Chemical Bonding and Molecular Geometry: Ionic, Covalent, and Polarity
1/44
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
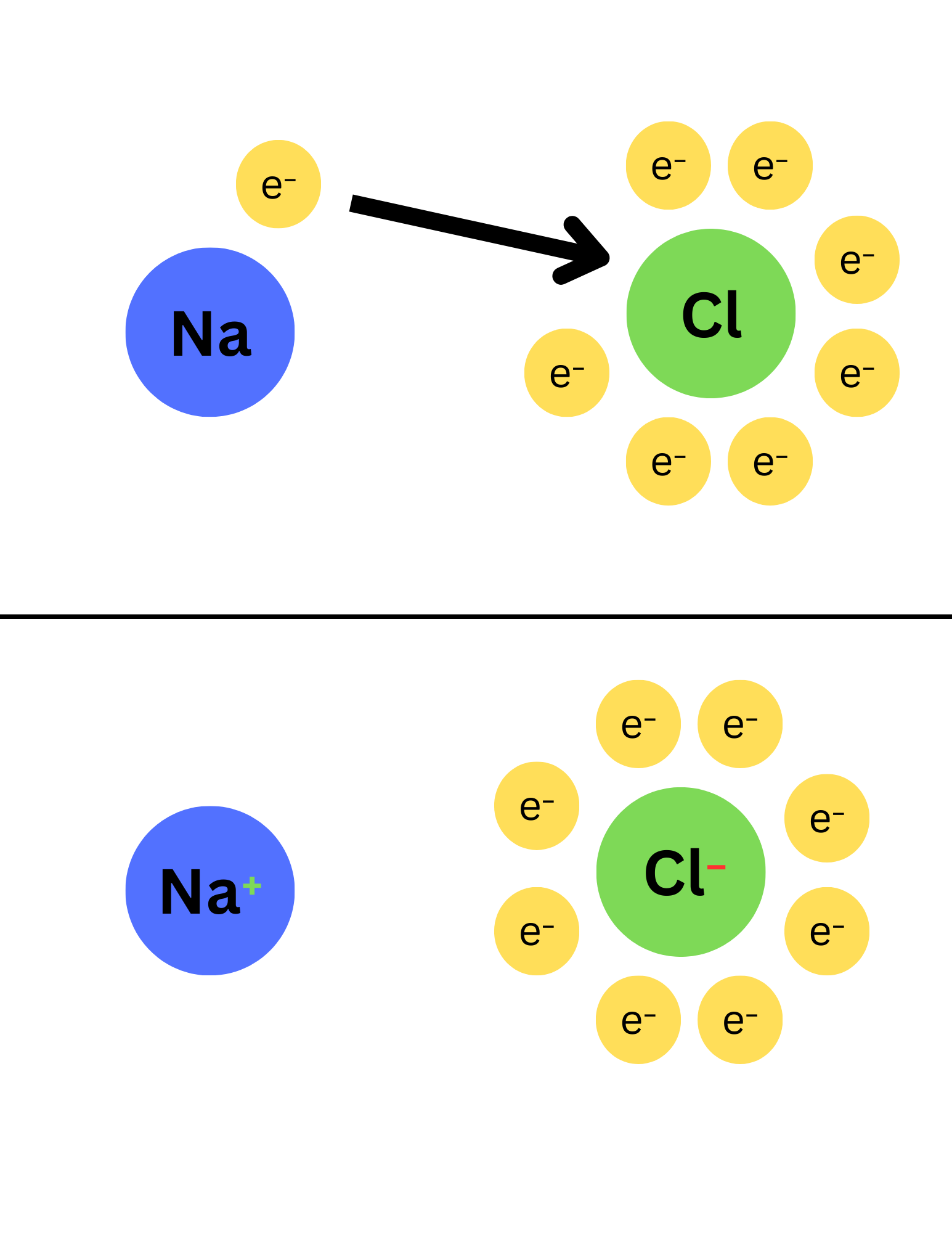
Ionic Compounds
-Metal + Nonmetal
-Transfer of electrons
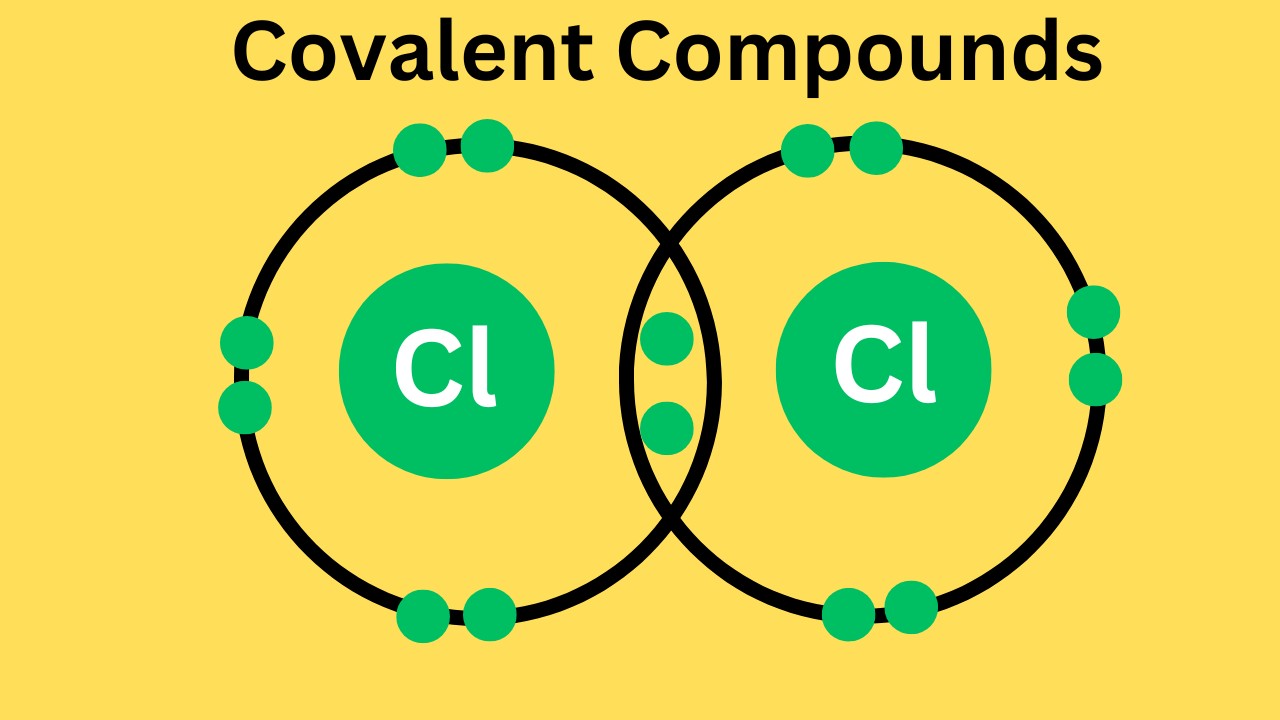
Covalent Bonds
-Two Nonmetals
-Electrons are shared
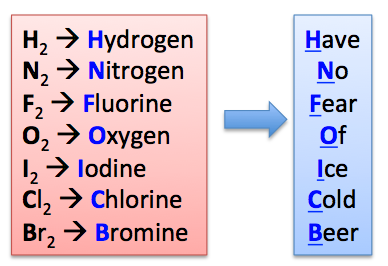
Diatomic molecules
Molecules consisting of two atoms, such as H2, N2, O2, F2, Cl2, Br2, I2.
H → (Hydrogen)
N → (Nitrogen)
O → (Oxygen)
F → (Fluorine)
Cl → (Chlorine)
Br → (Bromine)
I → (Iodine)
H → Have (Hydrogen)
N → No (Nitrogen)
O → Of (Oxygen)
F → Fear (Fluorine)
Cl → Cold (Chlorine)
Br → Beer (Bromine)
I → Ice (Iodine)
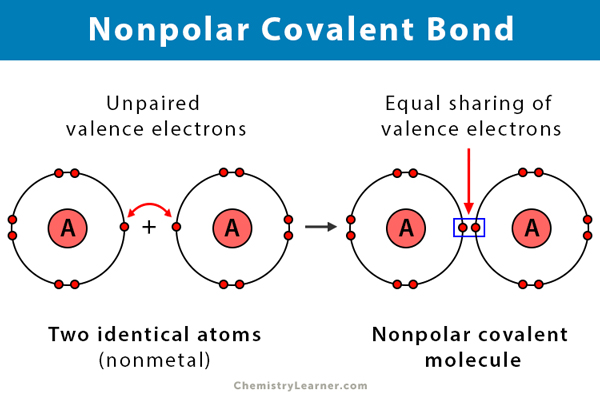
Nonpolar covalent bond
Covalent bond in which the atoms are identical (diamotics), so the electrons are shared equally or a bond between atoms with similar electronegativities.
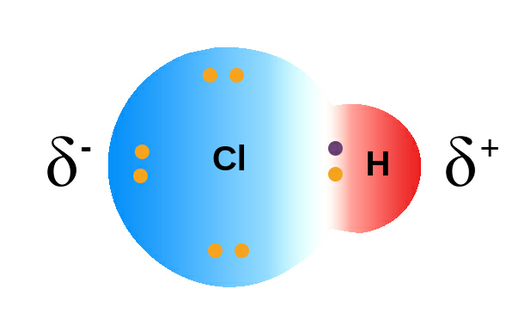
Polar covalent bond
A bond with unequal sharing of electrons, as one atom attracts the electrons more than the other atom. Resulting in partial positive and negative charges.
Polar Molecule
(also dipole) molecules with an overall dipole moment
Electronegativity
The relative ability of an atom in a molecule to attract shared electrons.
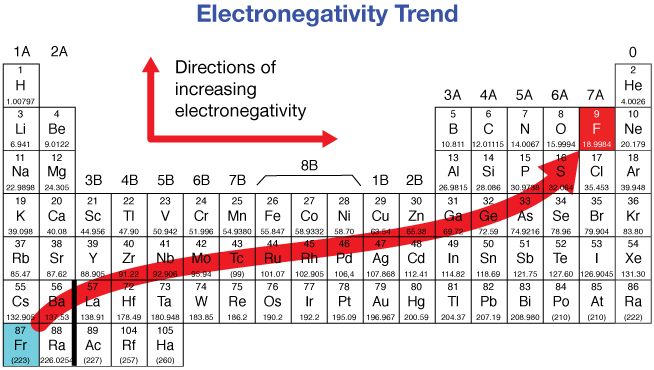
How does electronegativity change on the periodic table?
It generally increases across a period and decreases down a group.
What is the electronegativity range for elements?
From 4.0 for fluorine (most electronegative) to 0.7 for cesium and francium (least electronegative).
How to calculate electronegativity
Polarity of a bond depends on the difference between the electronegativity values of the atoms forming the bond

Electronegativity difference of Less than 0.4 is what bond type?
Nonpolar Covalent Bond

Electronegativity difference of Between 0.4 and 1.8 is what bond type?
Polar Covalent Bond

Electronegativity difference of Greater than 1.8 is what bond type?
Ionic Bond
Strong Electronegative Atoms…
-Pull electrons closer to them, resulting in electrons spending more time around one atoms compared to the other
-Partial change at the end of a bond
Symbol used to represent partial change
Delta (δ+, δ-)
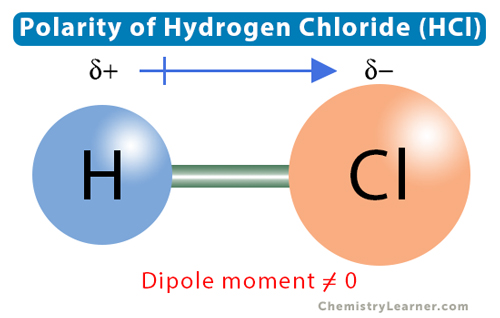
What does a dipole moment represent?
The charge distribution in a molecule with a center of positive and a center of negative charge.
Dipole character of a molecule is represented…
By an arrow
The arrow in a dipole molecule…
Points to the negative charge center, and its tail indicates the positive center of charge
The partially negative (δ-) side of a dipole molecule is…
Where more electrons will be because electrons have a negative charge, making the area partially negative
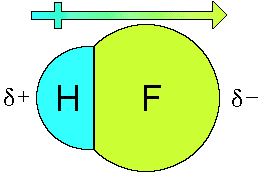
Explain Dipole Moment of HF
A hydrogen fluoride (HF) molecule will indeed have a dipole due to the difference in electronegativity between the hydrogen and fluorine atoms. In HF, the electronegativity of hydrogen is 2.2, and that of fluorine is 4.0, creating a significant polarity in the molecule. Because F is more electronegative, it’s partially negative while H is partially positive.
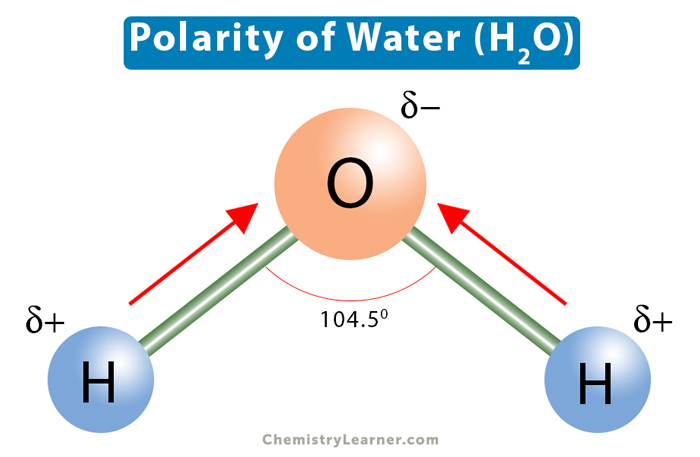
Dipole Moment in Water Molecule
-Permits ionic compounds to dissolve in it
-Causes water to remain a liquid at the temperatures on the earth’s surface
What is a Lewis Dot Diagram used for?
To describe valence electrons around atoms.
What is the octet rule?
Main group elements should have eight electrons around them to be stable.
What is the exception to the octet rule?
Hydrogen, which only needs 2 electrons (a duet) to fill its valence shell.
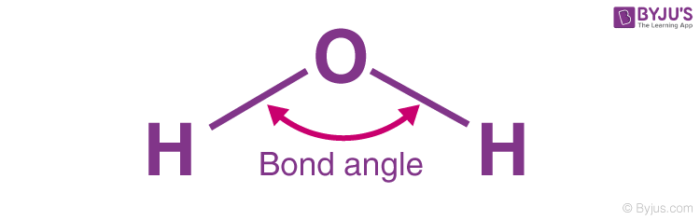
Bond Angle
Angle between any two covalent bonds that share a common atom
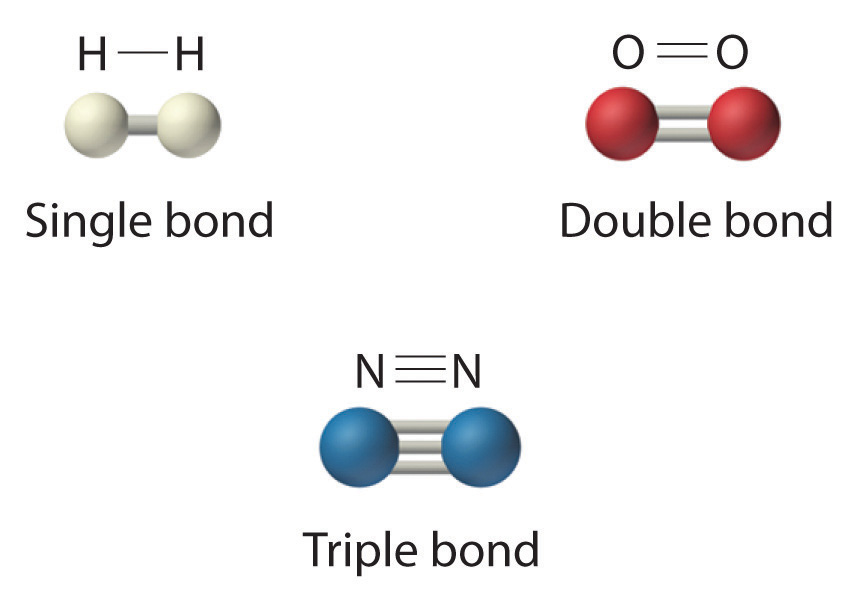
What are the types of covalent bonds based on shared electron pairs?
Single (1 pair), double (2 pairs), and triple (3 pairs) covalent bonds.
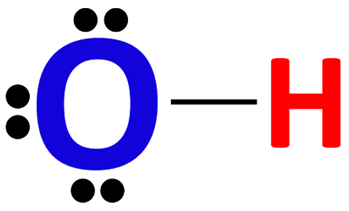
Lone Pair
(a pair of) valence electrons that are not used to form a covalent bond
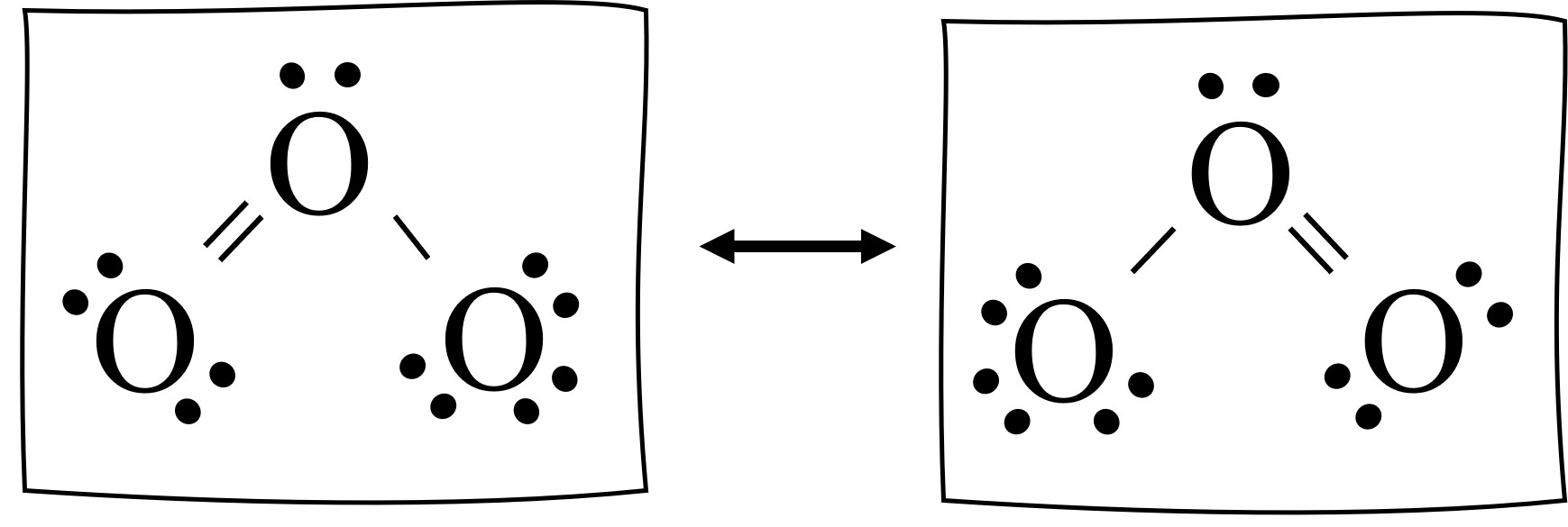
What is resonance in molecular structures?
When more than one Lewis structure can be drawn for a molecule. (use ↔ to separate resonance structures)
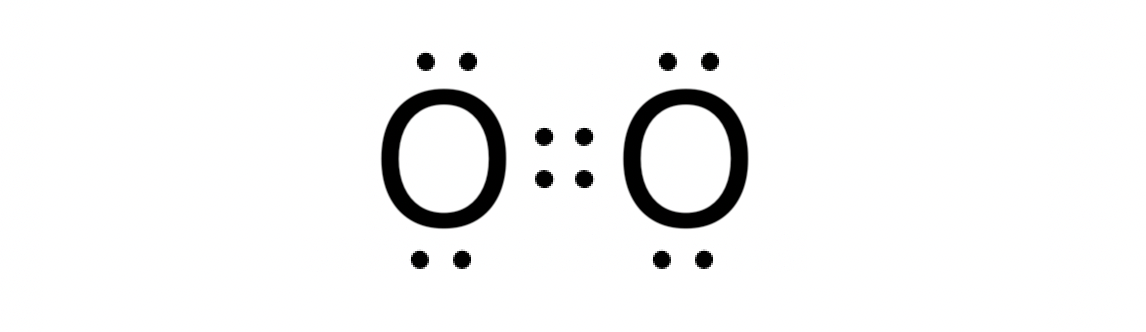
Give an example of a molecule with a double bond.
O2 (oxygen).

Give an example of a molecule with a triple bond.
N2 (nitrogen).
What does a Lewis structure show?
How atoms are bonded and the number of bonds between them, including lone pairs.
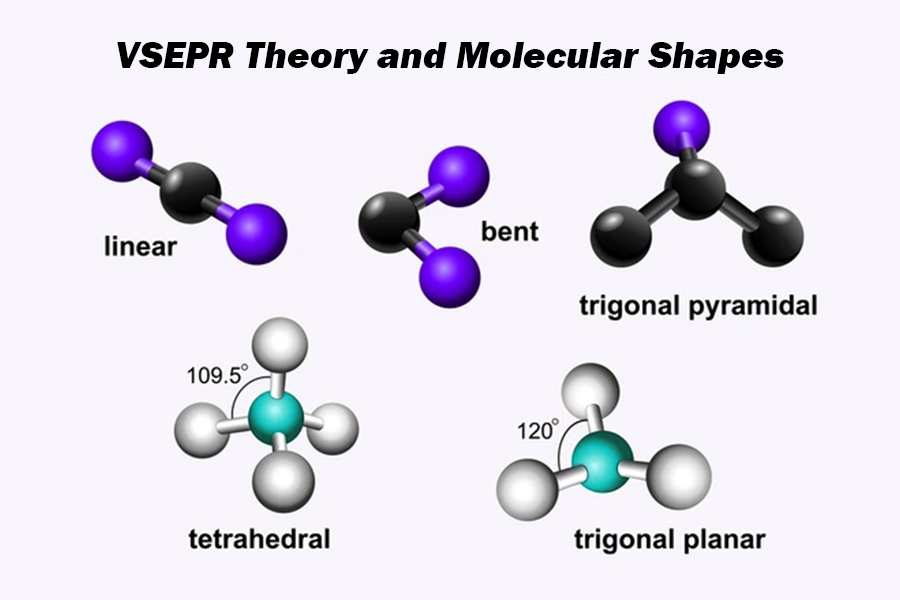
Valence Shell Electron Pair Repulsion theory (VSEPR)
A model used to predict 3-D molecular geometry based on the number of valence shell electron bond pairs among the atoms in a molecule or ion
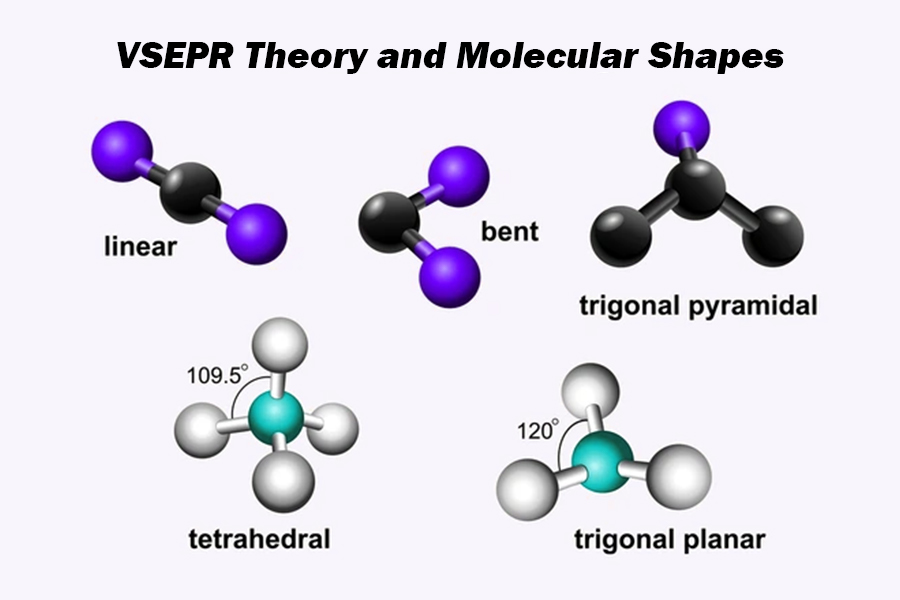
VSEPR
predicts the 3D shape of molecules by minimizing electrostatic repulsion between valence electron pairs (bonded and lone pairs) surrounding a central atom
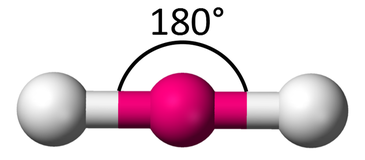
Molecular Shape: Linear
Total Electron Pairs: 2
Shared Pairs: 2
Lone Pairs: 0
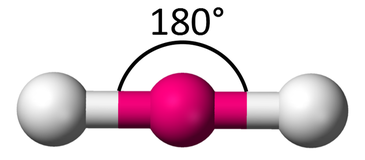
Angle of Linear
180°
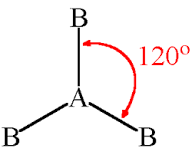
Molecular Shape: Trigonal Planar
Total Electron Pairs: 3
Shared Pairs: 3
Lone Pairs: 0
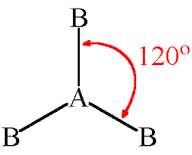
Angle of Trigonal Planar
120°
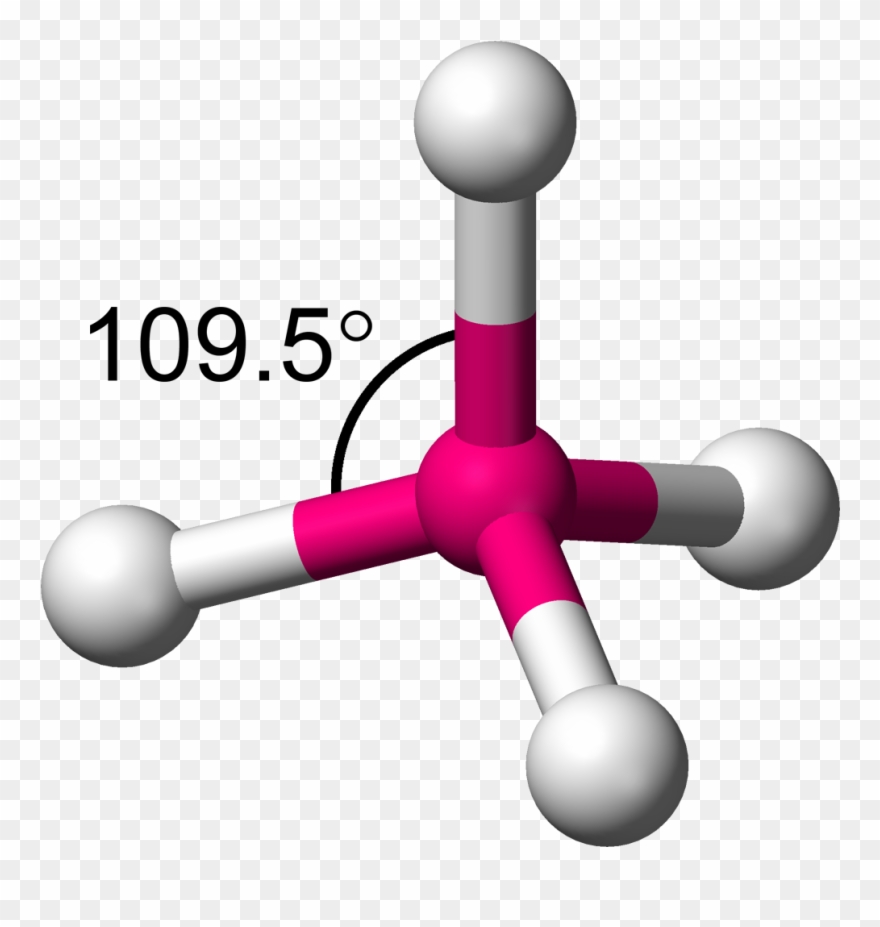
Molecular Shape: Tetrahedral
Total Electron Pairs: 4
Shared Pairs: 4
Lone Pairs: 0
Angle of Tetrahedral
109.5°
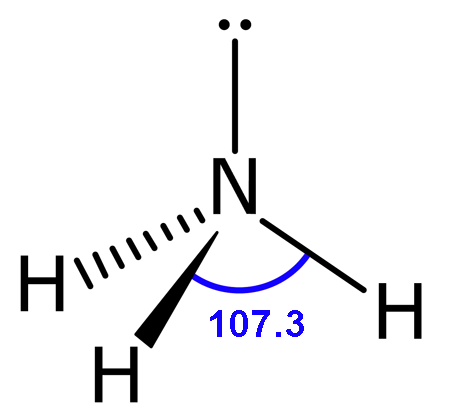
Molecular Shape: Trigonal Pyramidal
Total Electron Pairs: 4
Shared Pairs: 3
Lone Pairs: 1
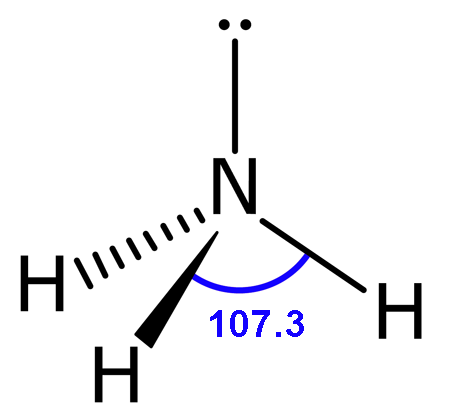
Angle of Trigonal Pyramid
107.3°
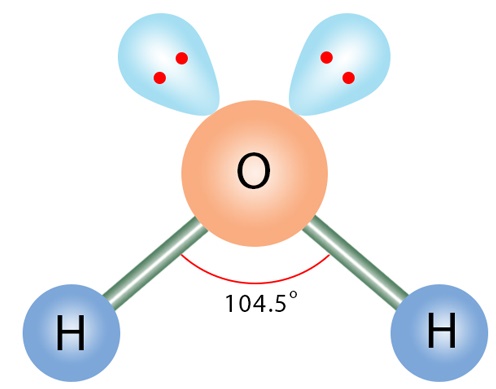
Molecular Shape: Angular / Bent
Total Electron Pairs: 4
Shared Pairs: 2
Lone Pairs: 2
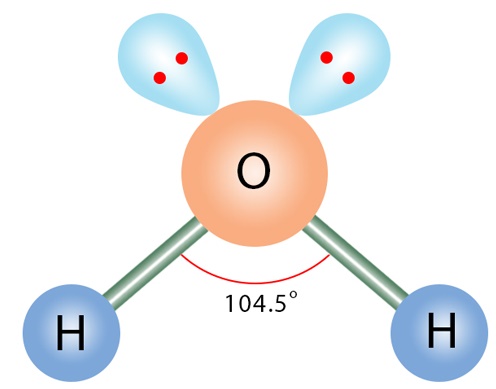
Angle of Angular / Bent
104.5°
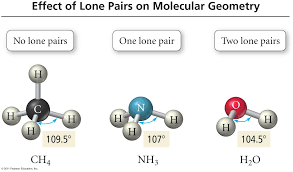
Lone Pairs affect Molecular Geometry by…
taking up space (changing the bond angle) and pushing atoms down instead of straight across