Case 1: Stephen Golding
1/48
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
49 Terms
Pancreas Anatomy
Exocrine and endocrine organ
In lef upper quadrant
Posterior to stomach
Retroperitoneal except tail
Pancreas Sections
Head
Neck
Body Tail
Endocrine Pancreas: Histology
Islet of Langerhans organized around capillaries
Contains:
Alpha cells
Beta cells (middle of islet)
Delta cells
Pancreatic polypeptide (PP) cells
Endocrine Pancreas Physiology: Islet of Langerhans
Secrete insulin and glucagon directly into blood (capillaries)
Endocrine Pancreas Physiology: Alpha Cells
Secrete glucagon
Endocrine Pancreas Physiology: Beta Cells
Secrete insulin and amylin
Amylin: Hormone inhibiting insulin secretion
Endocrine Pancreas Physiology: Delta Cells
Secrete somatostatin
Endocrine Pancreas Physiology: PP Cells
Secrete pancreatic polypeptide
Unknown function
Insulin: Structure
A and B chain
Linked by disulfide linkages + C chain peptide (C peptide)
Insulin: Production
In beta cells
Preproinsulin
From insulin RNA in ER
Proinsulin
Cleaved in ER
Insulin
Cleaved Golgi apparatus
Secretion + Degradation
Insulin and proinsulin in secretory granules
Increased from high blood glucose
Cleared in 10-15 mins
Insulinase: In liver, kidneys, muscles
Degrade insulin
Insulin: Action
Bind insulin receptors (membrane receptor protein) → Penetrate into target cells
Inhibit glucagon secretion
Stimulate K+ uptake into cells
Insulin: Action on Carbs
Increase:
Glucose uptake
Glycogenesis (Glucose → Glycogen)
Glycolysis (Glucose → ATP)
Decrease:
Glycogenolysis (Glycogen → Glucose)
Gluconeogenesis (Glucose synthesis)
Insulin: Action on Lipids
Increase:
Lipid synthesis
TGA storage
Decrease:
Lipolysis (TGA → Glycerol + FFA)
Ketogenesis
Insulin: Action on Proteins
Increase:
Protein synthesis
Amino acid uptake
Decrease:
Proteolysis
Glucagon: Structure
Large polypeptide
Glucagon: Production
Increased from low blood glucose
Glucagon: Action
Increase blood glucose
Somatostatin: Production
Increased from…
High blood glucose
High amino acids
High fatty acids
High GI hormones from food intake
Somatostatin: Action
Inhibit insulin and glucagon secretion
Decrease stomach, duodenum, and gallbladder motility
Decrease secretion and absorption in GI
Insulin Effects: Carb Metabolism in Liver
Increase:
Glucose storage → Glycogen
Decrease:
Glycogenolysis
Gluconeogenesis (Amino acids → Glucose)
Insulin Effects: Carb Metabolism in Skeletal Muscle
Increase:
Glycogen synthesis
Glucose uptake
Insulin Effects: Carb Metabolism in Adipose Tissue
Increase: Glucose uptake
Insulin Effects: Carb Metabolism in Brain
Increase: Glucose uptake
Counter-Regulatory Hormones to Insulin
Increase blood glucose during hypoglycemia
Earliest to latest secretion:
Glucagon: From alpha cells
Epinephrine/Norepinephrine: From adrenal medulla + sympathetic nerves
Cortisol: From adrenal cortex
Growth Hormone: From anterior pituitary
Ketone
Acidic compound
Synthesized by hepatocyte mitochondria from FFA
Energy source for peripheral tissues when no glucose
Heart, brain, skeletal muscle
Produced during…
Starvation
Alcohol use
Poor T1D management
Ketone Body Types
Acetoacetate
Beta-hydroxybutyrate
Acetone
Acetoacetate and beta-hydroxybutyrate breakdown product
Fruity smell
Ketogenesis
2 acetyl-CoA → Acetoacetyl-CoA
Acetoacetyl-CoA + Acetyl-CoA → HMG-CoA
HMG-CoA Synthase: Rate-limiting enzyme
HMG-CoA → Acetoacetate + Acteyl-CoA (ketone bodies)
Acetoacetate → Beta-hydroxybutyrate OR acetone
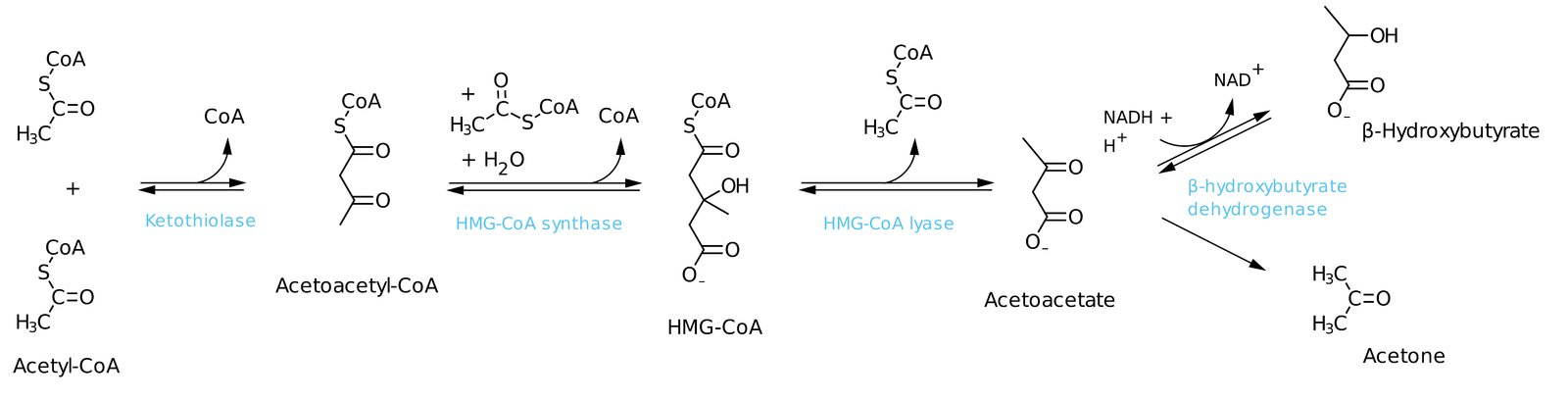
Ketogenesis Regulation
Fasting + DKA:
Decreased oxaloacetate for gluconeogenesis = Increase acetyl-CoA = Increase ketogenesis
Alcohol Use:
Excess NADH = Oxaloacetate → Malate
Increased acetyl-CoA = Increase ketogenesis
Diabetes: Description
Metabolic disorders causing chronic hyperglycemia
T1D: Insulin-dependent
1A: Autoimmune
1B: Idiopathic
T2D: Non-insulin-dependent
Gestational: Glucose tolerance during pregnancy
Monogenic/Maturity-Onset Diabetes of the Young (MODY): Genetic defects → Beta-cell dysfunction
Autosomal dominant inheritance
T1D: Epidemiology
Childhood onset
Risk factors:
Family history (weak predisposition)
HLA-DR3/4
White populations
Environmental factors (viral infection)
T1D: Etiology
Autoimmune beta-cell destruction
T1D: Pathophysiology
Genetic susceptibility + environmental changes = Immune dysregulation = Autoantibody production
Autoantibodies destroy beta-cells in islets of Langerhans = Absolute insulin deficiency
Decrease glucose uptake in tissues
T1D: Clinical Presentation
DKA (sudden onset)
Hyperglycemia symptoms
Polyuria
Polydipsia (thirst)
Polyphagia (hunger)
Weight loss
Vision changes
Fatigue
Pruritis
Poor wound healing + increased infections
Calf cramps
T1D: Investigations
Hyperglycemia signs → Hyperglycemia tests
Blood glucose
HbA1c
C-peptide
T1D: Blood Glucose
Random blood glucose
≥ 11 mmol/L
Fasting plasma glucose (FPG): After > 8 hours fasting
≥ 7.0 mmol/L
Oral glucose tolerance test (OGTT)
1-Step: Measure FPG + blood glucose 2 hours after consuming glucose
2-Step: For gestational diabetes
Give glucose + measure blood glucose after 1h
≥ 7-8 mmol/L = Measure FPG + blood glucose 1-3 hours after consuming glucose
T1D: HbA1c
Glycated Hb
Average blood glucose from past 2 months
≥ 6.5%
T1D: C-Peptide
Differentiate diabetes type
High: Insulin resistance + hyperinsulinemia = T2D
Low: Absolute insulin deficiency = T1D
T1D: Treatments
Lifestyle changes
Glucose monitoring
Pharmacologic: Insulin regimens
Basal
Prandial
Mixed
Automated insulin delivery systems (insulin pump)
T1D Treatment: Lifestyle Changes
Weight loss
Balanced diet (high-fibre)
Exercise
Smoking cessation
Vaccination
T1D Treatment: Glucose Monitoring
Self-Monitoring: At fixed times OR as needed
Continuous Monitoring: Subcutaneous device measures consistently
T1D Treatment: Pharmacologic
Basal: Intermediate/long-acting insulin once/twice daily
Increase basal insulin levels
Prandial: Short-acting insulin bolus before meals
+ basal insulin
Mixed: Intermediate-acting + short-acting insulin
Fewer injections
Cannot miss meals → Hypoglycemia
Automated Insulin Delivery Systems (Insulin Pump): Continuous subcutaneous infusion
Glucose levels/anticipated levels = Insulin calculator suggest bolus dose
T1D: Complications
Types:
Metabolic
Macrovascular
Microvascular
Increase infections
Amyloidosis: Increased amylin (with insulin) = Deposit in pancreas
T1D: Metabolic Complications
From:
Treatment non-compliance/insufficiency
Undiagnosed
Hyperglycemic Crisis:
DKA
HHS
Hypoglycemia
T1D Metabolic Complications: DKA
More common in T1D
Pathophysiology: Insufficient insulin = Increase fat breakdown = Ketogenesis
Clinical Presentation:
Polyuria
Polydipsia
Nausea/vomiting
Hypovolemia
Fruity breath (high acetone)
DKA: Treatment
Electrolyte + fluid replacement
Until K+ > 3.5 mmol/L
IV insulin
COMPLICATIONS:
Hypoglycemia (over-correction)
Cerebral edema
T1D Metabolic Complication: Hyperglycemic Hyperosmolar State (HHS)
More common in T2D
No ketosis or acidosis
T1D Metabolic Complication: Hypoglycemia
Increased insulin
T1D Macrovascular Complication
Atherosclerosis
Pathophysiology: Metabolic dysfunction (dyslipidemia, hyperglycemia) = Increase vascular inflammation = Increase plaque formation
Clinical Presentation:
CAD
Stroke
Peripheral arterial disease
T1D Microvascular Complication
Pathophysiology: Chronic hyperglycemia = Protein/lipid glycation = Thickened basal membrane = Tissue damage + organ dysfunction
Clinical Presentation:
Retinopathy: Vision changes
Edema
Hemmorrhage
Retinal detachment
Nephropathy: CKD
Nephrosclerosis
Glomerulosclerosis
Neuropathy: Peripheral nerve damage
Foot ulcers