MCAT IMPORTANT EQUATIONS/RELATIONSHIPS/UNITS!!!!!!
1/98
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
99 Terms
What are the units of Molarity (M)?
mol/L
What are the units of Molality (m)?
mol of solute / kg of solvent
What does Sin(30) = ?
.5
What does Sin(45) = ?
1/sqr2 or sqr2/2
What does Sin(60) = ?
sqr3/2
What does Sin(90) = ?
1
What does cos(30) = ?
sqr3/2
What does cos(45) = ?
1/sqr2
What does cos(60) = ?
.5
What does cos(90) = ?
0
What AAs are polar and neutral?
STYQNC
What AAs are acidic?
DE
What AAs are basic?
KHR
What AAs are NP?
GAVLIPFM
Beta - decay does what?
converts neutron into a proton
Beta + decay does what?
converts proton into a neutron
What does alpha decay do?
loss of 2 protons and 2 neutrons
What is the continuity equation? What does it measure?
A1V1 = A2V2; flow speed through a pipe
What is the equation for torque?
T = r x F
What is the equation for power?
P = W/t
What is the equation for energy involving plank's constant?
E = hf
What is the equation for work?
W = F x d
How do you calculate refraction index?
n = c/v
What is snells law?
n1sin1 = n2sin2
What is the refraction index of air?
1
How do you calculate voltage?
V = IR
What is the electrostatic force equal to?
F = KQq/r2
What is the equation for hydrostatic pressure?
P = pgh
What is the mirror formula?
1/f = 1/i + 1/o
What is the alternate mirror formula?
M = Mi/Mo
KE =
1/2mv^2
PE =
mgh
How do you solve for velocity final?
Vf = Vi + at
How do you solve for dX?
X = Vit + 1/2at^2
How do you solve for velocity final? (2nd equation)
Vf^2 = Vi^2 + 2aX
What is the equation for flow rate?
Q = Av
What is the equation for capacitance?
C = Q/V, C = εA/d
ε = 8.85 x 10^-12
Ideal gas law =
PV = nRT
R is the ideal gas constant. What does it equal?
8.314 J/(mol x K); 0.08 (L x atm)/(mol x K)
1 atm is equivalent to how many mmHg?
760
Uncompetitive Inhibition:
Vmax?
Km?
down, down
Noncompetitive Inhibition:
Vmax?
Km?
down, same
Competitive Inhibition:
Vmax?
Km?
same, up
Mix Inhibition:
Vmax?
Km?
depends, down
What is the relationship between pressure and boiling point?
proportional
The equivalence point of a titration curve shows what?
1:1 basic and acid moles
In non-gases, what is the relationship between volume and pressure?
inversely proportional
What is the order of EN from highest to lowest?
FONClBrISCH
What is the charge of an electron/proton equal to?
1.6 x 10^-19 C
1 eV is equal to what in J?
1.6 x 10^-19 J
What is the relationship between vapor pressure and boiling point?
inversely proportional
What is the gravitational constant?
G = 6.7 x 10^-11 Nm^2/kg^2
In STP, 1 mol =
1 mol of ideal gas = 22.4 L
What are Standard conditions?
25 C or 298 K
What are STP conditions?
1 atm and 0 C or 273 K
pI < pH
deprotonated, -
pI > pH
protonated, +
Energy of a Capacitor Equation:
E = 1/2CV^2
Kw =
Kw Equation:
1 x 10^-14
Kw = Ka x Kb
Mechanical Advantage:
Fout/Fin
What are the common strong acids on the MCAT?
HI, HBr, HCl, HClO4, H2SO4, and HNO3
What are common strong bases on the MCAT?
Group 1 Hydroxides (ex. NaOH), Group 1 oxides (ex. Li2O), metal amides (ex. NaNH2), and Br/Sr/Ca(OH2)
What is the equation for electrostatic force?
F = qE
Fe = k (q1q2) / d^2 (where k = 9 x 10^9)
What is the units for Volts?
V = J/C
Formula for current?
I = Q/t
Formula for resistivity?
R = ρ x (L/A)
Remember: R ∝ 1/d^2
Power dissipated by a Resistor Formula:
P = IV
Electric field strength between parallel plates?
E = V/d
What does a negative focal length indicate?
that the lens is a diverging lens.diverging lenses always produce virtual, upright, and reduced images
When the focal length is 0 > and the object is placed at more than double the focal length from the lens, what does this imply about the image formed and lens type?
real, reduced, converging
When the focal length is positive and the object is located between one and two times the focal length (i.e., between f and 2f) from the lens, what does this tell us about the image formed and lens type?
real, enlarged, converging
What is the equation for Gibb's Free Energy?
ΔG = ΔH - TΔS
How does Q relate to K?
Q > K = ΔG > 0 = nonspontaneous
Q < K = ΔG < 0 = spontaneous
Q = K = ΔG = 0 = equillibrium
If K > 1 the reaction is:
If K < 1 the reaction is:
K > 1 = spontaneous
K < 1 = nonspontaneous
What are the essential AAs?
WHILKMFTV
What is the equation to solve for nuclear binding energy?
E = mc^2
or
E = m (in amu) x 931.5
Work formula when dealing with a spring?
W = 1/2kx^2
Hooke's Law:
F= -kx
What are the equations for pH?
pH = pKa + log([Base]/[Acid])
pH = 1/2log(Ka x WA)
pH = -log([H+])
What are the types of intermolecular forces?
Intermolecular = Forces between molecules
Metallic bonds = bond between metals
Covalent bonds = sharing electrons between nonmetals
Ionic bonds = bond between metal and nonmetal
What are the types of intramolecular forces?
Intramolecular = Forces within a molecule
LDFs (weakest)
Dipole (moderate)
Hydrogen (strongest)
What is Avogrado's Number?
6 x 10^23
Change in Electric Potential Energy:
PE = qV
What is the equation for ΔG′° ?
ΔG′° = –RTlnK
How many cm^3 in 1 liter?
1000
How do you solve for pressure given force?
P = F/A
How does bond enthalpy, length, and heat of combustion relate to stability?
↑ Stability = ↑ Bond Enthalpy = ↓ Bond Length = ↓ Heat of Combustion
What is Kcat?
Vmax/[E]
What is Vmax?
Maximum rate of reaction
What is Km?
the substrate concentration at 1/2 Vmax
What is V0?
initial rate of reaction
Competitive Inhibition Graph:
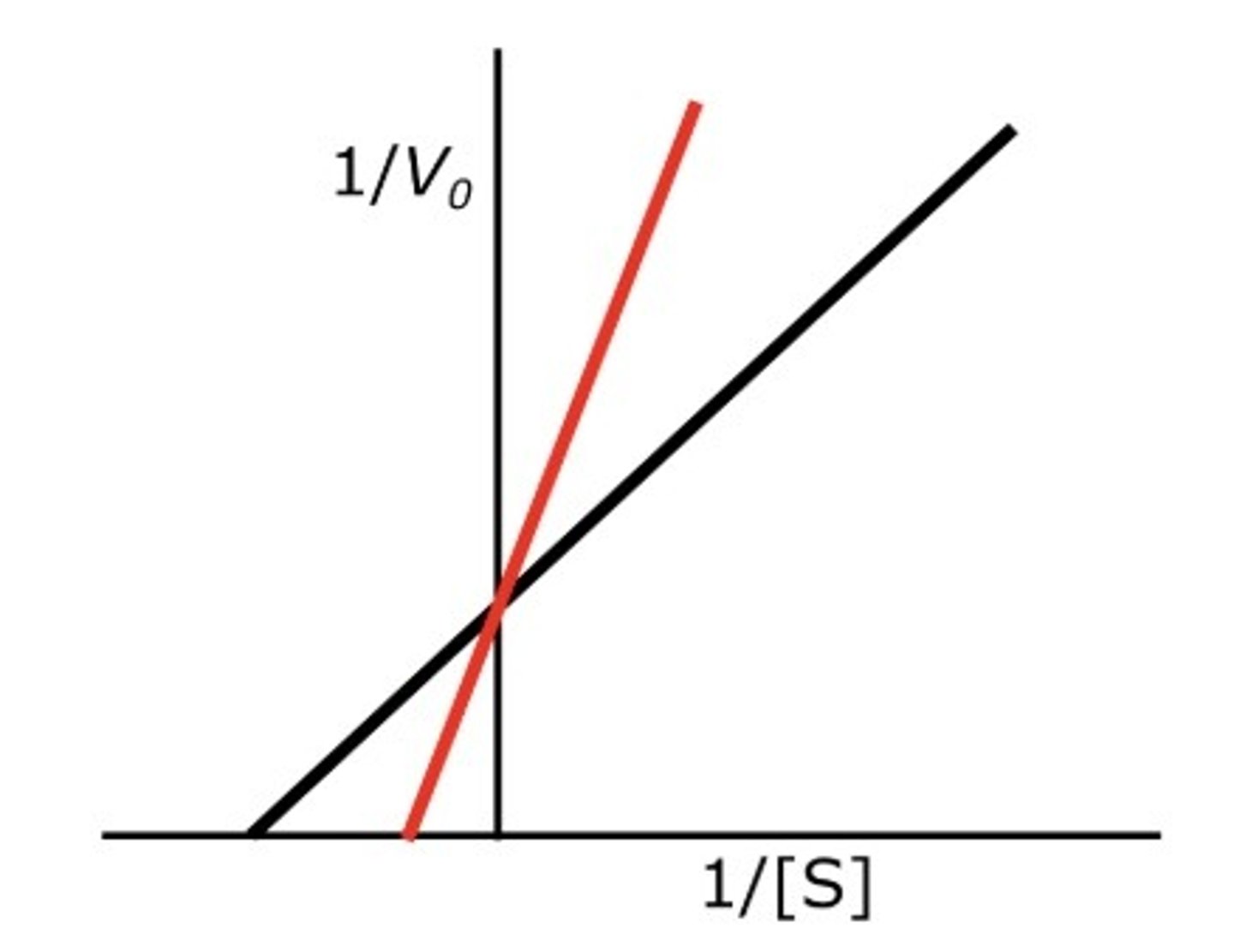
Uncompetitive Inhibition Graph:
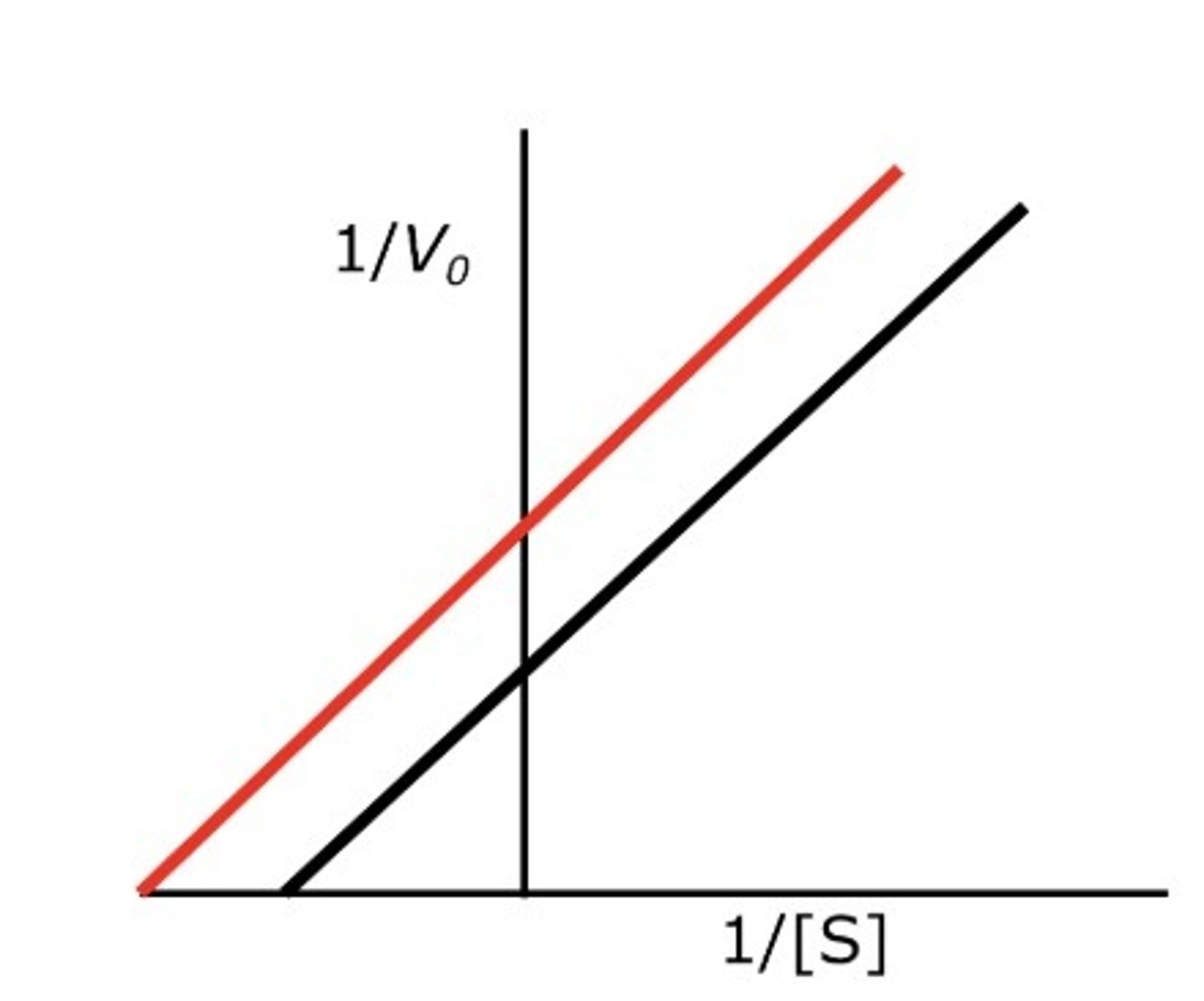
Noncompetitive Inhibition Graph:

Km is inversely proportional to _______. Why?
Km is inversely proportional to affinity.
A lower Km means the enzyme needs less substrate to reach half its maximum velocity, indicating a stronger attraction or higher affinity for the substrate.
If an enzyme has a high KM value, what does this suggest about its interaction with its substrate?
A. The enzyme is near Vmax
B. The enzyme has low affinity for the substrate
C. The enzyme is highly efficient
D. The enzyme is operating under allosteric regulation
B;
Km is inversely proportional with affinity.
If Km is higher, this means that more substrate is needed for an enzyme to reach half of its reaction rate.
In an enzyme-catalyzed reaction, which of the following conditions will directly increase Vmax?
A. Increasing substrate concentration at low [S]
B. Increasing enzyme concentration
C. Increasing temperature above the enzyme's optimal range
D. Decreasing the KM of the enzyme
B;
Increasing enzyme concentration increases the # of active sites available for a reaction to occur. As Vmax is the rate in which a reaction occurs, increasing the # of sites is directly proportional.
In an enzyme-catalyzed reaction where enzyme concentration is held constant and substrate concentration is relatively low, which kinetic parameter will increase with the addition of more substrate?
(Note: Other than substrate concentration, assume no other changes to reaction conditions.)
A. KM
B. kcat
C.Vmax
D. V0
D;
increasing substrate concentration will increase the initial rate of a reaction.
Equation for Cardiac Output:
CO = SV x HR