Topic 8 energetics
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
definition of enthalpy change
heat energy change measured at constant pressure
standard conditions
100Kpa and 298k
c to k is +273
what's an exothermic reaction and how how does enthalpy level diagram look like?
-give out heat energy
-negative sign
-temp often goes up

whats an endothermic reaction and what does the enthalpy level diagram look like?
-absorb energy
-positive sign
-temp often falls

the less enthalpy a substance has , is it more or less stable?
more
reaction profile diagram for exothermic reaction and endothermic reaction.

definition of Standard enthalpy change of reaction
Enthalpy change when reaction occurs in the molar quantities shown in the chemical equation under standard conditions.
definition of standard enthalpy change of neutralisation
Enthalpy change when an acid and alkali react together under standard conditions to form 1 mole of water
- always exothermic
definition of Standard enthalpy of combustion
Enthalpy change when 1 mole of a substance is completely burned in oxygen under standard conditions.
definition of Standard enthalpy change of formation
Enthalpy change when 1 mole of a compound is formed from its elements in their standard states under standard conditions.
what is calorimetry used for
to find enthalpy changes of combustion
describe apparatus of calorimetry
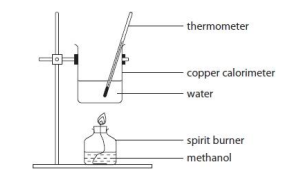
what are possible errors in measuring enthalpies of combustion using a calorimetry?
• Energy losses from calorimeter
• Incomplete combustion of fuel
• Incomplete transfer of energy
• Evaporation of fuel after weighing
• Heat capacity of calorimeter not included
• The experiment was not carried out under standard conditions because water formed as H₂O(g) rather than H₂O(l), which is the standard state in the data booklet. This means the measured enthalpy change is less exothermic than the data-booklet value
what are the possible errors in measuring enthalpy change for a reaction experimentally using a calorimeter?
• energy transfer from surroundings (usually loss)
• approximation in specific heat capacity of solution. The method assumes all solutions have the heat capacity of water.
• neglecting the specific heat capacity of the calorimeter- we ignore any energy absorbed by the apparatus.
• reaction or dissolving may be incomplete or slow.
• density of solution is taken to be the same as water.
equation for enthalpy change of combustion
q = mc∆T
q is KJ
J to KJ is divide by 1000
whats hess's law and why is it used?
The total enthalpy change of a reaction is always the same, no matter which route is taken
-to find enthalpy changes that cant be worked out experimentally
enthalpy change of formation for elements is
zero
where does arrows point in enthalpy change of formation
upwards
it always products minus reactants for the route
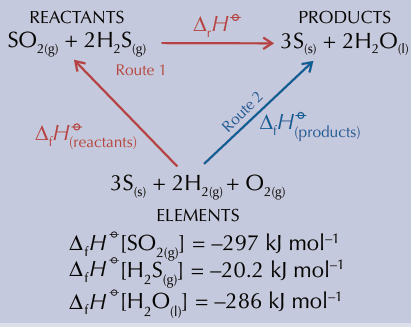
where does arrows point in enthalpy change of combustion
downwards
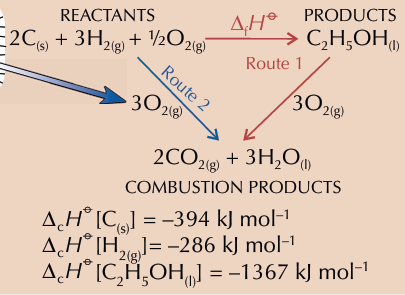
what happens when bonds break in reactants?
energy is absorbed
endothermic process so delta H is positive
what happens when bonds are formed in products?
energy is released
exothermic process so delta H is neg
define bond enthalpy
is the amount of energy required to break one mole of a type of bond in a molecule in the gas phase
why would the data booklet values for mean bond enthalpy be different to actual ones?
mean bond enthalpy is the energy needed to break one mole of bonds in the gas phase, averaged over many different compounds.
write the formula for enthalpy change of reaction involving bond enthalpies
sum of bond enthalpies of reactants - sum of bond enthalpies of products