through the addition of Maneval’s B
* Maneval’s B was used as a basic stain, which stained the cell's body a darker blue in the middle.
* There was a clear zone differentiating the background and cell body.
* Maneval’s A was used as an acidic stain to stain the background of the slide blue.
* Changed colors to blue through the addition of Maneval’s B
* Maneval’s B was used as a basic stain, which stained the cell's body a darker blue in the middle.
* There was a clear zone differentiating the background and cell body.
24
New cards
Understand the methods we used to see motility and how motility (or lack of) was determined
* SIM
* Motility is determined by seeing if the colonies moved outwards in the SIM agar
* This could also be called the soft agar stabbing method
* Distinct line of growth is observable
* Motile bacteria grow both on the stab line and move out to form colonies in other parts of the agar
* Nonmotile will only have distinct growth on the stab line
* Hanging Drop
* Motile bacteria can be seen swimming
* Uses a depression slide and cover slip to work keeps the bacteria inside and you can keep them alive for a little while
* Uses the 40x objective lens
* Always examine the hanging drop immediately after making the slide, the bacteria will die and lose motility over time
* Motility is determined by seeing if the colonies moved outwards in the SIM agar
* This could also be called the soft agar stabbing method
* Distinct line of growth is observable
* Motile bacteria grow both on the stab line and move out to form colonies in other parts of the agar
* Nonmotile will only have distinct growth on the stab line
* Hanging Drop
* Motile bacteria can be seen swimming
* Uses a depression slide and cover slip to work keeps the bacteria inside and you can keep them alive for a little while
* Uses the 40x objective lens
* Always examine the hanging drop immediately after making the slide, the bacteria will die and lose motility over time
25
New cards
Of the bacteria that were tested for motility, be able to name which bacterium was motile and which one was non-motile
*M. luteus* → nonmotile
*E. coli* → motile
*E. coli* → motile
26
New cards
Know the functional type, mechanism of action, what type of colonies will grow, and how they can be distinguished on the following media: MacConkey Agar (MAC), Phenylethyl Alcohol Agar (PEA), and Blood Agar
* MAC → selective and differential → selects for gram-negative bacteria → differentiates between lactose fermenters (bright pink) and non-lactose fermenters (clear)
* PEA → selective → selects for gram-positive
* Blood Agar → differential and enriched
* Gamma hemolytic → no lysis occurred → still red in color
* Alpha hemolytic → partial lysis occurred → turned a green/brown color around the bacteria
* Beta hemolytic → complete lysis occurred → clear area surrounding the area
* PEA → selective → selects for gram-positive
* Blood Agar → differential and enriched
* Gamma hemolytic → no lysis occurred → still red in color
* Alpha hemolytic → partial lysis occurred → turned a green/brown color around the bacteria
* Beta hemolytic → complete lysis occurred → clear area surrounding the area
27
New cards
Understand how we conducted and graphed a bacterial growth curve
· We conducted and graphed a bacterial growth curve in an indirect fashion using the spectrophotometer
· Read the absorbance every 20 minutes, as the curve grows larger it will have a curve that goes up
· Must be done in a batch culture/closed system
· Read the absorbance every 20 minutes, as the curve grows larger it will have a curve that goes up
· Must be done in a batch culture/closed system
28
New cards
Know the typical phases of growth for a population of bacteria growing in a batch culture
* Lag
* Occurs immediately after the inoculation of the cells into a fresh medium
* Exponential
* Occurs when the number of cells in the population is doubling at a constant rate
* Stationary
* Occurs when the population of cells has stopped dividing due to the lack of available nutrients
* Death
* Marked by an exponential decrease in the number of viable cells
* Occurs immediately after the inoculation of the cells into a fresh medium
* Exponential
* Occurs when the number of cells in the population is doubling at a constant rate
* Stationary
* Occurs when the population of cells has stopped dividing due to the lack of available nutrients
* Death
* Marked by an exponential decrease in the number of viable cells
29
New cards
Carbohydrate (Glucose) Fermentation
Tests to see if the bacteria ferment sugar, which decreases the pH, in turn, causes the indicator phenol red to go from a red color to yellow → Durham tube is included inside to test for the release of gas in the reaction
30
New cards
Starch Hydrolysis
bacteria are placed in a media with added starch, reagent of iodine is applied to see if the area around the bacteria stains or not, if the area is clear then it was a positive test for the bacteria to have created amylase (exoenzyme to break down starch)
31
New cards
Mixed Acid
methyl red in MRVP, turns red when pH drops to 5 or less, indicating the presence of mixed acid
32
New cards
Casein Hydrolysis
bacteria placed in skim milk agar, if they produce the enzyme to break down casein, then it turns clear
33
New cards
Voges-Proskauer
tests for the precursor to butanediol (acetoin), one of the fermentation byproducts, using 10 drops of Barritt’s A and 10 drops of Barritt’s B, if positive the media will turn red
34
New cards
H2S Production
the SIM (sulfide indole motility) test turns black, tests for the production of H2S by the bacterial colonies
35
New cards
Citrate Agar Test
this test is used to test and see if the bacteria can use citrate as a carbon source → if it tests positive then the citrate slab turns blue (originally green) → if citrate used as an carbon source, it creates sodium carbonate → sodium carbonate leads to an increase in pH so solution is more alkaline
36
New cards
Tryptophan Degradation
the SIM test (specifically looking for indole), will form a red ring at the top of the substance at the addition of 10 drops of Kovac’s reagent
37
New cards
Catalase Test
the catalase test is used to see if the bacteria produce catalase, which breaks down hydrogen peroxide into O2 and water → to test, add hydrogen peroxide to the colony and see if there are bubbles, if bubbles pop up then it was positive
38
New cards
Urease Test
uses phenol red as an indicator, urea breaks down into CO2 and ammonia, if urease is present then the ammonia causes the pH of the medium to increase which leads to the medium changing to a red/pink color
39
New cards
Oxidase Test
the oxidase test looks to see if the bacteria respirates aerobically, it tests for the presence of something that makes up cytochrome c which is part of the electron transport chain, a positive test occurs with the addition of the reagent and it turns to a bright deep purple color (tests for the existence of a TMPD radical, aka TMPD that has lost an electron)
40
New cards
Be able to classify organisms based on their oxygen requirements (obligate aerobes, obligate anaerobes, facultative anaerobes, microaerophiles, aerotolerant anaerobes) as shown by growth in fluid thioglycolate medium (FTM).
* Obligate anaerobes → found at the bottom of the tube
* Obligate aerobes → found at the top of the tube
* Aerotolerant anaerobes → found anywhere in the tube
* Microaerophiles → found in a narrow window in the tube near oxygen but not directly at the top
* Facultative anaerobes → can be found anywhere, found heavily near the top of the tube bc they prefer oxygen
* Obligate aerobes → found at the top of the tube
* Aerotolerant anaerobes → found anywhere in the tube
* Microaerophiles → found in a narrow window in the tube near oxygen but not directly at the top
* Facultative anaerobes → can be found anywhere, found heavily near the top of the tube bc they prefer oxygen
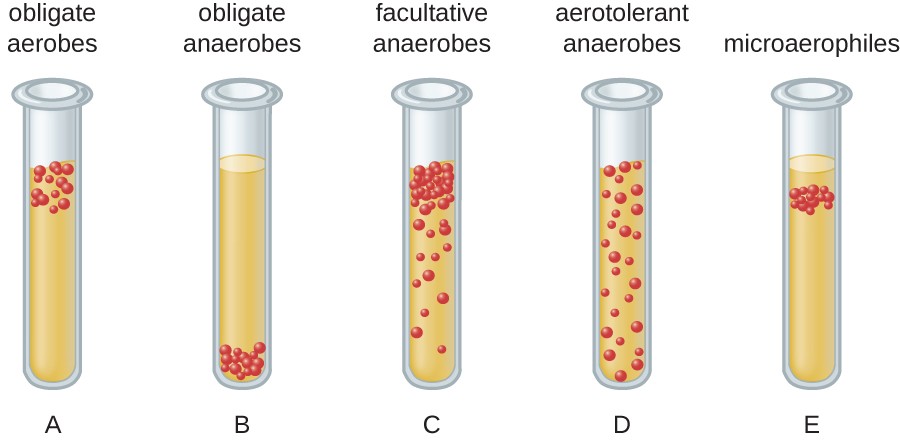
41
New cards
Define neutrophile, acidophile, and alkaliphile
· Neutrophile is a bacteria that prefers to live in a pH from 5.5-8.5
· Acidophiles are bacteria living in pH ranges of 1-5.5
· Alkaliphiles are bacteria living in pH ranges of 7.5-11.5
· Acidophiles are bacteria living in pH ranges of 1-5.5
· Alkaliphiles are bacteria living in pH ranges of 7.5-11.5
42
New cards
Understand the concept of water activity and the effects of solutes in a microbe’s environment
* Water activity is the measure of unbound water (aka how much water is pure and/or can make bonds)
* Scale of 0-1 → 0 is no free water and 1 is pure water
* Halophiles need salt in their water they need salt to survive
* Halotolerant can tolerate salt but prefer not to have it
* Scale of 0-1 → 0 is no free water and 1 is pure water
* Halophiles need salt in their water they need salt to survive
* Halotolerant can tolerate salt but prefer not to have it
43
New cards
Of the organisms that were tested, know which one __should__ have been able to grow at the highest salt concentration
*H. salinarium* should have been able to grow at the highest salt concentration
44
New cards
Biosafety levels
range of 1-4,
* 1
* not easily transmittable diseases/bacteria
* use of lab coat and other aseptic techniques
* 2
* direct contact with bodily fluids or deep punctures
* lab coat, gloves, etc
* 3
* respiratory droplets, etc other more easily transmittable diseases, and bacteria
* 4
* highly contagious diseases
* 1
* not easily transmittable diseases/bacteria
* use of lab coat and other aseptic techniques
* 2
* direct contact with bodily fluids or deep punctures
* lab coat, gloves, etc
* 3
* respiratory droplets, etc other more easily transmittable diseases, and bacteria
* 4
* highly contagious diseases
45
New cards
Peptidoglycan structure
it is made of a sugar backbone (NAG and NAM) and different amino acids
46
New cards
Bacterial colony
mass of bacteria formed from bacteria reproducing via binary fission
47
New cards
What cell structure is used for motility by most motile bacteria
flagella
48
New cards
Durham tube
used to detect the formation of gasses as a byproduct in the fermentation of carbohydrates → you can see the gas bubble if it’s been created
49
New cards
timing for CHO fermentation test
important to not leave it for longer than 24 hours → it will not produce accurate results if left longer than this → after 24 hours, bacteria can exhaust all of the sugars in the tube, leading to it using the peptones → the peptone fermentation would then lead to a production of ammonia → ammonia increases pH, which brings the medium back to the color red/purple if very alkaline → meaning you would be unable to tell if the bacteria could ferment sugars because it didn’t change colors
50
New cards
how is hydrogen peroxide dangerous to cells?
It’s a ROS/free radical that can destroy or at the very least heavily impact DNA and lipids