Bile Salts & Steroid Biosynthesis
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms

this lecture follows the cholesterol metabolism lecture because the liver cells synthesize bile salts from cholesterol.
once synthesize, bile salts are then stored in the gallbladder and released in the small intestine to aid in digestion and absorption of fats.


we must recognize that the conversion of cholesterol to 7a-hydroxy cholesterol is the rate limiting step in bile salt synthesis.
This step is crucial because it is the first committed step in the bile salt synthesis pathway of bile salts.
the reaction is catalyzed by the enzyme cholesterol-7a-hydroxylase, also known as CYP7A1.
bile salts synthesize from cholesterol inhibit their production by feedback inhibition.
chenodeoxycholic acid and cholic acid from cholesterol synthesis involve several enzymatic reactions within the hepatocytes.
bile salt conjugations, consist of attachment of taurine, or glycine, to the carboxyl group of the bile acid molecule.


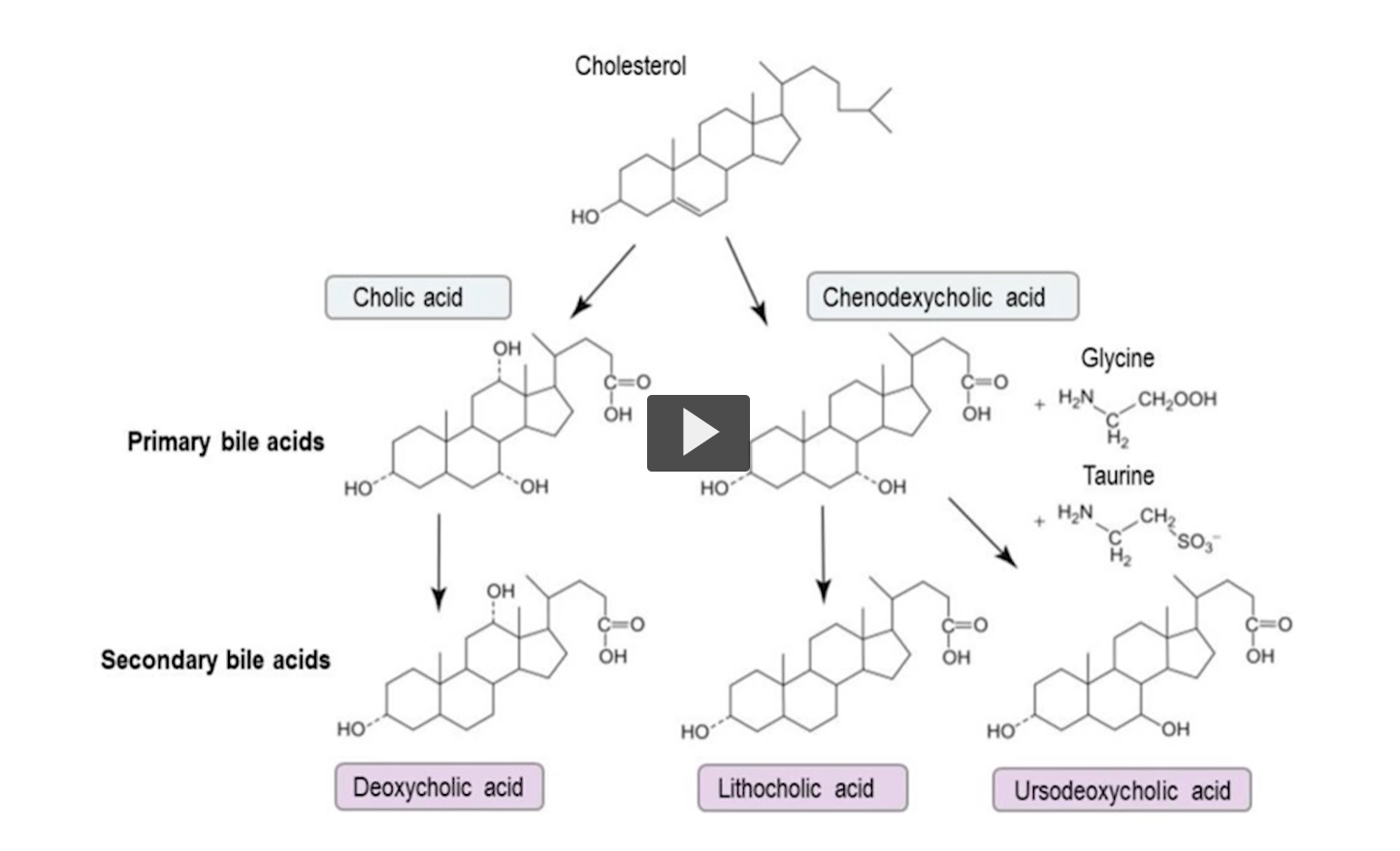
we will contrast primary verses secondary bile salts.
Primary bile salts are synthesized by the liver from cholesterol, secondary bile salts are formed from the action of intestinal bacteria on primary bile salts.
the fate of primary bile salts and secondary bile salts involves dynamic interplay between absorption and excretion.
lastly, we will explore steroid biosynthesis, a tightly regulated process involving multiple enzymes and intermediate molecules.
steroids regulate various physiological processes, including metabolism, reproduction, immune function and stress response
bile salts are detergents used in our body to solubilize lipids for digestion. Lipids are not soluble in water, thus bile salts make them accessible to processing enzymes that aid in digestion in digestion and absorption.
Bile salts do function chemically by having the access of polar lipids, meaning that there is hydrophobic and polar portions of these molecules.
Those molecules are setup in the water, they allow for you to aggregate polar lipids in an aqueous environment that then allow for accessing of enzymes, that is how detergents work.
Bile acids are synthesized in the liver by a multistep, multiorganelle pathway.
Hydroxyl groups are inserted at specific positions on steroid structure. The double bond on the cholesterol B ring is reduced. The three carbons shorten the hydrocarbon chain, introducing a carboxyl group at the end of the chain.

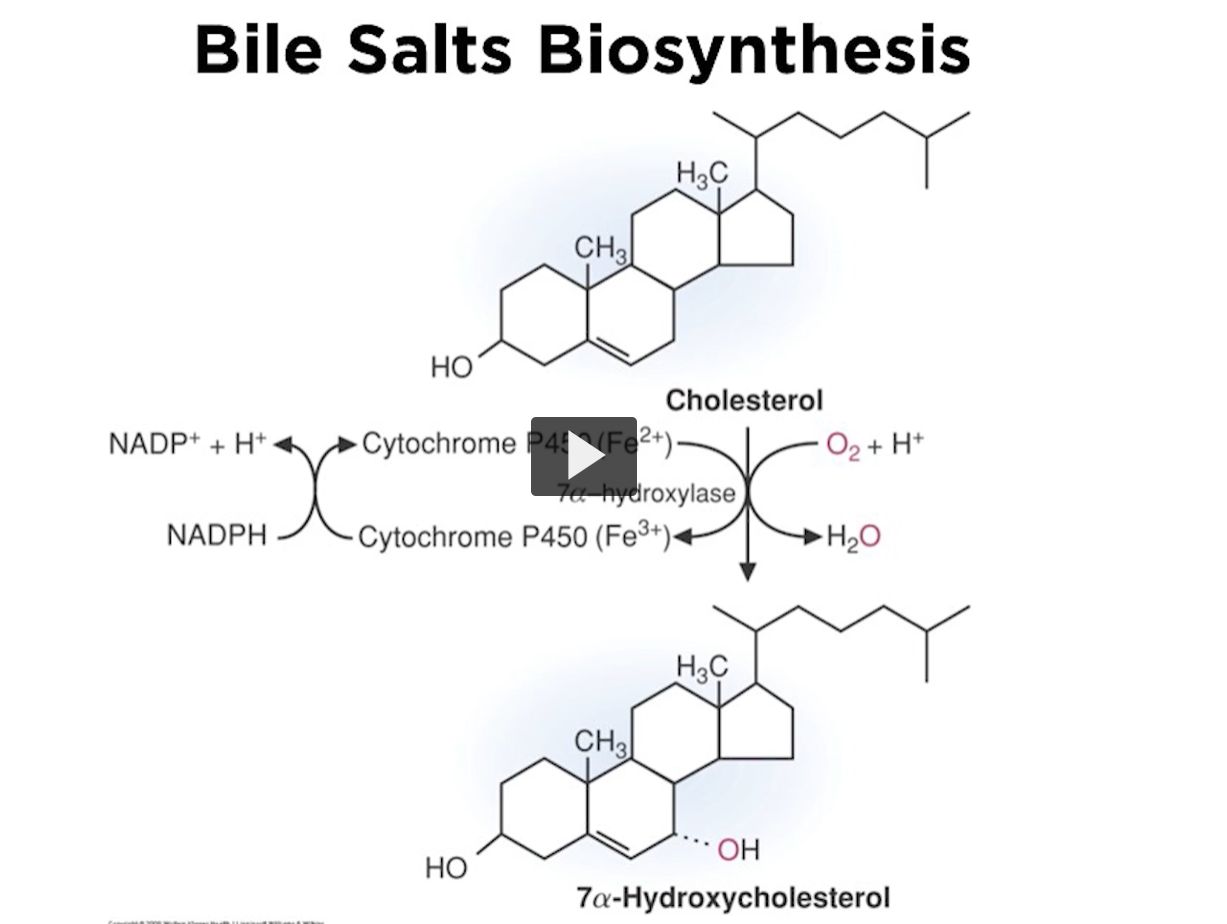
bile salt biosynthesis
Here is the first reaction in which bile salts are synthesized.
cholesterol is made into 7a-hydroxycholesterol.
this is the first step, the committed step, and the step that is under control.
the enzyme carrying out this reaction is 7a-hydroxylase. Like any hydroxylase, it uses NADPH as a reducing agent.
You use cytochrome P450 and iron.
There is an incorporation of oxygen at position #7, this is a monoxygenase enzyme, meaning that there are two atoms in the oxygen molecule and the one of the atoms ends up in organic moiety.
It is essential to point out that 7a-hydroxylase recognizes cholesterol as the only substrate.
This is a very rigid reaction, because the enzyme only recognizes cholesterol.
7A-hydroxylase is controlled by feedback, the concentration of the end product will regulate the combined, committed step.
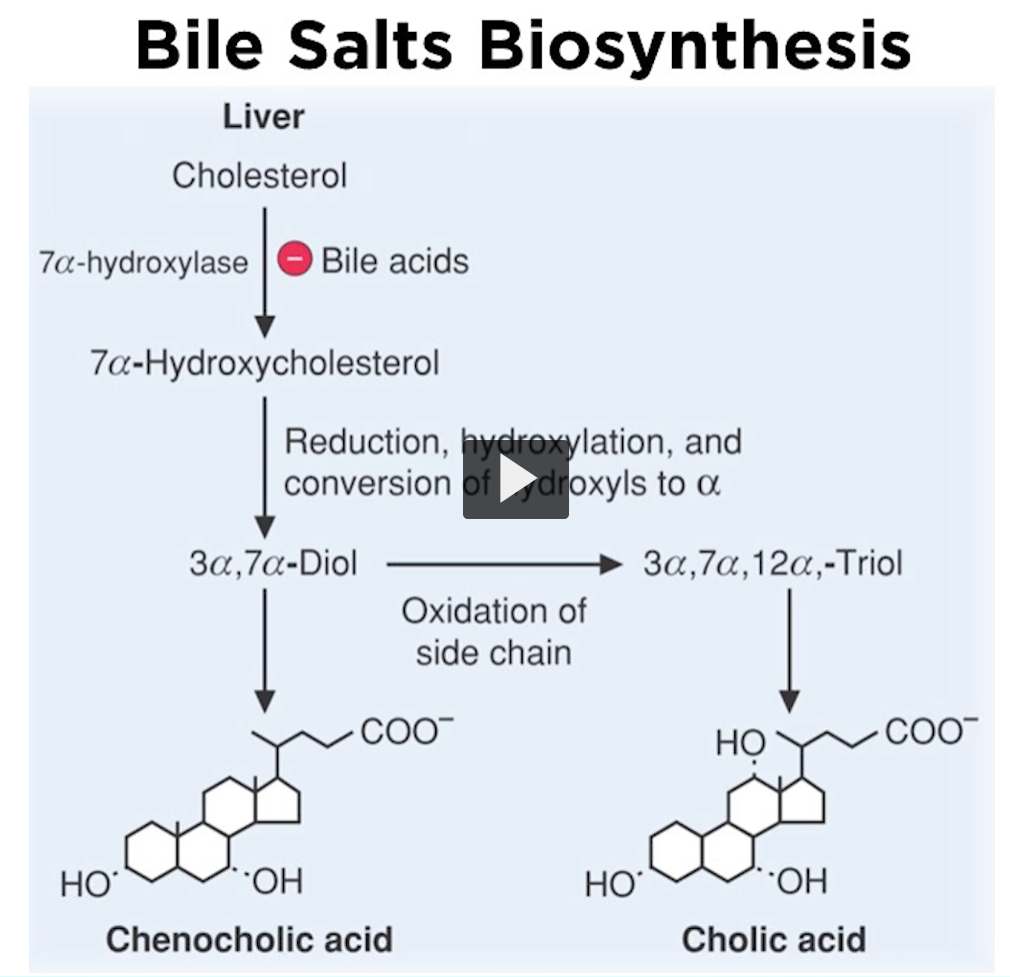
so, what happens after you formed 7a-hydroxylcholesterol. Remember, that you want bile salts to be detergents, you want to make these molecules polar, so they can form micelles in an aqueous environment.
In the micelle center is the hydrophobic portion, where other lipids can solubilize.
A serious of reactions must occur, including reductions, hydroxylation, and the oxidation of the side chain of cholesterol
Remember that the side chain of cholesterol is carbon and hydrogen that you can use to oxidize to form carboxylic acid.
These reactions allow you to form chenocolic and cholic acids, both are bile salts.
They have a negatively charged carboxyl and hydrophobic region, so chenocolic, and cholic acids are called unconjugated bile salts. You recognize unconjugated bile salts because the carboxyl group is free. If you attach an amino acid to a carboxyl group, then you have a conjugated bile salt.
So, this is an important point. Here is the conjugated bile salt where the pH~ 6.
The conjugation of cholic acid is outlined here.
In the first reaction, the cholic acids carboxyl group, reacts with the sulfhydryl group as H of coenzyme A, forming a thioester bond. CoA synthetase is the enzyme that catalyzes the reaction between Colic Acid and CoA. Remember that CoA Synthetase also catalyzes the formation of the Acyl-CoA derivatives need for fatty acid metabolism or beta oxidation.
Before fatty acids enter the mitochondria, they must be activated by CoA synthetase. CoA synthetase uses ATP as an energy source, the overall reaction is shown here. You have a carboxyl group, CoA, ATP, forming an Acyl CoA, and AMP, PPI, Cholyl CoA, Cholic Acid CoA, and Cholae CoA is ready to react with amino acids. We have seen two examples, taurine and glycine
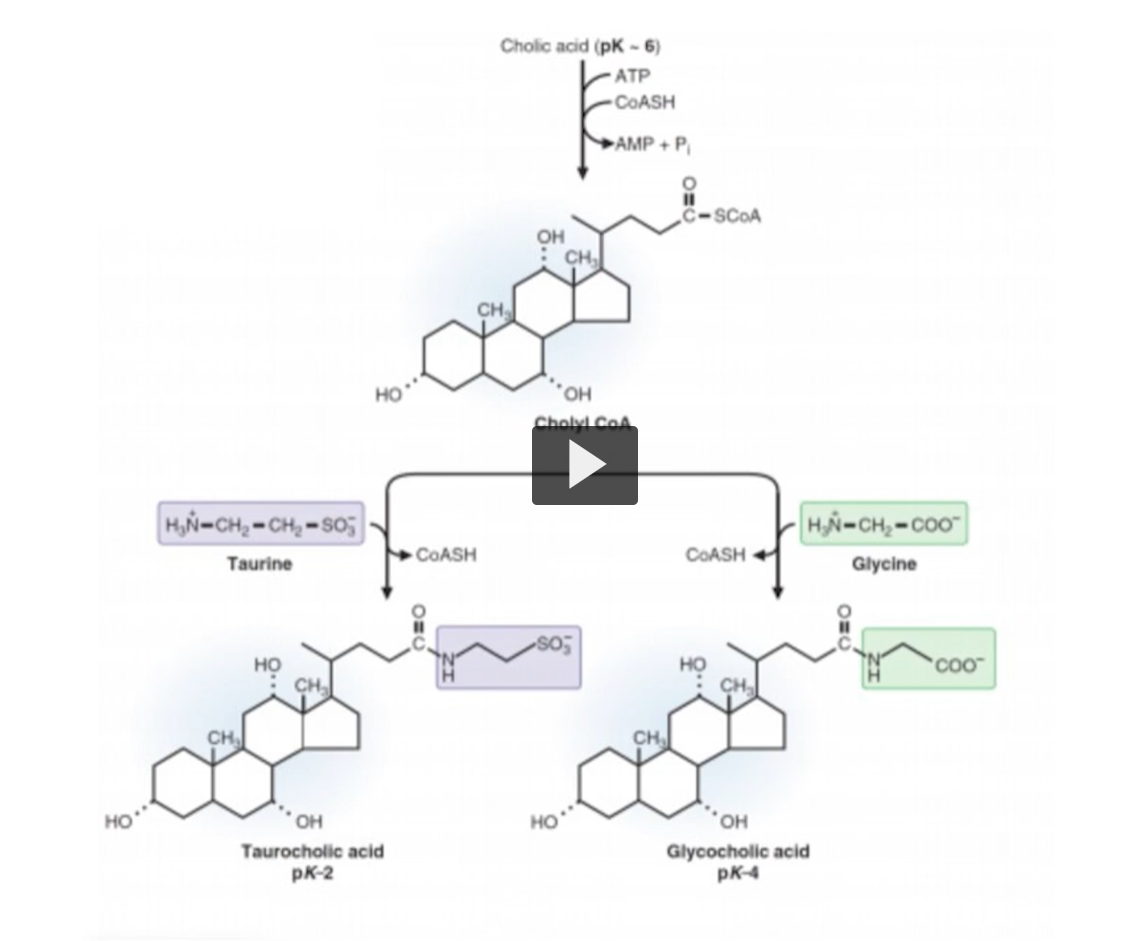
Taurine is an amino acid that is not found among the amino acids used in the genetic code.
Taurine has a simple chemical structure of an amino group and a sulfonic acid group.
Notice that there is NO carboxyl group.
Then there is glycine, which is a smaller amino acid.
Taurine and glycine are two examples of two amino acids reacting with Cholyl CoA
The reaction between choline and taurine produces taurocholic acid. The reaction of Cholyl CoA and glycine produces glycocholic acid.
Remember that you identify conjugated vs unconjugated bile salts by looking at the carboxyl group. Notice that the pKas are two for taurocholic acid and four for glycocholic acid. The low pKa means 100% of these molecules at physiological pH are negatively charged
Bile salts are produced FIRST in an unconjugated form, then you add the amino acids to form the conjugated form.
The conjugated bile salts enter the digestive system to act as detergents, but in the digestive system, the intestinal flora have enzymes that deconjugate the bile salts, meaning that they remove the amino acids in a hydrophilic reaction.
Then they have enzymes that remove OH groups. The effect of removing polar groups is that the remaining non-polar compound could be excreted.
One of the characteristics of bile salts is that they can be re-used 7 times. Every time they are re-used in digestion, approximately 90 to 95% are recovered. 5% of the molecules are not soluble enough because the intestinal flora removed the polar compounds, meaning OH and amino acids.
Let’s take for example, These two at the end here. Which would you predict that would be more soluble. The one with the higher number of OH groups. Primary and second bile salts illustrated here, remember that primary bile salts have OH at a position 7, secondary bile salts lack an OH at position 7 due to intestinal floral activity.
After reabsorption, the bile salts can have amino acids or OH placed, the exception is with OH at position 7. Why is that? Because 7a-hydroxylase is specific for cholesterol, Bile salts are no longer cholesterol.
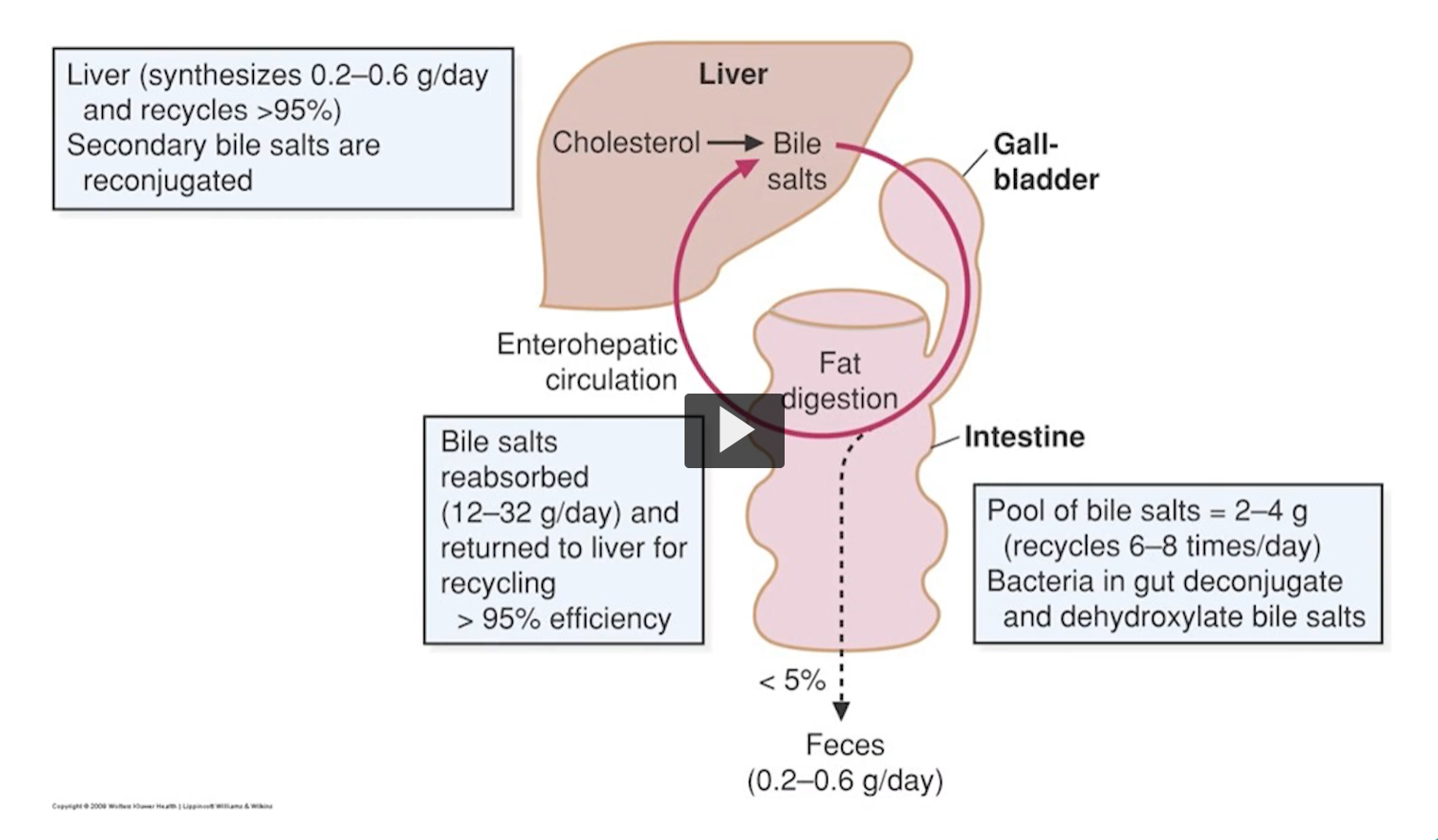
Here is the summary of bile salt synthesis, reabsorption and loss. The liver synthesizes about 0.2 to 0.6 grams of bile salts per day.
Bile salts are secreted from the liver into the bile ducts and stored in the gallbladder between meals.
After eating a meal containing fats, the gallbladder contracts, releasing the bile into the duodenum. After aiding in fat digestion, most bile salts are reabsorbed in the terminal ileum. That is a recycling efficiency of 95%.
Secondary bile salts are reconjugated, meaning that they can have an amino acid removed by the intestinal flora, and then added back. There is 5% loss in every cycle where Bile salts are used
steroid hormones
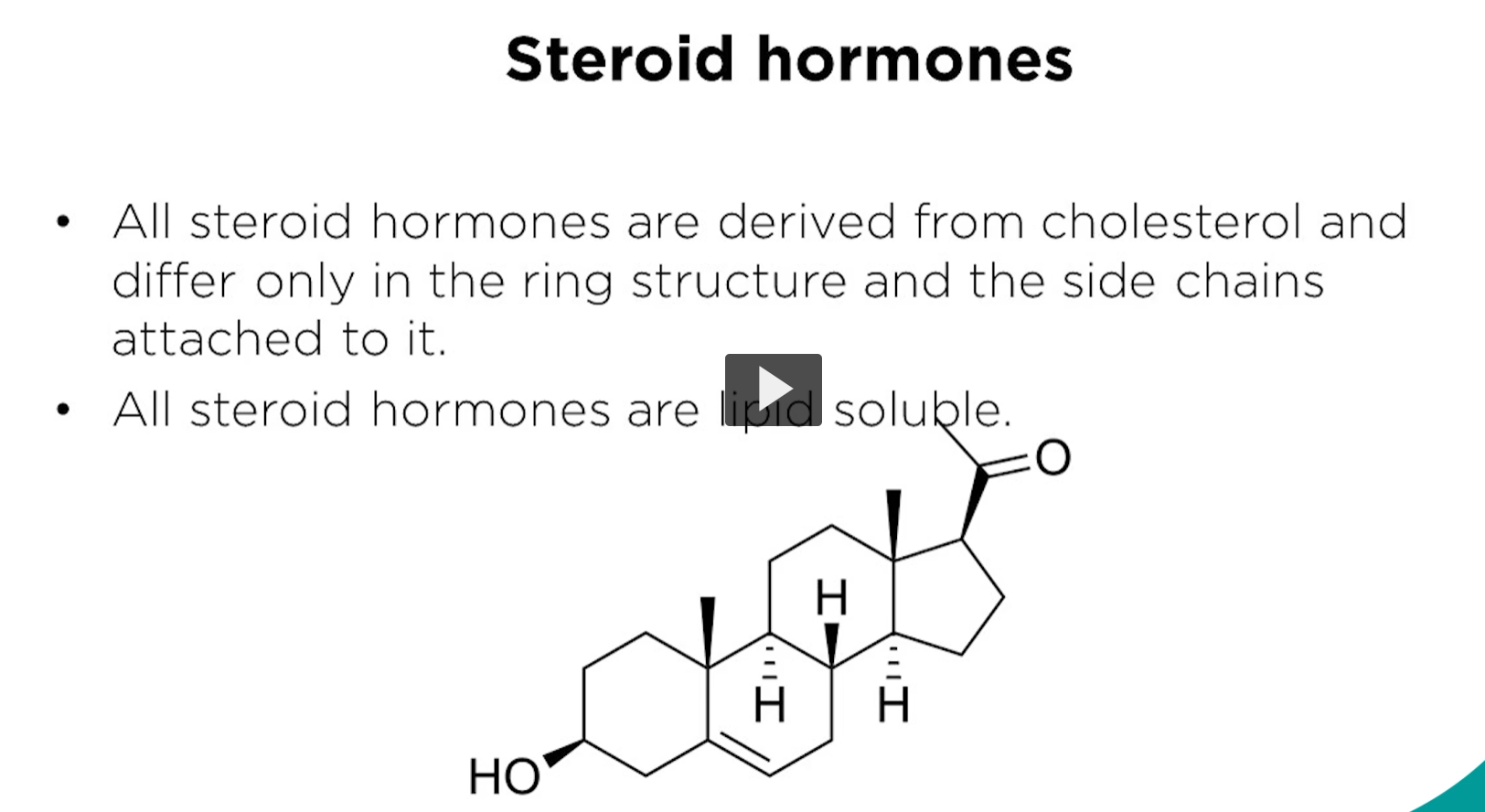
let’s move to steroids. steroids are synthesized from cholesterol in different tissues. Steroids are easy to recognize because they share the typical four ring structure which you observe in cholesterol.

steroid hormones
This is a typical steral structure. The difference in function relate to the chemical groups on the sterol. Remember that the steroid hormones are not soluble in water, but are soluble in organic solvents.
steroids behave like other lipids
steroids behavior like other lipids, let us review the following steroid hormones.
glucocorticoids, we have discussed previously, an example is cortisol, which plays a role in various physiological processes, including metabolism, immune response and stress regulation. Cortisol is an anti-inflammatory steroid that also induces the expression of the gluconeogenic pathway enzymes
mineral corticoid: with aldosterone is an example? Aldosterone plays a crucial role in regulating electrolyte balance, blood pressure, and fluid volume in your body.
Androgens are primarily known for their roles in male sexual development and reproductive function. Androgens also influence both males and females mood, cognition, and behavior.
Estrogens are a group of steroid hormones primary associated with female reproductive function.
Progesterone is a steroid hormone primarily associated with female reproduction function, particularly, during the menstrual cycle and pregnancy.


steroid hormones
in biochemistry one, we studied how lipid soluble hormones, and non-lipid soluble hormones differ in their chemical properties, mechanisms of actions and modes of transportation within the body.
Steroids has lipid soluble hormones are released from the cells as they are synthesized and are not packaged.
Here, we can compare steroids to neurotransmitters. Neurotransmitters are stored in vesicles and are secreted in a quantum package. Here, we can compare steroids to neurotransmitters. Neurotransmitters are stored in vesicles and are secreted in a quantum package.
Steroids are synthesized from cholesterol by enzymes located primarily in the mitochondria and the smooth ER.
steroid hormones
steroids travel through the bloodsystem using binding proteins or lipoproteins.
an example would be corticosteroids, like cortisol, which are transported by globulin. Globulin also carries other steroids, such as testosterone and estradiol.
The difference between steroids and other hormones is that the steroid hormones can be converted to another steroid hormone in the target cell. For example, insulin is secreted as specialized cells and act as insulin in target cells. These steroids can be converted into different steroid molecules in the target cell.

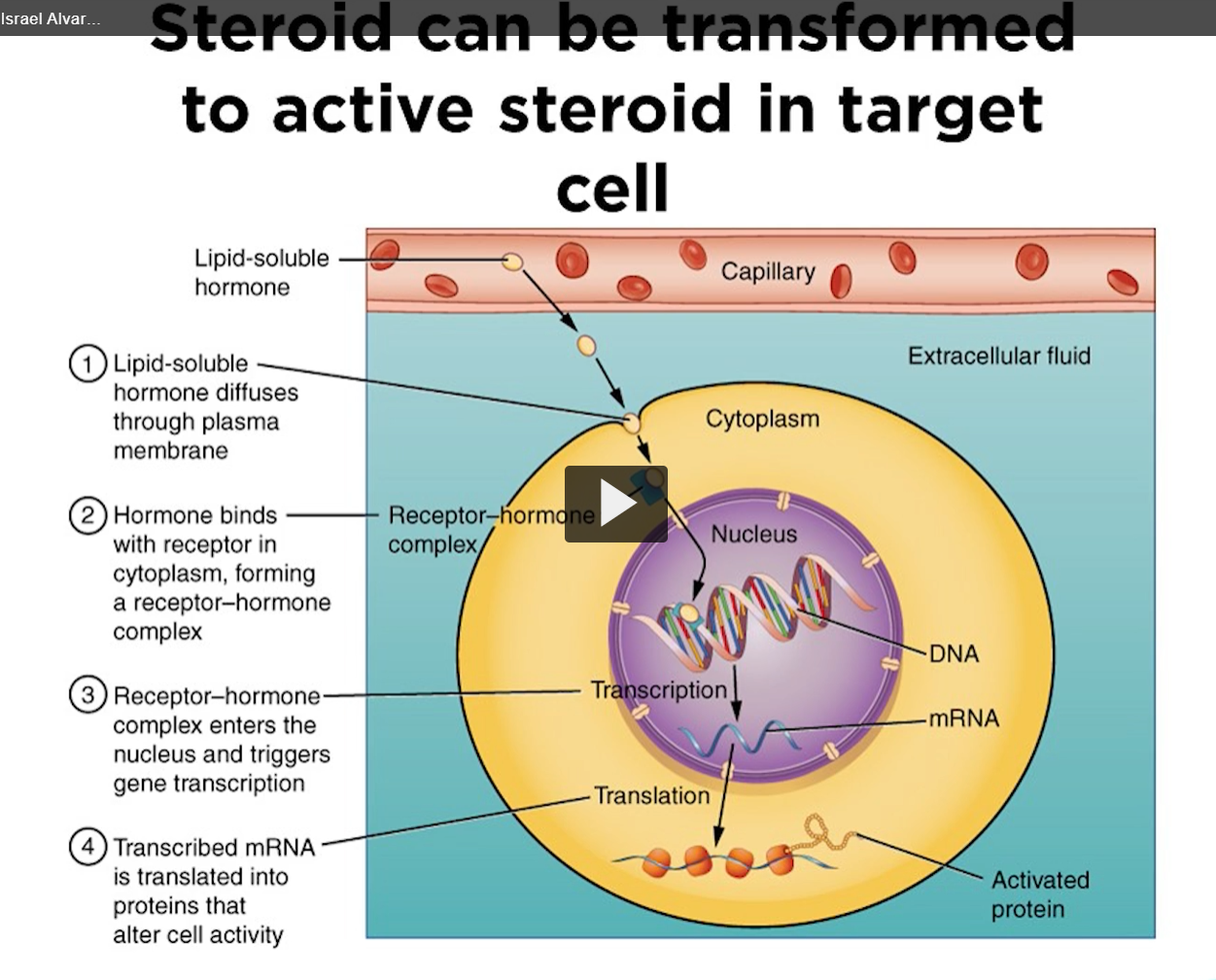
for example, testosterone is transported via binding of lipoprotein to target cells. In one target cell, testosterone is converted to dihydrotestosterone and estradiol in another cell. So, this is the unique aspect of steroid hormones.
steroid hormones
steroids are lipids that can cross the membrane freely, steroids don’t need a permease or an ion channel, or a membrane bound receptor. Steroid receptors are found inside the cell in the cytoplasm.

functions of steroid hormones
The effects of steroids are enormous.
carbohydrate regulation
mineral balance
reproductive function
inflammatory response
stress response
bone metabolism
cardiovascular fitness
behavior
cognition
mood.
source of cholesterol for steroid synthesis
We require cholesterol to synthesize steroids. We have reviewed de novo cholesterol synthesis from acetyl-CoA by condensation. The critical enzyme or pacemaker is HMG CoA Reductase, which you have discussed in previous lectures


sources of cholesterol for steroid synthesis
Cholesterol is also to be obtained from LDL.
LDL in the cell, these are free cholesterols, or cholesterol esters
Cholesterol esters have fatty acids esterified onto the OH group.
LDL can carry large amounts of esterified cholesterol.
Studies that per LDL particle, you can transport 1,500 esterified cholesterol molecules.
The LDL is captured and transported through a receptor. This is called receptor mediated endocytosis.
The LDL receptor allows for everything to be internalized inside the cell. Esterified cholesterol is broken by an esterase.
source of cholesterol for steroid synthesis
This is a summary of the two sources of cholesterol
de novo synthesis of free cholesterol from acetyl-CoA in the fed state conditions
OR free cholesterol from the LDL receptor mediated process.
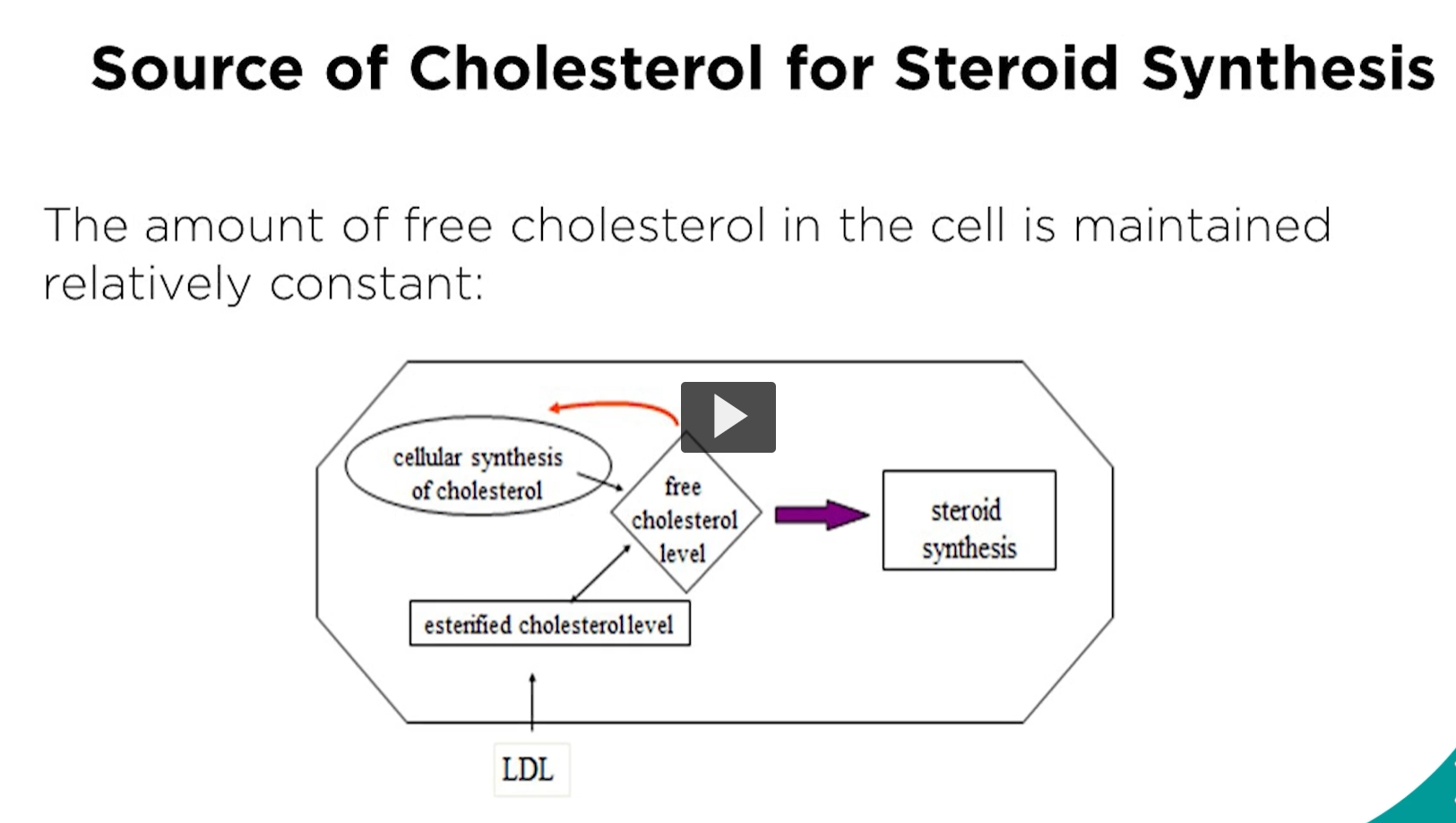

cellular localization of cholesterol metabolism for steroid production
in steroid biosynthesis, the first enzymatic step is conversion of cholesterol into pregnenolone. This is regardless of which steroid is being made.
The enzyme is in the inner mitochondrial membrane.
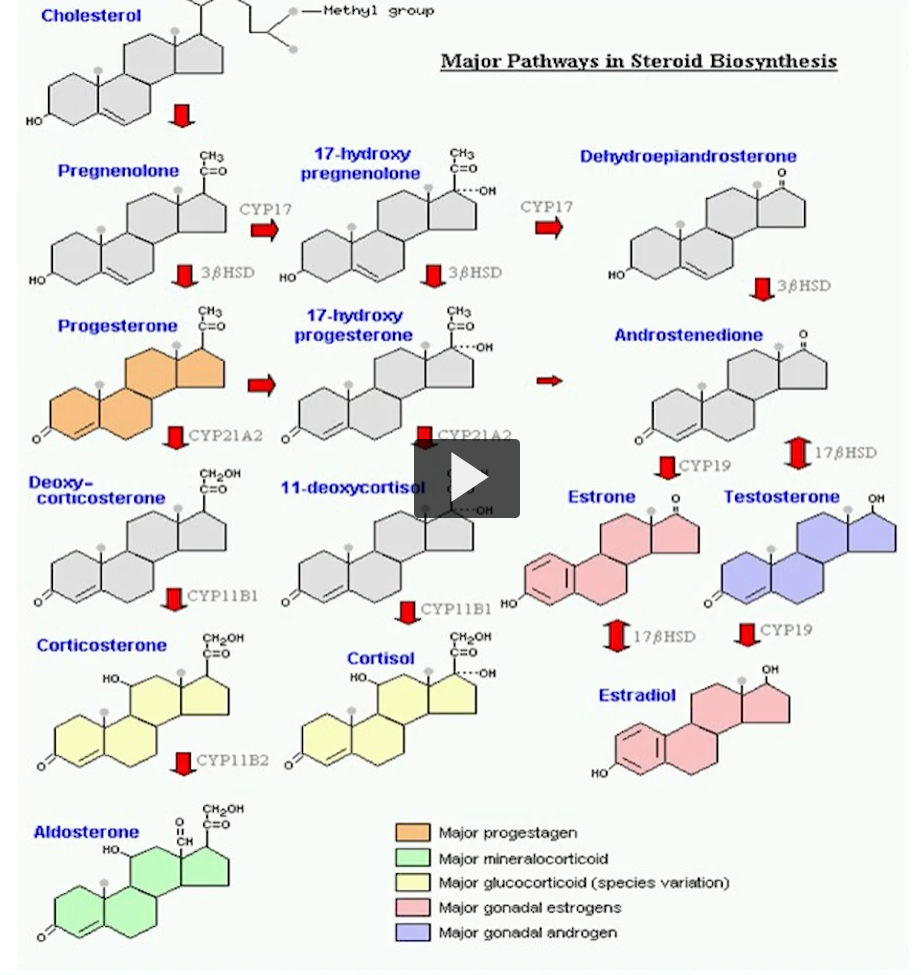
major pathway in steroid biosynthesis
This is the biosynthesis of different steroid molecules.
The initial step of cholesterol is to go into pregnenole, this is the first OBLIGATORY step for what you are going to produce.

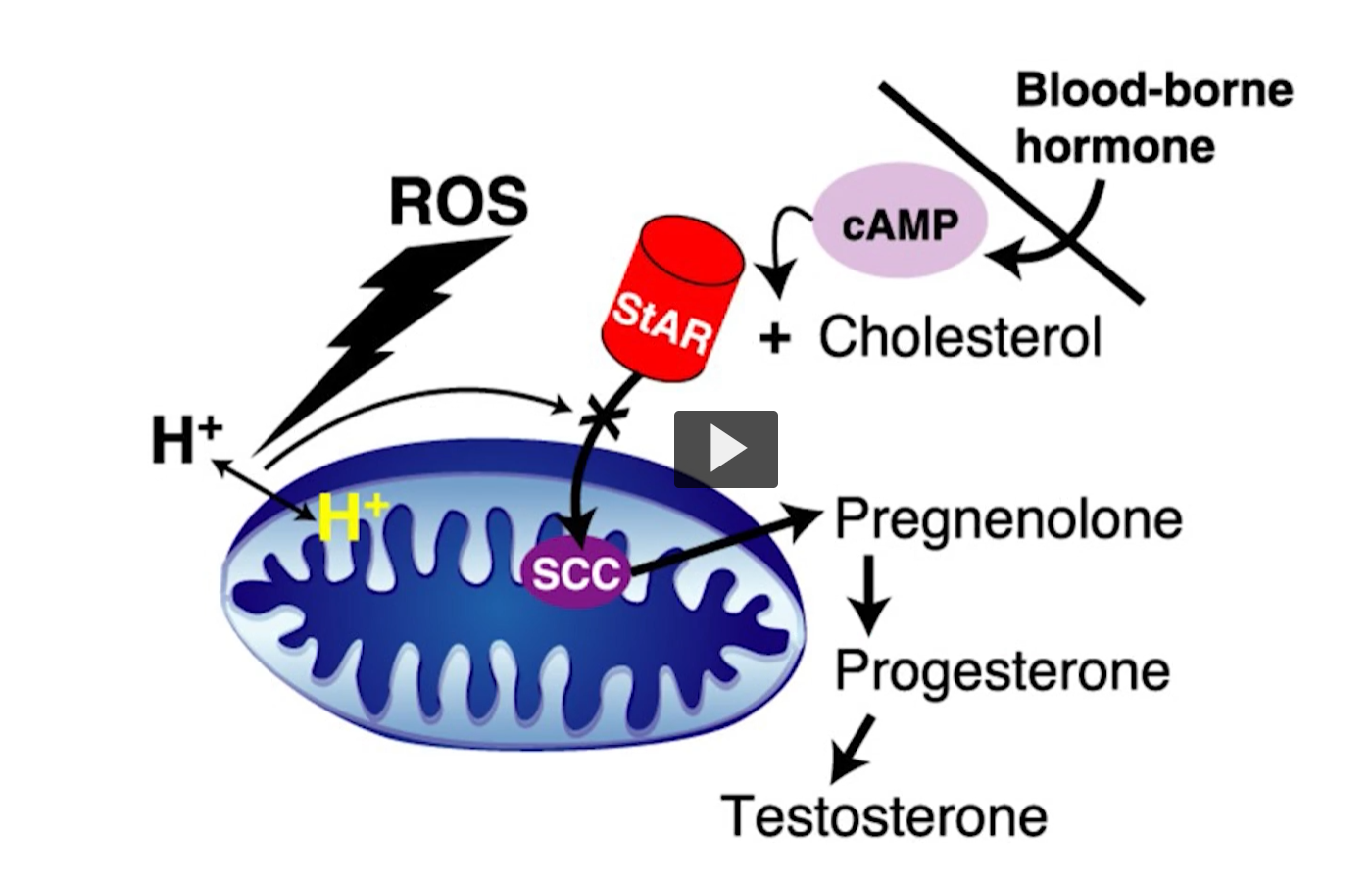
steroid hormone synthesis
All steroid hormones are derived from cholesterol.
The enzyme reactions occur in the mitochondria and the smooth ER. The rate limiting step is entry into the mitochondria for the first reaction. The process of cholesterol getting into the matrix of the mitochondria, the star protein is steroidogenic acute regulatory protein (StAR). This protein plays a crucial role in synthesizing steroid hormones.
Its primary function is to facilitate the transport of cholesterol, a precursor molecule, across the mitochondrial membrane into the site of hormone synthesis.
This is a cartoon. You see cholesterol entering the mitochondria through a star A permease.
Once the cholesterol enters, it is converted into pregnenolone on the first reaction and so on.
This rate limiting step, is the entry of cholesterol into the mitochondria
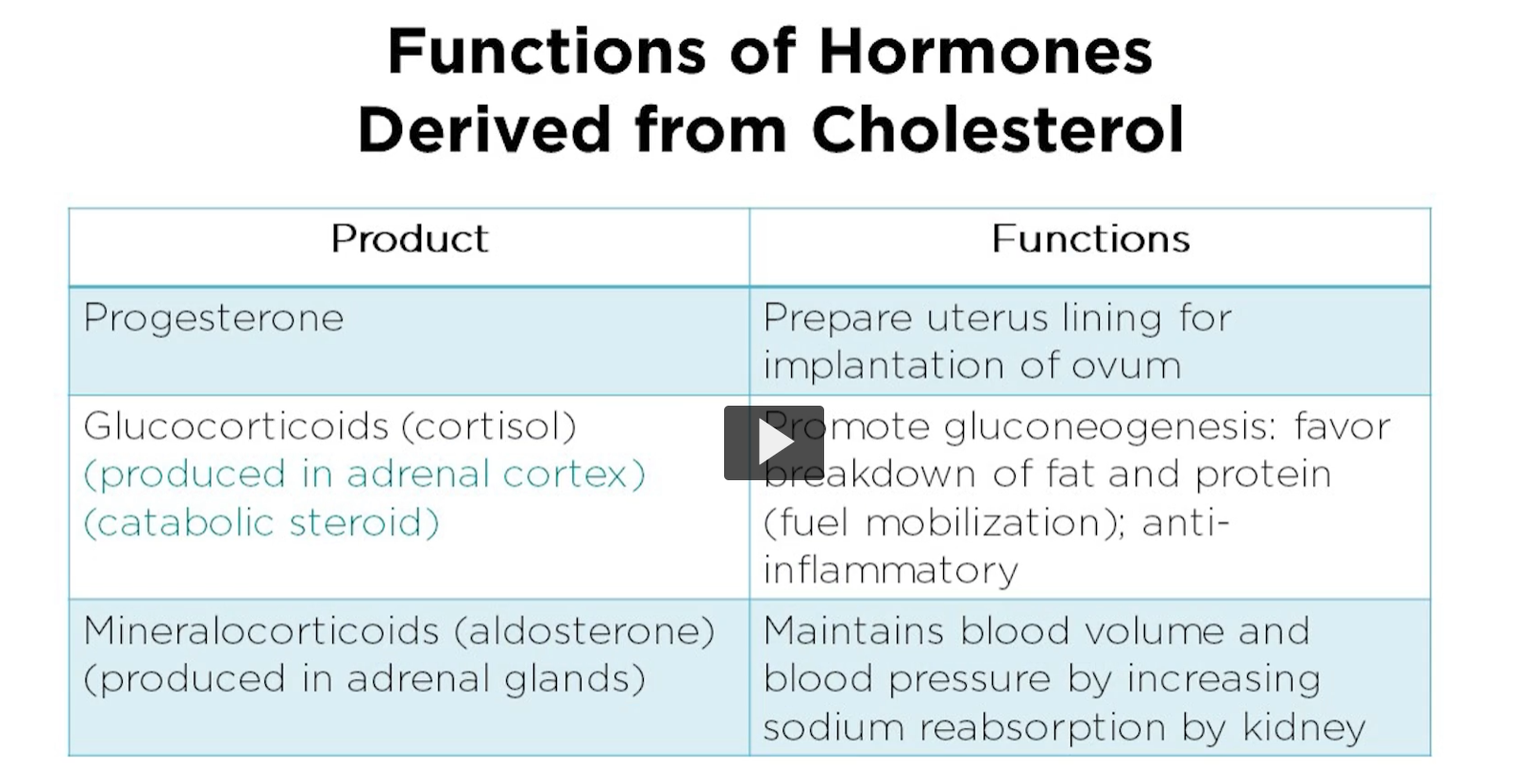
functions of hormones derived from cholesterol
Here we have the function of steroid hormones.
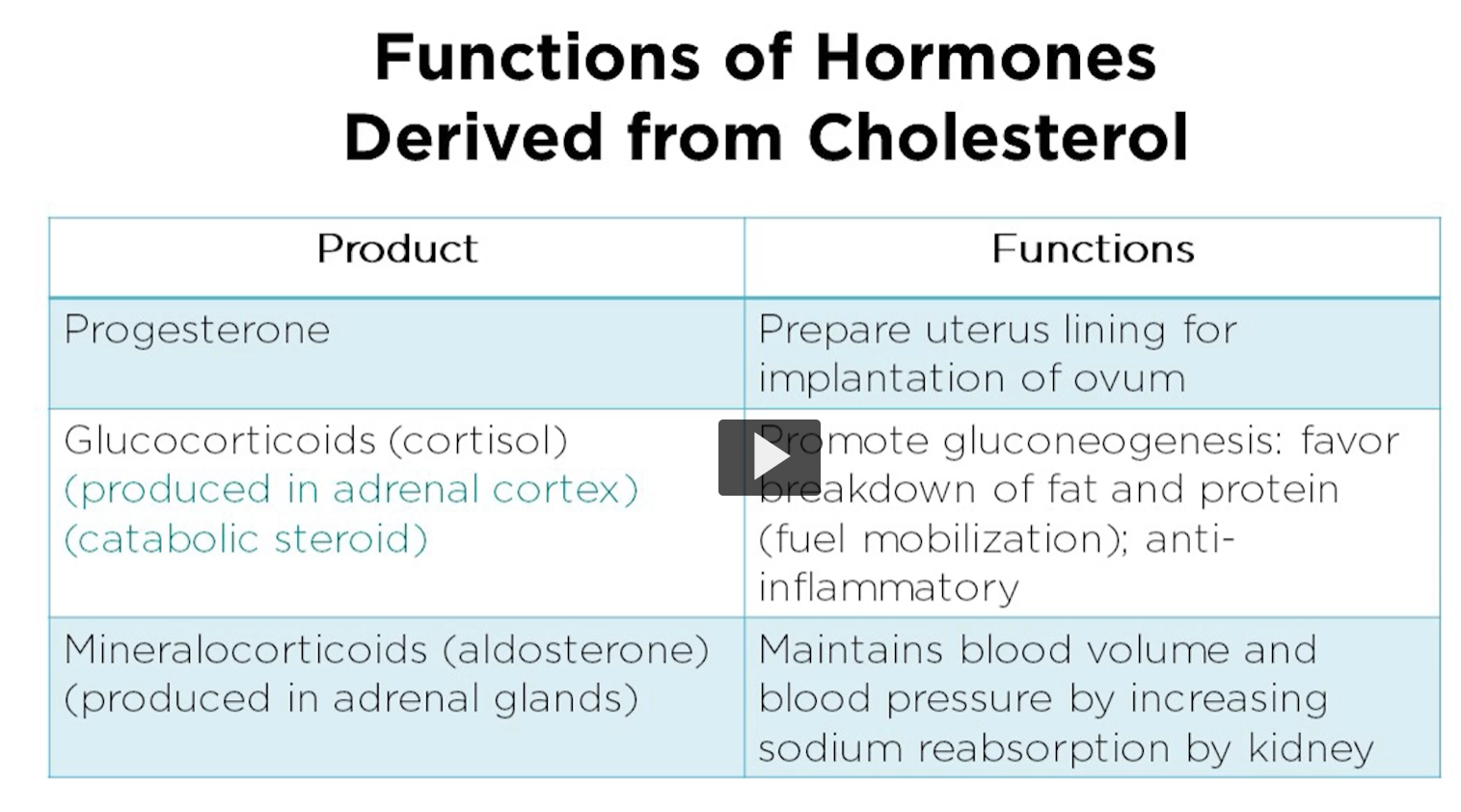
Progesterone plays a crucial role in the female reproductive system. Progesterone helps to prepare the uterus for implantation of fertilized egg by thickening the uterine lining. It also maintains the uterine lining throughout the pregnancy and prevents contractions of uterine muscles, thus supporting the development of the fetus. Additionally, progesterone is involved in the regulation of the menstrual cycle and helps prepare the mammeric glands for lactation.
functions of hormones derived from cholesterol
glucocorticoids are essential for regulating the various metabolic processes in the body.
Cortisol, the primary glucocorticoid, is involved in metabolism of carbohydrates, proteins and fats.
It helps regulate blood sugar levels by promoting gluconeogenesis, from the synthesis of glucose from non-carbohydrate sources, Inhibiting glucose uptake in perilous tissues, and stimulating glycogen synthesis in the liver.
Cortisol also has an anti-inflammatory and immunosuppressive effects, which help the body respond to and regulate the immune system.
Abnormalities in glucocorticoid levels can lead to metabolic disorders, such as Cushing’s syndrome, excess Cortisol, or Addison’s Disease, in deficiency of Cortisol.

functions of hormones derived from cholesterol
mineralocorticoids are primarily involved in regulating electrolyte and fluid imbalance in the body, particularly sodium and potassium levels.
Aldosterone, the primary mineral corticoid, acts on the kidneys to increase the reabsorption of sodium and water and enhance potassium secretion, thereby regulating the blood pressure and blood volume.
It also plays a role in maintaining the acid base balance
functions of hormones derived from cholesterol
Here we have antigens, the strongest is testosterone, which is produced in the testes.
The functional development of male secondary characteristics and bone reabsorption is involved.
Estrogen is produced in the ovaries and is involved in developing female secondary characteristics and is involved in bone resorption.
Vitamin D is not a structurally a steroid, but its behavior in the cell is similar. The way vitamin D reacts with the receptor in the cytoplasm and that complex goes to the nucleus to exert a change in gene expression. That function resembles steroids.
Even though the structure is different in terms of functionality, it is very similar to the way steroids effect gene expression in the cell.
Vitamin D is involved in intestinal calcium absorption which promotes bone formation and prevents phosphate loss through the kidneys.

synthesis of vitamin D
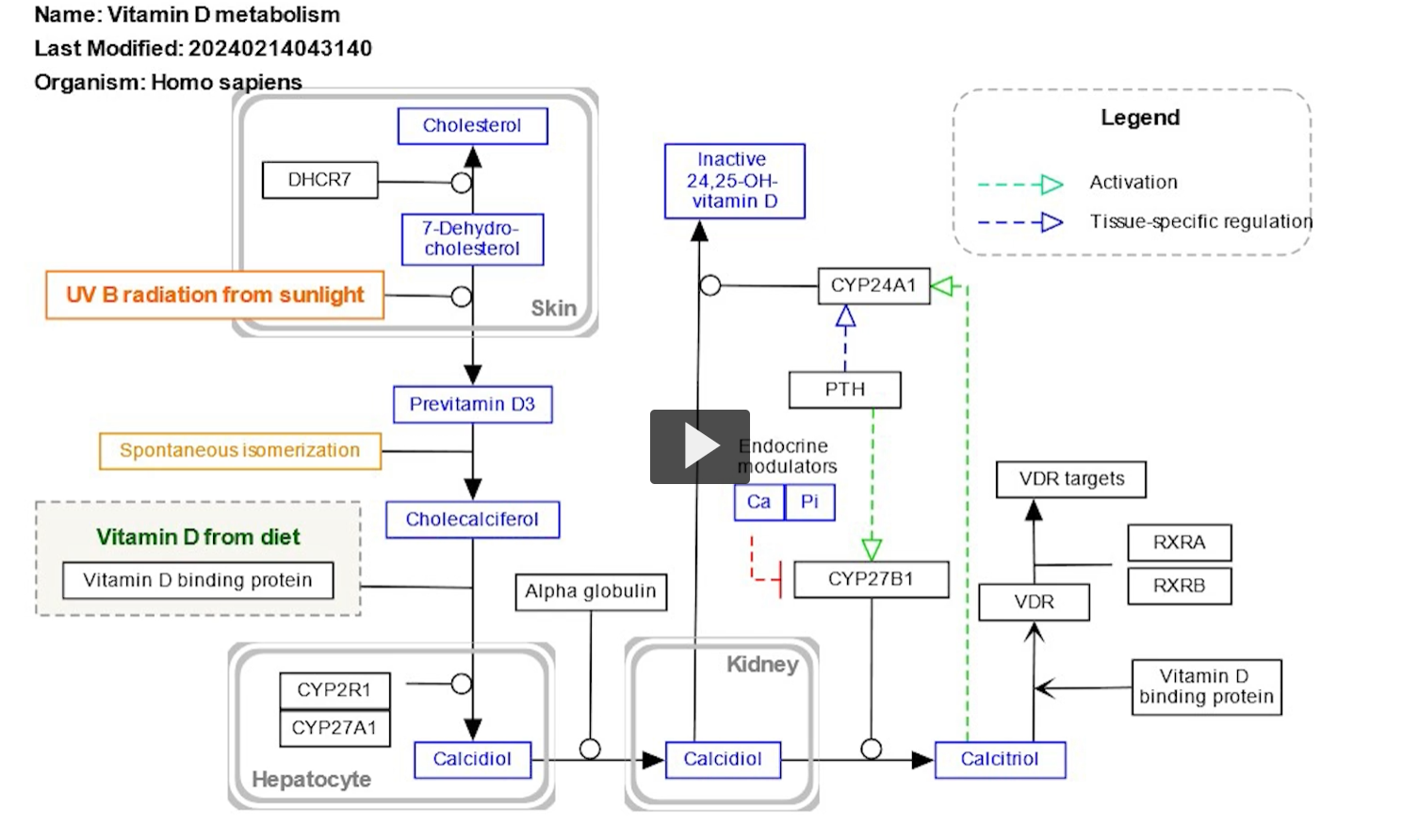
the initial precursor of Vitamin D is pro-vitamin D3 or 7-dehydroxylcholesterol.
7-dehydroxylcholesterol is synthesized from oxidation of cholesterol, resulting in a double bond.
when the skin is exposed to the ultra-violet light, radiation from the sun, particularly UV radiation, triggers a conversion of 7-dehydroxylcholesterol to pre-vitamin D3 and then vitamin D3.
Vitamin D3 binds to vitamin D binding protein DBP for transportation into various tissues.
In the liver, vitamin D3 undergoes hydroxylation, which is the addition a hydroxyl group at the 25th carbon position. This is catalyzed by the enzyme 25 hydroxylase to form 25 hydroxy vitamin D3.
This is also known as calcidiol or calcifederol
25 hydroxy vitamin D is further transported to the kidneys, in the kidneys 25 hydroxy vitamin D undergoes hydroxylation at the first carbon position. This is catalyzed by the enzyme one alpha hydroxylase, also known as CYP27B1. To form the biologically active form of vitamin D, 1-25 dihydroxy D3, also known as calcitriol.
We have seen that vitamin D is different from other vitamins. We have all the enzymes to synthesize vitamin D, which is not true for other vitamins. We must obtain other vitamins from our nutrition.