topic 8 - control of gene expression
1/110
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
111 Terms
What is a mutation?
A change in the DNA sequence
What are the 6 types of mutation?
Substitution
Deletion
Addition
Duplication
Inversion
Translocation
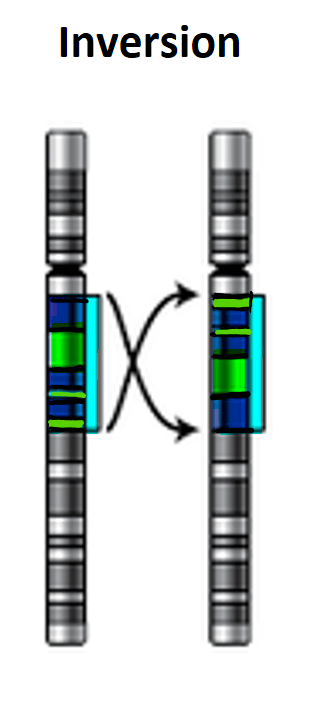
What is an inversion mutation?
Where a section of DNA detaches from the sequence and reattaches but the sequence of DNA bases in the region is inverted. This means a completely new set of amino acids will be coded for in the inverted region so the whole protein will be effected.
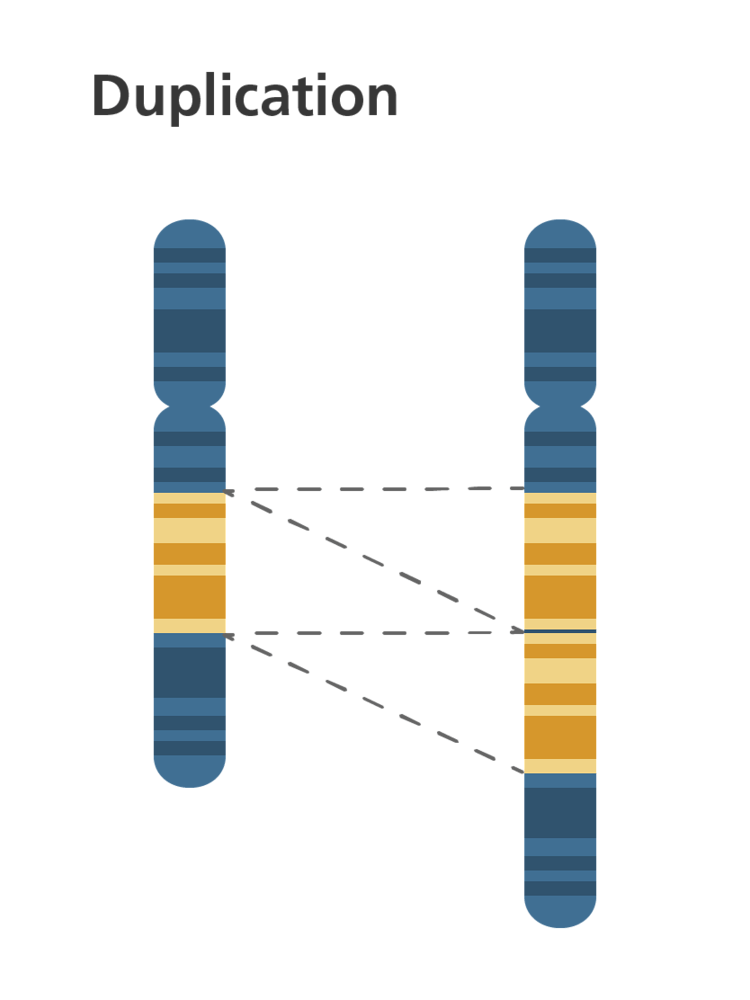
What is a duplication mutation?
When a base or multiple bases in a DNA base sequence are duplicated at least once causing a frame shift to the right. From that point onwards a completely new set of amino acids will be coded for.
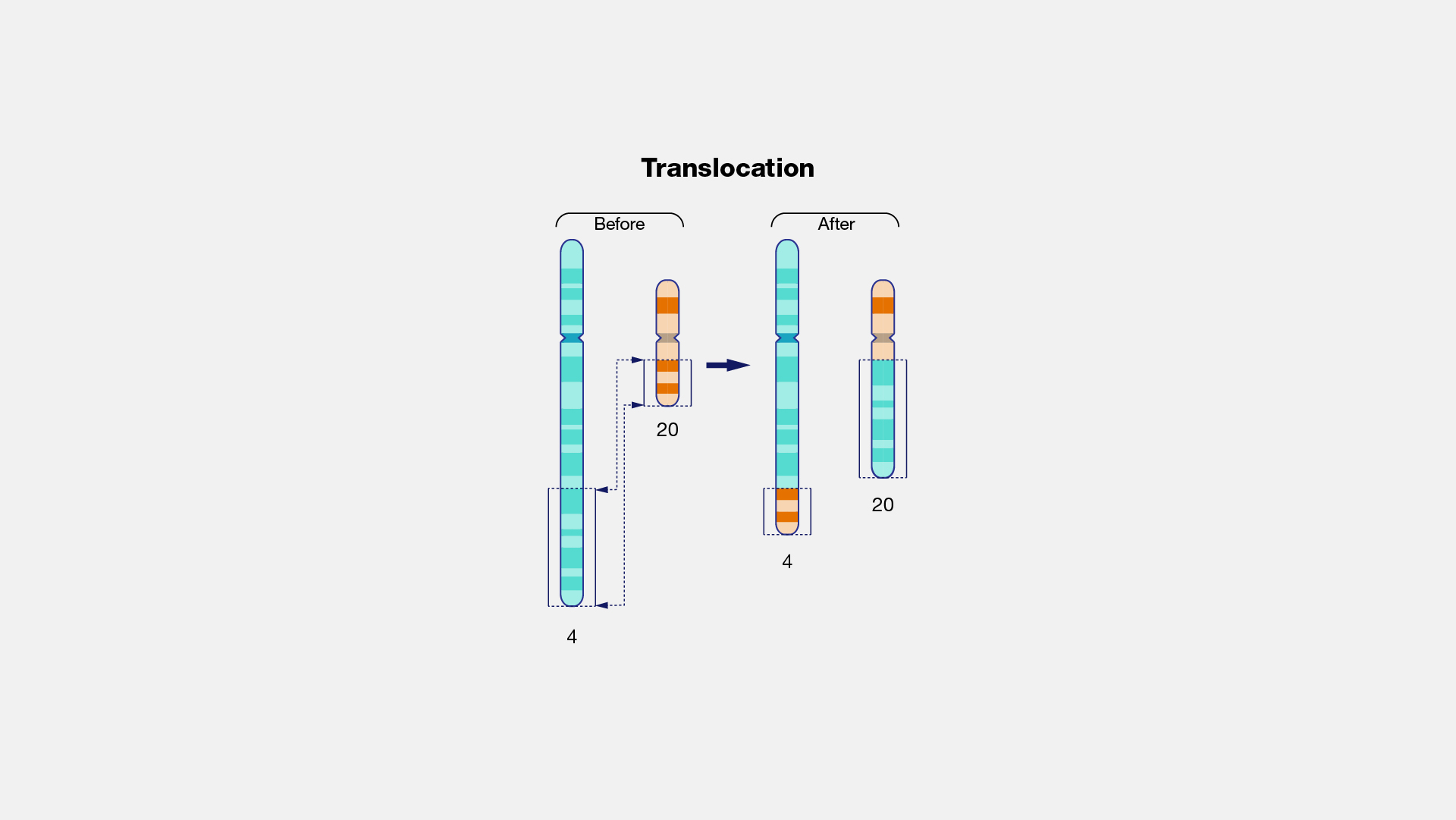
What is a translocation mutation?
When a region of DNA on one chromosome detaches and reatached to a different chromosome or a different area of the same chromosome. This will significantly alter both chromosomes involved so the whole phenotype is likely to be affected.
What are mutagenic agents?
Things that increase the rate of mutations - you can have physical mutagens like ionising radiation e.g. UV, chemical mutagens aka carcinogens, or biological agents like HPV.
How can mutagenic agents increase the rate of mutation? ( 3 ways)
Acting as a base- hemicals called base analogs can substitute for a base
Altering bases - some chemicals can delete or alter bases
Changing the structure of DNA - some types of radiation can change the structure of DNA
What are the different ways a mutation can effect the gene & protein produced?
Silent mutation - (usually only substitution) a mutation occurs but the triplet still codes for the same amino acid → there is no effect on the polypeptide
Misense mutation - a mutation which results in a different amino acid being coded for → if only one triplet is affected only that amino acid will be changed so the polypeptide is likely to be the same
Nonsense mutation - a stop codon is coded for → truncated polypeptide as transcription stops part way through a gene
Why might some mutations have no affect on the gene & so protein coded for?
They are silent or point misense mutations
The mutation happens in a non-coding region/intron
The change in the amino acid has no effect on the tertiary structure of the protein or the activity of the protein
The mutation occurs in a recessive allele in a heterozygous individual
What is a hereditary mutation?
When a gameate containing a mutation for a type of cancer or genetic disorder is fertilised then it will be present in new foetus
What are acquired mutations ?
Mutations that occur in individual cells after fertilisation
How can uncontrolled division develop?
Mutations to genes controlling cell division - mutations to these genes will result in non-functional proteins leading to too much cell division
Epigenetic changes to the expression of genes controlling cell division - this could be inhibited expression of a gene which halts the cell cycle etc or increased expression of a gene which starts cell division etc
Increased oestrogen concentration - or generally increased transcription factors either increasing transcription of genes encouraging cell division or inhibition of genes stopping the cell cycle
What is a benign tumour?
A non-cancerous tumour meaning there is a clump of cells undergoing uncontrolled cell division. But they will not metastsise aka spread to other tissue so the effect is localised.
Why are benign tumours easier to treat than maligant tumours?
This is because the tumour cells produce adhesive molecules which stick the cells together and to tissue, some also have capsules around them. These tumours also have a slower rate of growth than malignant tumours so are more easily able to be removed in surgery.
However depending on where the tumour is e.g. the brain, benign tumour removal can also be life threatening.
What is a maligant tumour?
A cancerous tumour. They don’t have sticky molecules or a capsule so can metasasise. They also have a faster rate of growth than benign tumours.
How is a maligant tumour different to normal body cells & benign tumour cells?
The cells nuclei become large and the cell can become unspecialised again
The tumour can grow projections and develop its own blood supply
Removal of the tumour needs supplmentary treatment - chemotherapy & radiotherapy - but reccurrence is still more likely
What are six ways in which tumour cells differ from normal cells?
Larger nucleus
Darker nucleus sometimes more than one
Irregular shape
Don't produce all proteins needed to function
Different antigens on surface
Don't respond to growth regulated processes
Divide by mitosis more frequently
How are benign & maligant tumours different - summary?
Benign -
Not cancerous
Enscapsulated so can’t metastasise
Nuclei can’t change
Can’t grow their projections and their own blood supply
Easier to remove & treat
Recurrence is less likely
Maligant -
Cancerous
Not enscapsulated so can’t metastasise
Nuclei change leading to cells becoming undifferentiated
Can grow their projections and their own blood supply
Harder to remove & treat
Recurrence is more likely
What are the 2 types of gene that control cell division?
Tumour suppressor genes
Photo-oncogenes
What do tumour suppressor genes do?
Slow cell division by producing proteins that stop cells dividing or cause them to self destruct
What do proto-oncogenes do?
Stimulate cell division by producing proteins that make cells divide
What is a mutated protooncogene called?
Oncogene
What are 2 causes of tumour growth?
Abnormal methylation of dna
Increased exposure to oestrogen
Why is methylation important?
It regulates gene expression - controls whether gene is transcribed and translated when happening normally
What does hypermethylation of tumour suppressor genes cause?
Genes are not transcribed so proteins are not produced
What does hypermethylation of protooncogenes cause?
Causes them to act as oncogenes
What are the 3 theories as to why increased exposure to oestrogen can increase risk of breast cancer?
can stimulate certain breast cells to divide and replicate
if cells do become cancerous rapid replication could be further assisted by it
is able to introduce mutations directly into the dna of breast cells
What are the risk factors for cancer?
Genetic factors - inherited alleles
Environmental- radiation,smoking
What would be useful for curing and treating cancer?
Knowing which specific mutations cause which cancers so treatment can be made to target them specifically
What are stem cells?
Undifferentiated cells capable of dividing indefinitely and differentiating into one or more types of specialised cells, depending on its potency.
What are the different potencies of stem cells?
Totipotent - can mature into any type of body cell
Pleuripotent - can mature into any type of body cell except placental cells
Multipotent - can differentiate into a few different types of cell
Unipotent - can only differentiate into one type of cell
Where are different potencies of stem cells found?
Totipotent and Pleuripotent cells are only found in the embryo. Totipotent cells only occur for a very short period of time in early mamallian embryos.
Multipotent and Unipotent cells are found in mature mammals (and the embryo).
What can pleuripotent cells be used for?
They can be used in stem cell therapies/research as they can divide & differentiate into any cell type (-placenta). However there are issues surrounding the use of embryos to extract pleuripotent cells - need to create a theraputic clone of oneself and the embryo must be destroyed afterwards.
What are some examples of multipotent and unipotent cells?
Multipotent - bone marrow cells as they can divide into the multiple types of blood cells e.g. RBCs vs WBCs
Unipotent - cardiomyctes are unipotent stem cells as they can divide and differentiate into only cardiomyctes producing new heart muscle tissue but small amounts
Why and how do stem cells become specialised?
They only transcribe and translate part of the DNA - some genes expressed and other turned off in different conditions so they can now only produce a certain set of proteins and cell structures specific to a cell type.
E.g. skin stem cells differentiate into skin cells which can produce melanin but not haemoglobin and they don’t have sarcomeres etc.
What are the 3 sources of stem cells and how are they obtained?
Adult stem cells - from body tissue of adult
Embryonic stem cells - from embryos at early stages of development
Induced pluripotent stem cells (IPS) - created in labs
What are the advantages and disadvantages of using adult stem cells ?
Can be obtained from simple operation
Avoid ethical issues of using embryos
But aren’t as flexible as embryonic cells and can only differentiate into a limited range of cells
What are induced pleuripotent stem cells?
Adult somatic (body) cells (e.g. skin or blood cells) that have been genetically reprogrammed to behave like pluripotent embryonic stem cells. This means many different cells types can be made without the ethical issues of using embryos.
How can induced pluripotent stem cells be made?
Adult body cells (e.g. skin cells) are extracted.
Scientists insert genes into the cells that code for key pluripotency-inducing proteins.
These proteins are called transcription factors and can bind to DNA to initiate transcription of genes that were previously not expressed in the body cell as the had been turned off due to differentiation
The inserted genes reprogram the adult cell, causing transcription of genes that are active in embryonic stem cells, switching on gene expression.
The cell returns to a pluripotent state, forming an iPS cell.
How are stem cells currently used in medicine?
In bone marrow transplants - contains stem cells which make healthy new blood cells for patient with leukaemia and lymphoma
What are some diseases that stem cells could be used as treatment for in the future? How?
Spinal cord injuries - replace damaged nerve tissue
Heart disease/damage - replace damaged heart tissue
Bladder conditions- grow new bladder
Respiratory conditions- windpipes can be stripped down to collagen structure and new stem cells could generate new tissue for them
Organ transplants - organs can be grown
What is the control of gene expression?
The control over transcription and translation in protein synthesis which effects whether the protein coded by a gene is converted made and in what quantities.
What are the different processes which affect gene expression?
Effect transcription:
Methylation of DNA
Acetylation of histones
Effect translation/ post transcriptional edits
miRNA
siRNA
→ both are examples of RNAi/ RNA interference
What is transcription controlled by?
Transcription factors - proteins that are able to bind to specific DNA sequences (as their 3D structure is complementary).
Transcription factors tend to work on specific families of genes and are needed for RNA polymerase to bind to the DNA.
Where do transcription factors bind onto the DNA?
They bind onto promoter regions.
These are regions of DNA upstream of a gene which are non-coding and mostly unique to the gene.
All promoter regions have a TATA box where RNA polymerase binds to.

What are the different types of transcription factors?
Activators - increase (ease of) transcription by helping RNA polymerase to bind
Repressors - inhibit (ease of) transcription by blocking RNA polymerase binding
What is oestrogen an example of in gene expression?
Oestrogen is a transcriptional activator so it binds onto an activator transcription factor but doesn’t actually bind to the DNA itself.
This is because its a steriod hormone & so lipid not a protein.
How does oestrogen work?

Oestrogen diffuses into cell
It can then bind to the transcription factor ERa which is held in a protein complex
Its inactive without oestrogen present
Once oestrogen binds the ERa & oestrogen can move out of the protein complex
They/ERa can bind to the DNA at the right promoter
A cofactor can then bind & the complex formed can activate transcription
What is epigenetics?
The study of heritable changes in gene expression without changes to the DNA base sequence itself.
How does epigenetics effect gene expression?
Epigenetics involves chemical changes to the DNA and histones effecting how easily RNA polymerase can access a gene.
This means that transcription can be inhibited or increased depending on what changes have occured.
What is the epigenome?
All of the chemical modification made to the DNA and histones.
It is inherited but also changes due to environmental factors which means twins with the same inherited epigenome will have their epigenomes change in different ways.
What environmental factors can affect epigenetics/the epigenome?
Smoking, stress, exercise and diet
Internal signalling from the body's cells can also cause modifications to occur
What are the two processes involved in epigenetics and how are they different?
Methylation:
Effects the DNA not the histone proteins
Inhibits transcription
Isn’t dynamic - once methylation has occured it can’t really/ usually isn’t reversed - much more permanent
Acetylation
Effects the histone proteins not the DNA
Increases transcription
Is a dynamic change - acetylation and deacetylation both occur readily
What happens in methylation?
Methyl groups are added onto the DNA at the cytosine base on a nucleotide
This is carried out by enzymes called DNA MethylTransferase/DNMT
This mostly occurs at the GC island which is a region of DNA in the promoter region but upstream of the TATA box
It contains many cytosines (& guanines)
How does increased methylation affect gene expression?
Methylation attracts proteins to the DNA which cause the histones to pack closer together. This makes the promoter less accessible so transcription factors & RNA polymerase as less able to bind.
This means the methylated gene is expressed less.
What is the structure of the histones?
Histones are made up of 2 layers of 4 types of proteins - total = 8 proteins.
Each of the histone proteins has an amino acid tail which can be modified by attaching groups onto them etc.
What happens in acetylation?
Acetyl groups are added to the histone proteins on their tails by acetyl transferase
This causes the positive charge on the histones to be reduced
So there is less attraction between the positive histones and negative DNA
How does increased acetylation affect gene expression?
Acetylation results in less attraction between the histones & DNA so the DNA doesn’t wind around as tightly. This means the DNA is more exposed so the promoters are more accessible.
Transcription factors & RNA polymerase can bind more easily so the gene is expressed more.
How can drugs be used to treat diseases that are caused by increased methylation and decreased acetylation?
drugs can be used to stop dna methylation - treats disease caused by this
HDAC inhibitor drugs can inhibit the enzyme responsible for decreased acetylation treating the diesease
What is the problem with using drugs to counteract epigenetic changes?
They need to be really specific or they could damage normal cells which could turn cancerous
What is RNAi and how does it affect gene expression?
RNA interference occurs after a pre-mRNA strand has been made from template DNA and is where edits etc are made to the pre-mRNA. Splicing is an example.
RNAi also affects gene expression as changes can be made or the mRNA can be destroyed at this stage to reduce gene expression.
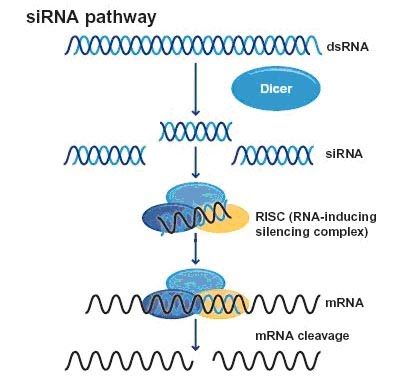
What is siRNA?
Short Interfering RNA. It’s a short, double-stranded RNA molecule which can interfere in protein synthesis by stopping the translation of an mRNA strand.
How does siRNA work?
siRNA is produced from double-stranded RNA in the cell or introduced synthetically.
It binds to a protein complex called the RNA-induced silencing complex.
The siRNA is separated into single strands, and the one strand remains attached to the protein complex.
This strand is complementary to a specific mRNA molecule.
siRNA guides the protein complex to the mRNA and binds to it via complementary base pairing.
An enzyme in the protein complex cuts the mRNA, breaking it into pieces.
The mRNA is degraded, so translation cannot occur, and the gene is effectively silenced.
What is miRNA?
Micro RNA are small RNA molecules which act in protein synthesis post-transcription so the mRNA affected is destroyed/can’t be translated.
How is miRNA different to siRNA?
Instead of cutting mRNA into fragments the miRNA protein complex blocks the translation of the target mRNA then it is moved into processing body where it can be degraded or stored
How does miRNA work?
miRNA isn’t fully complementary to its target mRNA
So it binds to multiple mRNA molecules which often means ribosomes are blocked from attaching or the mRNA strands are degraded
This means the gene is silenced in post-transcription
What happens if epigenetic marks on DNA are not removed between generations?
Expressions of some genes in the offspring can be affected by environmental changes that affected their parents or grandparents
How are twin studies useful?
Genetically identical so any differences in phenotype must be down to environmental factors
What are genome projects?
Where scientists work to determine the entire genome sequence of an organism.
Why is is relatively easy to determine the proteome of bacteria?
Because they don't have non coding dna
Why is it useful to determine the proteome of bacteria?
Useful in medical research and development- eg identifying the protein antigens on the surface of disease causing bacteria and viruses can help the development of vaccines,also allows pathogens to be monitored during outbreak of disease
Why is it difficult to translate a complex organisms genome into their proteome?
Because it's hard to find the bits that code for proteins among the non coding and regulatory DNA
What is pyrosequencing?
Recent technique that can sequence around 400 million bases in a 10 hour period
What does recombinant dna technology allow us to do and what does it involve?
Combine genetic material from different sources - involves transferring a fragment of dna from one organism to another
What is a transgenic organsim?
The organism that contains the transferred dna
What are the 3 methods for making dna fragments ?
Using reverse transcriptase
Using restriction endonuclease enzymes
Using gene machine
Explain how you would make dna fragments using reverse transcriptase
To make complementary dna
mRNA is first isolated from cells and then mixed with free nucleotides and reverse transcriptase. This then uses the mRNA as a template to synthesis new strands of cDNA ( complementary dna)
Explain how you would make dna fragments using restriction endonuclease
Dna sample with recognition sequences that you want is incubated with specific restriction endonuclease which cuts the dna fragment out with hydrolysis reaction and sometimes leave sticky ends which can then be used to bind the fragment to another piece of dna that has complementary sticky ends
What are restriction endonucleases?
Enzymes that recognise specific palindromic sequences and cut the dna at these places
What are palindromic sequences?
Sequences that consist of base pairs that read the same in the opposite direction
What are sticky ends?
Small tails of unpaired bases that are left at each end of a dna fragment
Explain how dna fragments can be made using a gene machine
Sequence that is required is designed
First nucleotide in the sequence is fixed to a bead
Nucleotides are added step by step in the right order including protecting groups which make sure they're joined at right point
Short sections of dna roughly 20 nucleotides long are made broken off and protecting groups removed then joined to others to make longer fragments
What are oligonucleotides?
Short sections of dna roughly 20 nucleotides long
Which two methods are used to amplify dna fragments
In vivo and in vitro cloning
What is in vivo cloning?
Where gene copies are made within a living organism- as the organism grows and divides it replicates the dna creating multiple copies of the gene.
What is in vitro cloning?
Where gene copies are made outside of living organism using the polymerase chain reaction
What are the 3 main steps in in-vivo cloning?
Making recombinant dna
Transforming cells
Identifying transformed cells
Explain the 1st step in in vivo cloning ( making recombinant dna)
Vector dna is isolated and then restriction endonucleases and dna ligase are used to stick the dna fragment and vector dna together by the sticky ends
What is ligation?
Dna ligase is used to join sticky ends of dna fragment to the sticky ends of the vector dna
Explain part 2 of in vivo cloning (transforming cells)
Vector with recombinant dna is used to transfer the gene into host cells
If plasmid vector- host cells have to be persuaded to take in the vector and it's dna
If a bacteriophage vector is used- bacteriophage will infect host bacterium by injecting dna into it - the phage dna then intergrates into the bacterial dna
What are transformed cells?
Host cells that take up the vectors containing the gene of interest
Explain the final stage of in-vivo cloning ( identifying transformed cells)
Marker genes inserted into vectors at same time as gene to be cloned so any transformed cells will contain gene and the marker gene. Host cells are grown on agar plates and each cells divides and replicates its dna creating a colony where transformed cells will produce a colony where all genes contain cloned gene and marker gene.
These identified cloned genes are allowed to grow more producing lots of copies
How can marker genes be used to identify transformed cells? (2)
Antibiotic resistance- host cells are grown on agar plates containing the specific antibiotic so only the transformed genes which have the marker gene will grow and survive
Fluorescence- when agar plates is placed under uv light only transformed genes will fluoresce
What would you need to do if you wanted transformed host cells to produce a protein coded for by the dna fragment?
Make sure that the vector contains specific promoter and terminator regions or else the dna fragment won't be transcribed and a protein won't be made
What method is used in in-vitro cloning?
Polymerase chain reaction (PCR)
Explain the 4 steps in the polymerase chain reaction
Reaction mixture is set up that contains the dna sample free nucleotides primers and DNA polymerase
Dna mixture is heated to 95degrees to break hydrogen bonds and then cooled to between 50-65 so primers can bind to strands
Reaction mixture is heated to 72 so DNA polymerase can work which lines up free nucleotides along template and specific base pairing allows compememnty new strands to form
Two new copies of fragment of dna are formed and one cycle complete- cycle starts again this time all 4strands are used (each cycle doubles dna)
Where can Recombinant DNA technology be used? (3)
Transformed organisms -
Transformed plants
Transformed animals
How can transformed genes be used in agriculture?
Crops can be made more nutritious and can be made more resistant to pests or droughts
How can transformed genes be used in industry's?
Industries use enzymes which can be produced in large quantities for less money
How can transformed genes be used in medicine?
Drugs and vaccines can be produced quickly and cheaply