PPFB 11- Drying in Pharmaceutical manufacturing
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
Why do we dry in pharmaceuticals?
Key reasons:
❑ stability
❑ flow properties
❑ tablet compression
Examples:
✓ wet granules before compression
✓ APIs after synthesis
What is drying ?
Drying refers to the removal of most or all liquid from a wet pharmaceutical product.
What does drying rely on ?
Evaporation or sublimation of the solvent.
Heat must be supplied efficiently to support this
What are the solvent types and safety ?
The solvent removed is often water, but can also be volatile organic solvents like isopropanol.
Physical drying principles are similar for aqueous and organic solvents.
Organic solvents:
Are recovered (not vented) for environmental and cost reasons.
Require careful handling due to toxicity and flammability concerns.
What is the principle of drying?
The focus is on drying to achieve a ‘dry’ solid state, whether starting from:
A wet solid, or A solution or suspension
What do most drying methods involve?
Exposure to dry, moving air (often heated).
Spray drying for converting liquids/suspensions into dry powders in one step.
What is residual moisture considerations ?
Dried materials usually still contain some water – they are not completely dry.
Moisture content varies with:
Temperature and humidity of the surrounding air
What is the moisture content?
Moisture content is defined as the amount of water (in kg) per kg of dry solid.
Example: A moisture content of 0.4 kg/kg = 40% moisture
It reflects how much water is present before and after drying
Total moisture includes all the water in a wet solid :
Free moisture - easily removed by evaporation
Bound moisture - difficult to remove due to chemical or physical interaction with the solid
When does drying stop?
Drying stops when the material reaches equilibrium with the surrounding air.
Depends on:
• temperature
• humidity
• material properties
Drying cannot reduce moisture below this level under normal conditions.
What are the practical implications ?
Dried solids reabsorb water if exposed to humid air — due to
equilibrium behaviour.
Over-drying is unnecessary — drying below the expected EMC under
normal use conditions is wasteful and reversible if relative humidity rises.
Materials should be dried only to the moisture level they will maintain during storage/use.
Why can moisture can be beneficial ?
A small amount of moisture can be beneficial:
Improves powder flow by reducing static charge.
Enhances tablet granule compaction (ideal: 1–2% residual moisture).
Very dry powders may flow poorly or cause issues in manufacturing.
What are the special cases ?
If a product is hydrolytically unstable, it must
be:
Dried to very low residual moisture.
Immediately sealed after drying to prevent moisture uptake.
Example: Use of desiccant packaging or airtight containers.
What are factors to consider when choosing a dry method ?
Key factors to consider when selecting a drying method:
✓ heat sensitivity
✓ solvent type
✓ scale
✓ sterility
✓ energy requirements
What are the major drying methods?
Fluidised-bed drying
vacuum oven drying
microwave drying
Spray dryers for solution/suspension
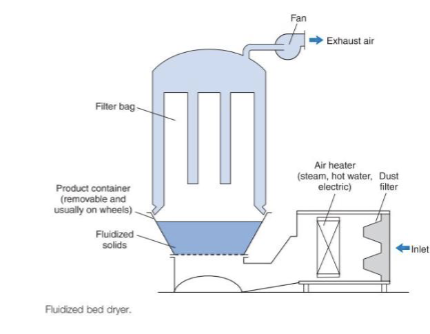
Describe the Fluidised- Bed Drying?
Principle:
Hot air suspends particles → Vigorous mixing leads to fast heat
and moisture transfer → rapid drying
Advantages:
-fast
-uniform drying
-widely used for granules
Limitations:
-fine particle loss
-static charge
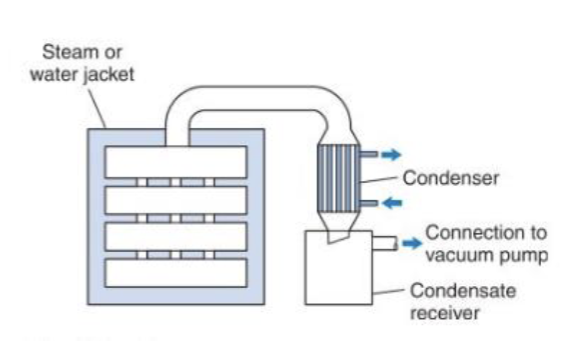
Describe the vacuum oven drying
Principle: The material is heated via conduction on trays under vacuum.
Vacuum lowers the boiling point of water → drying at low temperatures.
Advantages:
-good for heat-sensitive materials
-prevents oxidation
Limitations:
-batch process
-slower
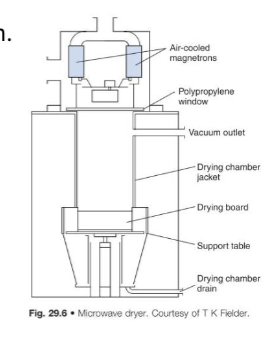
Describe microwave Drying?
Principle:
Uses microwave radiation (typically 2450 MHz) to heat water
molecules inside wet granules, causing rapid internal vaporisation.
Features: Often combined with vacuum (microwave-vacuum
dryer).
Suitable for moisture-sensitive products.
Advantages:
•very rapid drying
•uniform heating
Limitations:
• Small batch sizes (limited industrial scale)
• Microwave shielding required to protect operators
• Equipment is costly
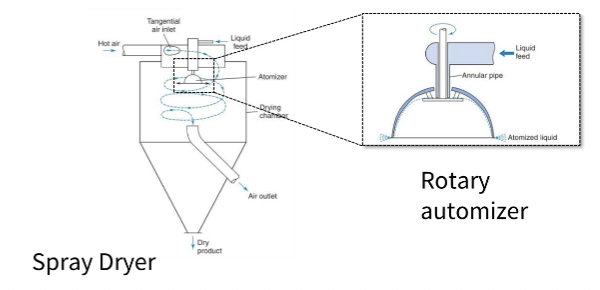
Describe spray dryers for solutions and suspensions?
Liquid solution/suspension is atomised into small droplets and rapidly dried in hot air.
Evaporation is very quick due to the large surface area.
Equipment includes an atomiser (nozzle or rotary), drying chamber, and exhaust air system.
Produces:
•fine powders
•spherical particles
Used for:
•inhalation powders
•amorphous dispersions
What are the advantages of spray dryers ?
Advantages:
• Ideal for heat-sensitive liquids
• Produces fine, uniform powders
• Can operate continuously
• Can generate particles in respirable size (1–7 μm) for inhalation
• Can be dry, sticky, and cohesive materials (with modifications)
• Suitable for sterile/aseptic processing
What are disadvantages of spray dryers?
Disadvantages:
• Bulky and costly equipment
• Complex system with high energy use
• Powder losses may occur if the filters not maintained
• Requires strict control of atomisation and drying parameters
What is the fluidised spray drying ( hybrid mechanism )
•A hybrid of fluidised bed and spray drying
•Allows drying of sticky or high-moisture materials
•Increases flexibility and product consistency
What is freeze drying ( lyophilization) ?
A drying method for extremely heat-sensitive materials such as proteins, blood products, biologicals, vaccines, and injectable products.
Removes water without damaging product integrity or viability.
Suitable for drying materials that would degrade under heat.
How does freeze-drying work?
Freezing: The liquid solution or suspension is frozen.
Primary Drying (Sublimation): Pressure is reduced below
the triple point, causing ice to sublime directly into vapour (solid → vapour).
Secondary Drying: Removes any remaining bound water by
slightly increasing the temperature.
The process avoids the liquid phase, thus preventing heat damage.
What are the advantages of freeze-drying?
• Low-temperature process minimises hydrolysis and chemical degradation.
• Product retains its original shape and structure after drying.
• Highly porous product, which dissolves easily and quickly.
• Minimal oxygen exposure due to vacuum conditions, reducing oxidation risk
What are the disadvantages of freeze-drying?
Disadvantages:
• The final product is very hygroscopic (easily absorbs moisture), so special packaging is not required.
• Freeze drying is slow and expensive, making it suitable only for high-value or heat-sensitive materials.
What are the quality issues with drying ?
Drying Can Affect Product Quality
Example problem:
Solute migration-Solutes move to the granule surface during drying.
Consequences:
•mottled tablets
•drug loss
•content variability
How do you reduce solute migration?
•faster drying
•fluidised bed drying
•higher viscosity granulation fluids
•smaller granules
summary
❑ Drying improves stability and processing
❑ Free water is easy to remove; bound water is harder
❑ Drying stops at the equilibrium moisture content
❑ Method depends on product sensitivity