Topic 4: Chemical changes
1/66
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
67 Terms
Draw and state what the pH scale measures. (2 marks)
- The pH scale shows how acidic or alkaline a solution is.
- Neutral solutions have a pH of 7.
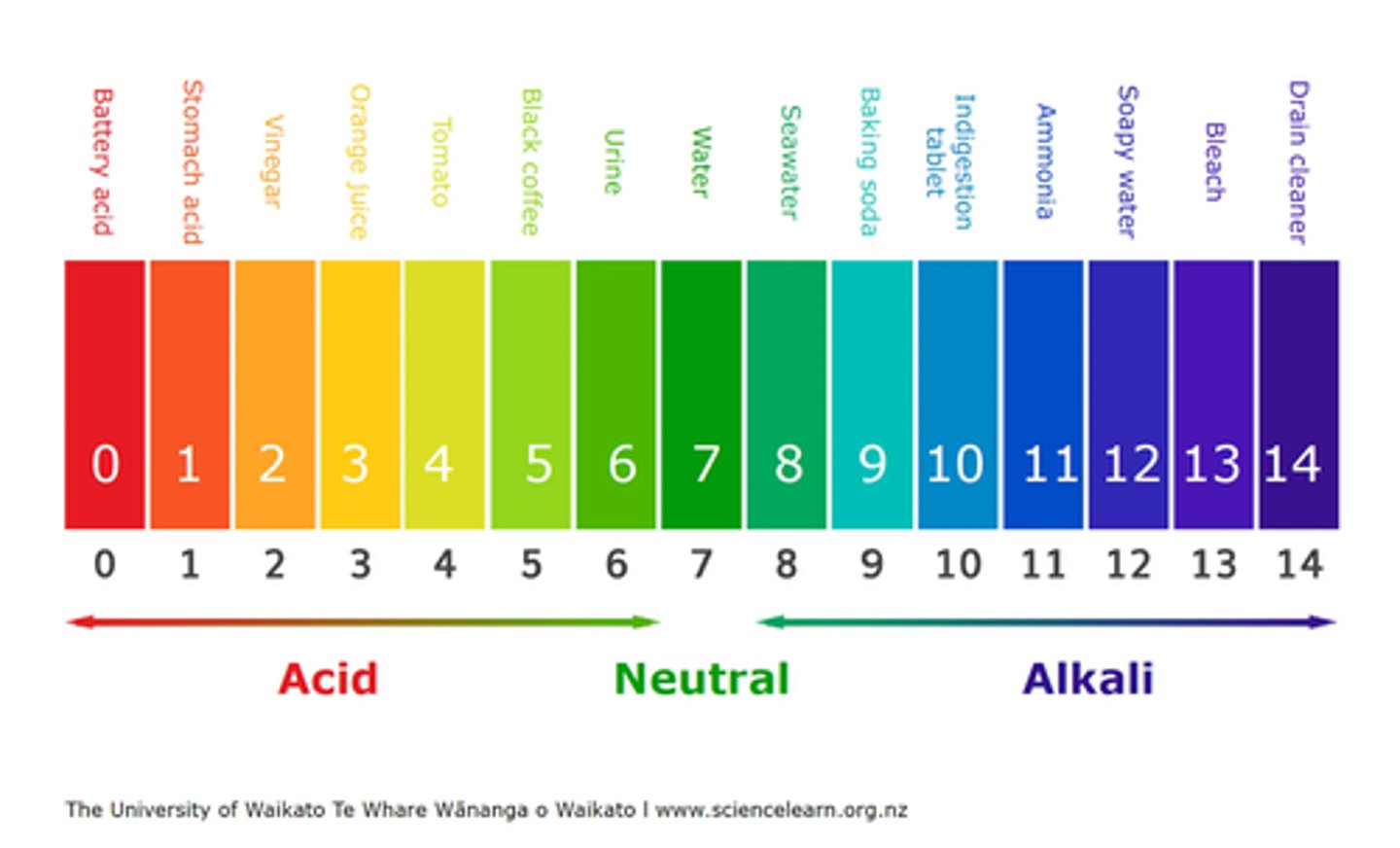
Explain how the pH of a solution can be measured. (3 marks)
- You can use an indicator that changes colour depending on the pH.
- Some indicators are universal and show a range of colours across the pH scale.
- A pH probe connected to a digital pH meter gives a numerical value for greater accuracy.
Define neutralisation and write the ionic equation for a neutralisation reaction. (2 marks)
- Neutralisation is the reaction between an acid and a base to form salt and water.
- H⁺ + OH⁻ → H₂O
Describe what happens to the pH during a neutralisation reaction. (2 marks)
- The pH moves towards 7.
- This happens because H⁺ ions from the acid react with OH⁻ ions from the alkali to make water.
State two substances formed when an acid reacts with an alkali. (2 marks)
- A salt is produced.
- Water is also formed.
State what ions acids and alkalis produce in solution. (2 marks)
- Acids release H⁺ ions.
- Alkalis produce OH⁻ ions.
Explain how to improve the accuracy of titration results. (3 marks)
- Repeat the titration until you get at least three similar results.
- Ignore any readings that don't fit the pattern (anomalous results).
- Calculate a mean value using only the consistent results.
State why you should do a rough titration first and explain why universal indicator is not suitable for titrations. (2 marks)
- To get an idea of roughly where the colour change (end-point) happens.
- It shows a gradual colour change instead of a sudden one at the end-point.
Describe the colour changes of three single indicators during titrations. (3 marks)
- Litmus: red in acid, blue in alkali.
- Phenolphthalein: colourless in acid, pink in alkali.
- Methyl orange: red in acid, yellow in alkali.
State what acids produce when dissolved in water and define the term ionisation in the context of acids. (2 marks)
- Acids release hydrogen ions (H⁺) when they dissolve in water.
- Ionisation is when acid molecules split up in solution to release hydrogen ions.
Describe the difference between strong acids and weak acids. (3 marks)
- Strong acids fully ionise in water, releasing all their H⁺ ions.
- Weak acids only partly ionise, so only some molecules release H⁺ ions.
- The ionisation of weak acids is reversible, forming an equilibrium between ions and un-ionised acid.
Describe how to carry out a titration to find the concentration of an acid or alkali. (6 marks)
1. Use a pipette and pipette filler to transfer a measured volume of the alkali into a conical flask.
2. Add a few drops of indicator to the alkali in the flask.
3. Fill a burette with acid of known concentration using a funnel, making sure the reading is below eye level.
4. Slowly add the acid from the burette into the flask, swirling the solution regularly.
5. Stop adding acid when the indicator shows that the solution has been neutralised (this is the end-point).
6. Record the final burette reading, subtract from the initial volume to calculate the volume of acid used.
State two examples of strong acids and state two examples of weak acids. (4 marks)
- Strong acids: hydrochloric acid, sulfuric acid.
- Weak acids: ethanoic acid, citric acid.
Explain why strong acids are more reactive than weak acids of the same concentration. (2 marks)
- Strong acids have more free H⁺ ions in solution.
- This increases the rate of reaction because there are more ions available to react.
State the relationship between pH and hydrogen ion concentration and state how the hydrogen ion concentration changes when the pH decreases by 1. (2 marks)
- A lower pH means a higher concentration of hydrogen ions.
- The H⁺ ion concentration increases by a factor of 10.
Explain what happens to the H⁺ ion concentration if pH drops by 2. (2 marks)
- The concentration increases by a factor of 100.
- Each pH unit represents a ×10 change, so 10 × 10 = 100.
Explain the difference between acid strength and acid concentration. (2 marks)
- Strength refers to how much the acid ionises in solution.
- Concentration refers to how many acid particles are dissolved in a set volume.
Describe how you can have a concentrated weak acid or a dilute strong acid. (2 marks)
- A concentrated weak acid has lots of acid particles but only partially ionises.
- A dilute strong acid has fewer acid particles but all of them ionise fully.
State the general word equation for the reaction between an acid and a metal oxide or metal hydroxide. (2 marks)
- Acid + metal oxide → salt + water.
- Acid + metal hydroxide → salt + water.
Describe what happens when acids react with metal oxides or hydroxides. (2 marks)
- These reactions are neutralisations and produce salt and water.
- Even if the base is insoluble, it can still react with the acid.
State two examples of reactions between acids and metal hydroxides. (2 marks)
- Sulfuric acid + potassium hydroxide → potassium sulfate + water
- Nitric acid + sodium hydroxide → sodium nitrate + water
State the general word equation for an acid reacting with a metal carbonate and state the symbol equation for HCL and CaCO3. (2 marks)
- Acid + metal carbonate → salt + water + carbon dioxide.
- 2HCL + Na2CO3 → 2NaCl + H2O + CO2
Describe how to make a soluble salt using an insoluble base. (5 marks)
1. Pick a suitable acid and an insoluble base, like a metal oxide or carbonate.
2. Warm the acid gently using a Bunsen burner, then turn the flame off.
3. Add the base slowly until no more dissolves and the excess settles.
4. Filter out the leftover base to get a solution of salt and water.
5. Heat the solution gently and leave it to cool so salt crystals form, then filter and dry them.
Write out the reactivity series and state what it shows. (11 marks)
It shows the order of how easily metals react compared to each other.
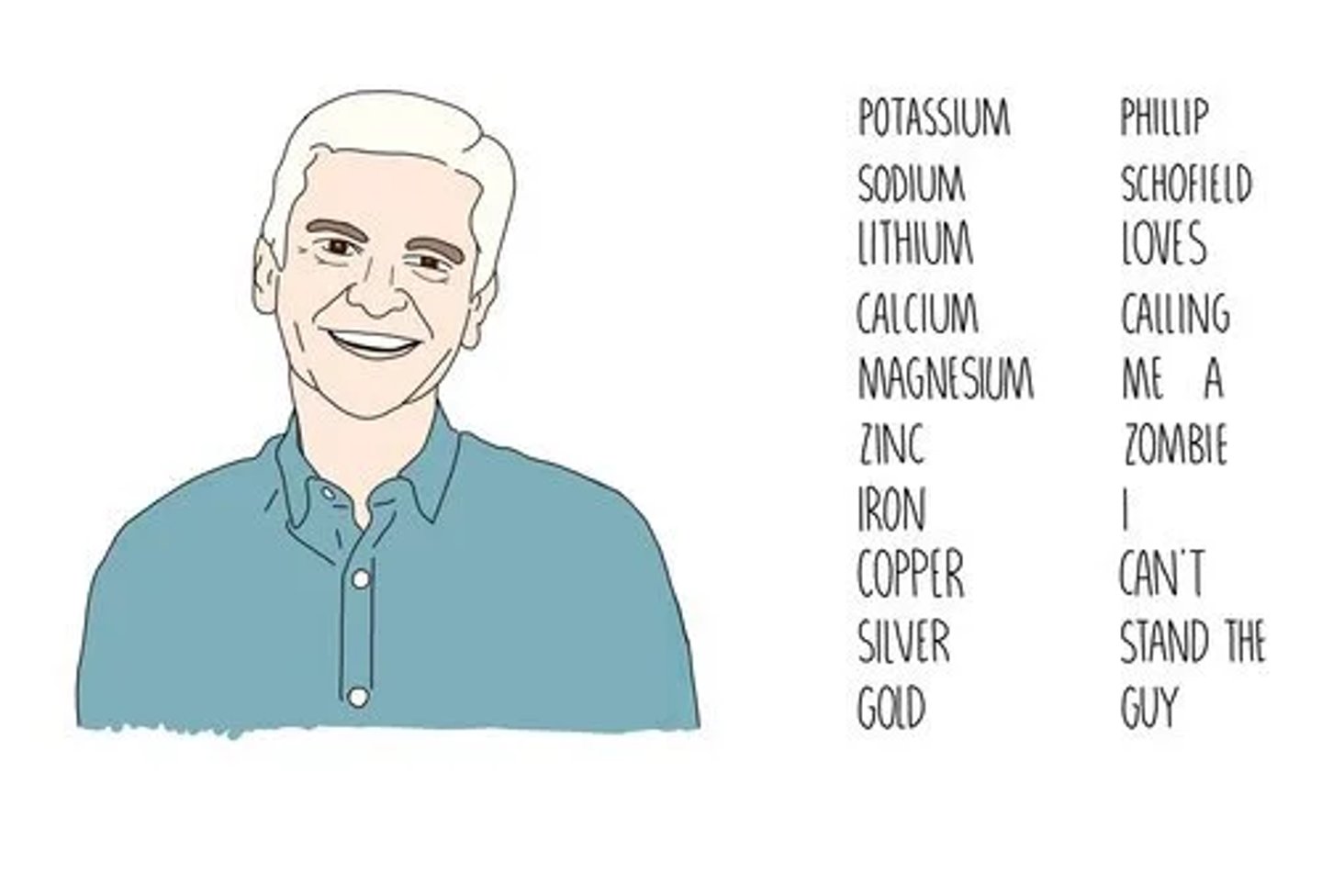
Explain what the reactivity of a metal depends on. (2 marks)
It depends on how easily it loses electrons.
The more easily it forms positive ions, the more reactive it is.
State the general word equation for a metal reacting with an acid and describe what happens when a metal reacts with an acid. (3 marks)
- Metal + acid → salt + hydrogen.
- It forms a salt and hydrogen gas.
- You can observe fizzing as bubbles of hydrogen are released.
Explain how the rate of a metal's reaction with acid indicates its reactivity. (2 marks)
- A faster reaction (more bubbles) shows higher reactivity.
- Very reactive metals react quickly and sometimes violently.
Describe how you could use temperature change to compare metal reactivities. (2 marks)
- Use the same amount and surface area of metal with acid each time.
- The metal that causes the biggest temperature rise is the most reactive.
State the general word equation for a metal reacting with water. (1 mark)
Metal + water → metal hydroxide + hydrogen
State examples of metals that react with cold water and do not react with cold water. (7)
- React with cold water = potassium, sodium, lithium, and calcium.
- Don't react with cold water = zinc, iron and copper.
State what must happen to metal oxides to extract the metal. (2 marks)
- The metal oxide must be reduced.
- Which means that oxygen is removed from it.
Define oxidation and reduction in terms of oxygen. (2 marks)
- Oxidation is the gain of oxygen.
- Reduction is the loss of oxygen.
Describe how carbon is used to extract some metals. (3 marks)
- The metal oxide is heated with carbon.
- The metal loses oxygen, so it's reduced.
- The carbon gains oxygen and is oxidised.
Explain why some metals can't be extracted using carbon. (2 marks)
- They are more reactive than carbon.
- Carbon can't remove oxygen from more reactive metals.
State which method is used to extract metals more reactive than carbon. (1 mark)
Electrolysis.
State the one metal that is found uncombined in the Earth. (1 mark)
Gold
Define oxidation and reduction in terms of electrons. (2 marks)
- Oxidation is loss of electrons.
- Reduction is gain of electrons.
Explain what happens in a redox reaction. (2 marks)
- One substance is oxidised and loses electrons.
- Another is reduced and gains those electrons.
Describe what happens to Fe atoms when they react with acid. (3 marks)
- Fe atoms lose 2 electrons and become Fe²⁺ ions.
- They are oxidised.
- Hydrogen ions gain electrons and form hydrogen gas.
Define a displacement reaction and state what happens when iron is placed in copper sulfate solution. (3 marks)
- In a displacement reaction, a more reactive metal replaces a less reactive metal in a compound.
- Iron displaces copper and forms iron sulfate.
- Copper metal is left behind.
Explain why displacement reactions are redox reactions. (2 marks)
- The more reactive metal is oxidised (loses electrons).
- The less reactive metal is reduced (gains electrons).
State what happens to electrons during displacement of copper by iron. (2 marks)
- Iron loses electrons and becomes Fe²⁺ (oxidised).
- Copper ions gain electrons and become copper metal (reduced).
State the ionic equations for the reaction of iron with copper sulfate. (2 marks)
- Fe → Fe²⁺ + 2e⁻ (oxidation)
- Cu²⁺ + 2e⁻ → Cu (reduction)
State what spectator ions are and describe the purpose of ionic equations. (2 marks)
- Ions that don't take part in the reaction and stay the same.
- They show only the substances involved in oxidation or reduction.
Define electrolysis and state what type of substances can be used as electrolytes. (2 marks)
- Electrolysis is the process of using an electric current to split up a compound.
- Electrolytes are molten or dissolved ionic compounds.
Explain what happens to positive and negative ions during electrolysis. (2 marks)
- Positive ions move to the negative electrode (cathode) and gain electrons.
- Negative ions move to the positive electrode (anode) and lose electrons.
Describe what happens to ions when they gain or lose electrons at the electrodes. (2 marks)
- They form neutral atoms or molecules.
- These are discharged from the electrolyte.
Explain why ionic solids cannot be electrolysed and state why molten ionic compounds can be electrolysed. (2 marks)
- Ionic solids' ions are locked in place and cannot move.
- Molten ionic compounds' ions are free to move and carry a charge.
State an example of a molten ionic substance and what it produces at each electrode. (2 marks)
- Molten lead bromide.
- Which forms lead at the cathode and bromine at the anode.
State the half equations for the electrolysis of molten lead bromide. (2 marks)
- Pb²⁺ + 2e⁻ → Pb
- 2Br⁻ → Br₂ + 2e⁻
Describe how aluminium is extracted from its ore. (3 marks)
- Aluminium is extracted by electrolysis from aluminium oxide (found in bauxite).
- The ore is mixed with cryolite to reduce the melting point.
- The molten mixture is electrolysed to separate the aluminium.
Explain why cryolite is used in aluminium extraction. (2 marks)
- It lowers the melting point of aluminium oxide.
- Which saves energy.
State what happens to aluminium ions during electrolysis. (2 marks)
- Al³⁺ ions go to the cathode and gain 3 electrons.
- This forms aluminium metal, which sinks to the bottom.
State what happens to oxide ions at the anode. (2 marks)
- O²⁻ ions lose electrons to form oxygen gas.
- The oxygen molecules are released at the anode.
Write the half equation for aluminium at the cathode, oxygen at the anode and the overall equation for the extraction of aluminium. (3 marks)
- Al³⁺ + 3e⁻ → Al
- 2O²⁻ → O₂ + 4e⁻
- 2Al₂O₃ → 4Al + 3O₂
Explain why the anodes used in aluminium extraction must be replaced regularly. (1 mark)
Oxygen reacts with the carbon anode to form carbon dioxide.
Explain why you must consider water when electrolysing an aqueous solution. (2 marks)
- Water also breaks down into ions: H⁺ and OH⁻.
- These ions compete with the ions from the dissolved compound at the electrodes.
Describe what is formed at the cathode during electrolysis of an aqueous solution. (2 marks)
- If the metal is more reactive than hydrogen, hydrogen gas is made.
- If the metal is less reactive than hydrogen, a layer of the pure metal forms.
State what is formed at the anode during electrolysis of an aqueous solution. (2 marks)
- If halide ions are present, molecules like Cl₂ or Br₂ are produced.
- If no halides are present, OH⁻ is discharged and oxygen is formed.
State the half equations for electrolysis of copper(II) sulfate solution. (2 marks)
- Cathode: Cu²⁺ + 2e⁻ → Cu
- Anode: 4OH⁻ → O₂ + 2H₂O + 4e⁻
Describe what you would observe during the electrolysis of copper(II) sulfate solution. (2 marks)
- A pink-brown layer of copper forms on the cathode.
- Bubbles of oxygen are released at the anode.
Explain why copper is produced instead of hydrogen in copper sulfate solution. (2 marks)
- Copper is less reactive than hydrogen.
- So it is discharged first.
State the ionic half equation for OH⁻ when no halides are present. (1 mark)
4OH⁻ → O₂ + 2H₂O + 4e⁻
State the half equations for electrolysis of sodium chloride solution. (2 marks)
- Cathode: 2H⁺ + 2e⁻ → H₂
- Anode: 2Cl⁻ → Cl₂ + 2e⁻
Describe what you would observe when electrolysing sodium chloride solution. (2 marks)
- Hydrogen gas is released at the cathode.
- Chlorine gas is released at the anode.
State how you could test the gases produced during electrolysis. (3 marks)
- Chlorine bleaches damp litmus paper, turning it white.
- Hydrogen makes a squeaky pop with a lit splint.
- Oxygen relights a glowing splint.
Still learning (11)
You've begun learning these terms. Keep up the good work!