CARBOHYDRATES - LECTURE 1
1/44
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
45 Terms
glycolysis
glucose → energy
gluconeogenesis
non carbs → glucose
glycogenesis
glucose → glycogen
glycogenolysis
glycogen → glucose
old definition of carbs
compounds with molecular formula Cn(H2O)n
limitations with the old definition of carbs
There were molecules which were very clearly carbs that did not follow the Cn(H2O)n formula as there was an oxygen missing
There were molecules which were not carbs which followed this definition such as acetic acid
how are carbs classified
sugars (saccharides)
non-sugars (polysaccharides)
how are sugars classified
mono, di, oligosaccharides (3-10 units)
how are monosaccharides classified
trioses
tetroses
pentoses
hexoses
what are the 2 types of hexoses - give examples
aldohexose - glucose
ketohexose - fructose
how are disaccharides classified
reducing - lactose, maltose, cellobiose
non reducing - sucrose
how are oligosaccharides classified
trisaccharides
tetrasaccharides
pentasaccharides
how are non sugars classified
homopolysaccharides
heteropolysaccharides
fischer projections
One of the simplest ways to represent carbs is in the straight chain form
Developed by emil fischer
how do fischer projections indicate stereochemistry
Horizontal = wedge i.e. coming out of the page
Vertical = dash i.e. pointing away from the page
how to identify R/S in fischer
Redraw the chiral centre in 3D if needed
Assign CIP priorities
Determine configuration
If lowest priority is on a vertical line - its ok
If lowest priority is on a horizontal line - disregard it, get the R/S configuration - then SWITCH
describe the process of ring closure of glucose in aqueous solution
in water glucose does not remain in the straight chain form
predominantly forms a ring structure due to intramolecular attack
It is a simple nucleophilic attack where hydroxyl on carbon 5 attack the carbonyl carbon of the aldehyde
A hemiacetal is formed and produces a six membered ring
haworth projections - what do they represent
a ring in a flat planar structure
difference between alpha and beta anomers
The anomeric carbon is newly formed and can have OH either
Down -> alpha anomer - this is when the nucleophilic attack comes from the bottom
Up -> beta anomer - this is when the nucleophilic attack comes from the top
what are the two ways substituents can be presented in chair conformations
axial - pointing straight up or down
equatorial - projecting outward roughly in the plane of the ring
which carbon is C6
CHOH group above the ring
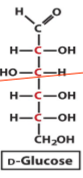
what are D and L isomers in relation to one another
enantiomers
D isomer
when hydroxy furthest away from the carbonyl group is on the right
L isomer
when hydroxy furthest away from the carbonyl group is on the left
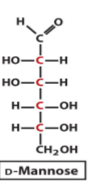
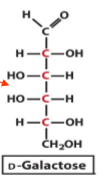
epimer
an epimer is a type of stereoisomer that differs from another molecule at only one specific chiral centre while all other chiral centres are the same
AKA diasteromer
mutarotation
The change in optical rotation due to the interconversion between alpha and beta anomers
it is catalysed by both acids and bases
when does mutarotation occur
when the cyclic form briefly opens to the linear aldehyde/ ketone form then recloses
approx equilibrium for glucose in water
α-D-glucose: 36%
β-D-glucose: 64%
Open-chain form: 0.02%
describe the formation of hemiacetals
A hemiacetal forms when a hydroxyl group attacks a carbonyl group intramolecularly
Hemiacetal carbon has both - OH and -OR groups
Reaction is reversible which allows the interconversion between alpha and beta
why is the beta anomer of glucose more stable
Beta anomer of glucose is more stable due to equatorial OH groups in chair form
how do you form acetals from hemiacetals
Adding extra alcohol in an acid catalysed reaction converts hemiacetals to acetals
steps:
Protonate OH - this then leaves as water
Alcohol attacks - we get an acetal with 2 -OR groups
Acetals are stable and do not form equilibria
Breaking acetals requires a strong acid and heat (boiling)
reducing sugars
can open to form free aldehyde or ketone
aldehydes reduce other compounds and become carbox acids
hemiacetals allow ring opening, enabling this to happen
why is sucrose non-reducing
Both anomeric carbons are full acetals - no free OH
Cannot open to form aldehydes/ ketones
Does not undergo mutarotation
Only hydrolysed under strong acidic boiling conditions
anomeric carbon
carbonyl carbon
anomers
isomers which differ in placement of hydroxyl on C1
slightly different chemical and physical properties
which monosaccharide is a structural isomer but NOT an anomer, epimer, diastereomer or enantiomer of D-glucose
fructose
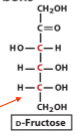
how does fructose form a ring
fructose is a ketose
its ring forms when a hydroxyl group attacks the ketone at C2
what are the 2 possible ring forms of fructose
furanose and pyranose
why is beta-D-fructopyranose the most stable form
Most hydroxy groups are equatorial, minimizing steric strain.
Beta anomer places the anomeric OH on C2 in a favorable orientation.
explain how fructose is a reducing sugar
Under basic conditions, fructose undergoes keto-enol tautomerization to form glucose and mannose intermediates.
These intermediates have an aldehyde group that can be oxidized.
Therefore, fructose gives a positive reducing sugar test
what is the optical rotation of fructose and what effects it
Fructose has a negative specific rotation, which changes slightly depending on which ring form predominates.
structure of D glucose
structure of D mannose
structure of D galactose
structure of D-fructose